Parenteral clomipramine has a strong clinical myth around it: bypass the gut, raise parent-drug exposure quickly, and maybe accelerate rescue in severe depression or OCD. A 2026 systematic review found the route advantage was not clearly proven: in depression, 5 RCTs without high risk of bias did not show clear IV or IM superiority over oral clomipramine within 2 weeks, and the poolable estimate was only −1.27 Hamilton Depression Rating Scale points (95% CI: −3.09 to 0.54; I² = 22%).1
Research Highlights
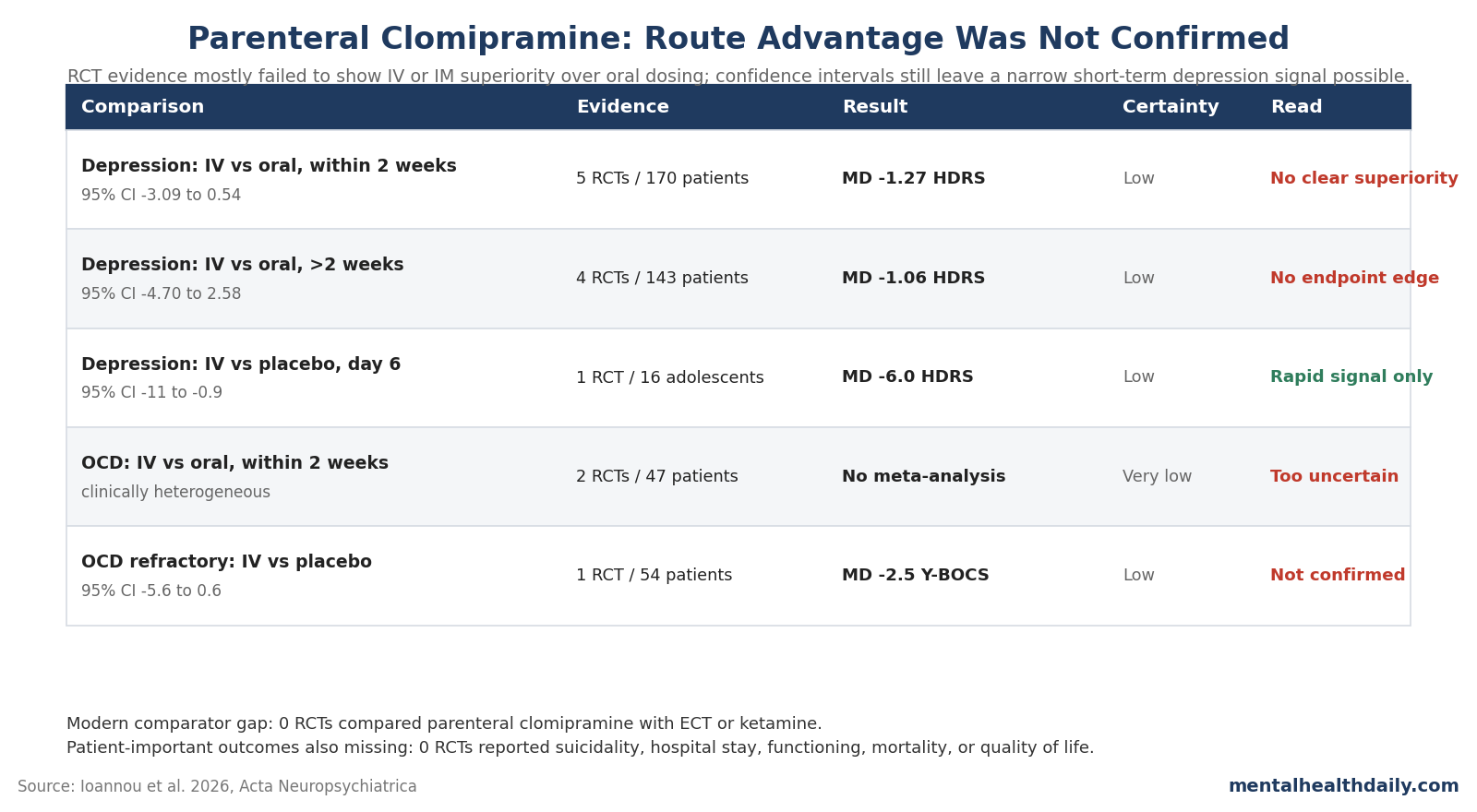
- Depression route advantage was unconfirmed: 5 RCTs with 170 patients compared parenteral vs oral clomipramine within 2 weeks; the 3 poolable RCTs produced a Hamilton Depression Rating Scale mean difference of −1.27 points (95% CI: −3.09 to 0.54), which failed to prove superiority.1
- Endpoint depression results stayed weak: after more than 2 weeks, 4 RCTs with 143 patients again showed no clear IV or IM edge over oral dosing, with pooled change of −1.06 Hamilton points (95% CI: −4.70 to 2.58; I² = 72%).1
- OCD evidence was thinner: 3 RCTs with 101 patients studied parenteral clomipramine for OCD, but the 2 IV vs oral trials were too clinically heterogeneous for meta-analysis, and certainty was rated very low.1
- Placebo-controlled signals were narrower: 1 adolescent depression RCT found a day-6 advantage over placebo (−6.0 Hamilton points; 95% CI: −11 to −0.9), while 1 oral-clomipramine-refractory OCD trial did not confirm superiority over placebo (−2.5 Y-BOCS points; 95% CI: −5.6 to 0.6).1
- Modern comparator evidence is missing: 0 RCTs compared parenteral clomipramine with electroconvulsive therapy (ECT) or ketamine, and 0 RCTs reported suicide attempts, mortality, suicidal ideation, global functioning, hospital stay, or quality of life.1
Clomipramine is a tricyclic antidepressant with unusually strong serotonin reuptake inhibition; it is licensed for obsessive-compulsive disorder (OCD) in the United States and has also been used in severe or treatment-resistant depression. Parenteral clomipramine means clomipramine given outside the gut, usually intravenously (IV) or intramuscularly (IM), rather than swallowed as an oral tablet.
The pharmacology rationale was never silly. Older pharmacokinetic work showed that route and metabolism can shift exposure to clomipramine and its active metabolite, desmethylclomipramine.2,4
The clinical question is whether that route shift produces better patient outcomes, and Ioannou et al. answered that question more skeptically than the old inpatient lore would suggest.1
2026 Review Tested Superiority and Mostly Did Not Find It
Ioannou et al. searched Medline, Embase, the Cochrane Library, and PsycInfo, then screened 4,973 unique publications. Only 14 randomized controlled trials met the review’s PICO question, and those trials were old: publication years ran from 1973 to 2008.1
The review asked a direct route question: whether parenteral clomipramine reduced depression or OCD symptoms more than oral clomipramine, other treatments, or placebo. Its conclusion ran opposite the clinical hypothesis.
The researchers wrote that parenteral clomipramine “may not be superior to oral administration in any respect,” while still acknowledging that a short-term depression benefit could not be excluded.1
That calibrated wording matters. The result is not “IV clomipramine never works.” Both oral and parenteral clomipramine arms often improved.
The failed claim is narrower: giving clomipramine by IV or IM did not reliably beat giving clomipramine by mouth.
Depression Data Leave a Possible Early Signal, Not a Proven IV Advantage
Depression evidence base: 11 RCTs, including 317 patients, studied parenteral clomipramine in depression. But the primary comparison was narrower: parenteral vs oral clomipramine within 2 weeks, using RCTs without high risk of bias.1
Across 5 such RCTs with 170 patients, 3 trials could be pooled. The mean difference in Hamilton Depression Rating Scale (HDRS) change was −1.27 points in favor of parenteral dosing, with a 95% confidence interval from −3.09 to 0.54 and I² = 22%.1
The negative point estimate leans toward IV or IM treatment, but the confidence interval crosses 0, so superiority was not statistically confirmed.
The minimum clinically important difference used by the review was 2 HDRS points. Because the confidence interval extended beyond that threshold, the researchers did not rule out a clinically meaningful short-term benefit.
That is the honest middle: point estimates lean parenteral, proof of route superiority is missing, and the uncertainty is still wide enough to keep a narrow specialist rationale alive.
Longer-term depression results were even less supportive. After more than 2 weeks, 4 RCTs with 143 patients contributed data; the 3 poolable trials gave a mean difference of −1.06 HDRS points (95% CI: −4.70 to 2.58; I² = 72%).1
Higher heterogeneity means those endpoint estimates disagreed substantially about magnitude.
Discontinuation: depression trials comparing parenteral with oral clomipramine produced a pooled Peto odds ratio of 1.05 (95% CI: 0.30 to 3.71; I² = 7%).1 That estimate is basically a wash: the point estimate sits near 1, and the confidence interval is too wide for a useful tolerability advantage claim.
Placebo-Controlled Depression Signal Does Not Answer the Oral-vs-IV Question
The most favorable depression result came from Sallee et al., a double-blind trial in 16 adolescents with major depression. A single IV pulse-load of clomipramine produced a larger day-6 HDRS reduction than saline placebo: −15.0 vs −9.0 points, for a mean difference of −6.0 points (95% CI: −11 to −0.9).6
That trial supports a rapid pharmacologic signal, but it does not prove that IV clomipramine is better than oral clomipramine. The comparator was placebo, the sample was only 16 adolescents, and the review downgraded certainty for serious indirectness and imprecision.1
Pollock et al. is the cleaner route comparison for pulse loading in adult major depression, and it did not favor IV dosing. HAM-D change numerically favored oral clomipramine at day 7 (−9.8 vs −11.4), day 14 (−12.9 vs −13.5), and endpoint (−14.7 vs −15.7).5
A fast-looking placebo result should not be stretched into an IV-over-oral claim when a route-comparison trial points the other way.
OCD Trials Split Between One Early Positive Study and Later Uncertainty
OCD evidence base: the route story should have been most interesting here because clomipramine remains a high-potency serotonergic option for patients who fail selective serotonin reuptake inhibitors. The evidence base was too small to carry that burden: 3 RCTs with 101 patients studied parenteral clomipramine for OCD.1
Koran et al. 1997 produced the strongest early IV signal. In 15 patients with OCD, week-1 Yale-Brown Obsessive Compulsive Scale (Y-BOCS) change was −11.0 after IV pulse loading vs −0.9 after oral pulse loading; week-2 change remained numerically favorable at −10.2 vs −1.7.7
That is a large short-term difference, but it came from a tiny trial.
The later Koran et al. 2006 trial tested a harder population: 32 patients with OCD who had failed 2 serotonin reuptake inhibitors. There, day-6 Y-BOCS change numerically favored oral dosing, not IV dosing (−2.75 vs −4.38), and week-12 percent change was similar at 24% vs 28%.8
Ioannou et al. did not meta-analyze those 2 OCD route trials because they were clinically heterogeneous.1 The practical read is not that the 1997 signal was fake.
It is that the early signal did not generalize cleanly when the question moved into treatment-resistant OCD.
Oral-refractory OCD: Fallon et al. studied 54 patients with OCD refractory to oral clomipramine. IV clomipramine reduced Y-BOCS scores by −3.4 points vs −0.9 with placebo by day 14, but among completers the mean difference was −2.5 points with a 95% CI from −5.6 to 0.6.9
Numerically favorable is not the same as confirmed superiority.
Metabolism Argument Weakened When Outcome Data Were Added
The route argument now has 3 weaker links:
- Bioavailability is not outcome superiority: a more predictable parent-drug exposure can be pharmacologically real without producing better symptom scores.
- Parent-drug dominance may not be the right target: clomipramine strongly blocks serotonin reuptake, while desmethylclomipramine has more norepinephrine reuptake inhibition, so route changes can alter the balance rather than simply make the treatment “stronger.”2
- Recent metabolite data complicate the old story: Vos et al. reported that a higher desmethylclomipramine-to-clomipramine ratio was more often observed in responders than non-responders in major depressive disorder.3
That third point does not prove oral dosing is superior. It does undercut the simple explanation that bypassing first-pass metabolism should automatically help because it pushes the drug profile toward the parent compound.
Ioannou et al. made the same mechanistic connection in the discussion: the clinical data did not behave the way the parent-drug rationale predicted.1
Safety and Logistics Push Oral Treatment Ahead in Most Cases
Parenteral clomipramine inherits the familiar tricyclic antidepressant safety problem and adds procedure burdens. In the depression IV vs oral trials, 3 double-blind RCTs with 121 patients reported infusion-related adverse events.
Because double-dummy designs exposed both groups to procedures, events could occur in either arm; the review counted 5 vs 2 cardiovascular-system events, including 4 vs 2 hypotension events, and 1 vs 1 thrombophlebitis events.1
In OCD, Koran et al. 2006 reported infusion-related adverse events in 2 of 16 IV patients and 2 of 16 oral-comparator patients; all were cardiovascular.8 The numbers are too small for a precise harm estimate, but they are enough to remind readers that IV administration is not a harmless delivery upgrade.
Tricyclics also carry anticholinergic effects, orthostatic hypotension, cardiac-conduction risk, seizure risk at higher exposures, and dangerous overdose toxicity. Those risks are not unique to IV clomipramine, but parenteral administration concentrates treatment in settings where monitoring, infusion access, and protocol discipline become part of the intervention.
Specialist Niche Is Narrower Than the Old Inpatient Lore
The review’s clinical-context numbers explain why this question still matters. A cited Swedish survey found that 31 of 37 responding clinics had used parenteral clomipramine in the prior 3 years, and 29 clinics (78%) considered it an alternative to electroconvulsive therapy (ECT) in certain cases.
At Sahlgrenska University Hospital, 4.1% of adults admitted for at least 6 days with a depressive state from 2016 to 2022 received parenteral clomipramine.1
Those numbers show ongoing clinical use. They also highlight the evidence gap: 0 RCTs compared parenteral clomipramine with ECT or ketamine, the very comparators that matter when clinicians are thinking about rapid treatment in severe depression.1
Oral treatment should remain the default route when patients can take it reliably. Parenteral clomipramine is easier to justify when the goal is monitored inpatient titration, oral administration is not feasible, adherence is impossible to verify, or a specialist program has a specific protocol and cardiac monitoring in place.
It is harder to justify as a general “faster and better” antidepressant or anti-OCD strategy.
Clomipramine Itself Is Not the Failed Claim
The review was about route, not whether clomipramine has pharmacologic value. In the depression trials, both oral and parenteral groups often had clinically meaningful symptom reductions.
In broader antidepressant evidence, clomipramine is active but not necessarily the most acceptable or best-ranked option compared with newer agents.10
For OCD, clomipramine remains an important serotonin reuptake inhibitor when standard selective serotonin reuptake inhibitor treatment fails. The 2026 review simply says the IV or IM route has not earned a broad superiority claim over oral clomipramine, and the evidence is especially shaky in oral-clomipramine-refractory OCD.
The practical verdict is direct: keep clomipramine in the specialist toolkit, but stop treating the parenteral route as if pharmacokinetic elegance has already proven clinical superiority. The route may help selected patients, but the pooled RCT evidence does not support routine IV or IM use for depression or OCD.
Questions About IV Clomipramine Evidence
Did IV or IM clomipramine beat oral clomipramine for depression within 2 weeks?
Not clearly. In the 3 poolable RCTs, the mean difference was −1.27 HDRS points in favor of parenteral dosing, but the 95% CI ran from −3.09 to 0.54, so superiority was not confirmed.1
Does the review rule out a short-term depression benefit?
No. The confidence interval crossed the 2-point HDRS threshold the review used as a minimum clinically important difference, so a meaningful short-term benefit remains possible despite the failed superiority claim.1
Why did the OCD route trials not settle the question?
The 1997 OCD pulse-loading trial strongly favored IV dosing at week 1, but it had only 15 patients; the 2006 treatment-resistant OCD trial numerically favored oral dosing at day 6 and looked similar by week 12.7,8
Is parenteral clomipramine an evidence-based substitute for ECT or ketamine?
Not on RCT evidence. Ioannou et al. found 0 randomized trials comparing parenteral clomipramine with ECT or ketamine, even though those are the clinically relevant rapid-treatment comparators in severe depression.1
When could parenteral clomipramine still make sense?
The narrowest rationale is selected inpatient or specialist care: severe depression or OCD, inability to use oral medication reliably, need for monitored titration, and enough cardiac and infusion monitoring to handle tricyclic risks.
What outcome gaps matter most?
The trials did not report suicide attempt, mortality, suicidal ideation, global functioning, hospital length of stay, or quality of life. Those missing outcomes are exactly the ones that would matter if IV treatment is being defended as a rapid inpatient intervention.1
References
- Ioannou M, Falk Ö, Gustavsson J, et al. Parenteral clomipramine for depression or obsessive-compulsive disorder: a systematic review and meta-analysis. Acta Neuropsychiatrica. 2026;38(e30):1–13. doi:10.1017/neu.2026.10074
- Balant-Gorgia AE, Gex-Fabry M, Balant LP. Clinical pharmacokinetics of clomipramine. Clinical Pharmacokinetics. 1991;20(6):447–462. doi:10.2165/00003088-199120060-00002
- Vos CF, Coenen MJH, Ter Hark SE, et al. A higher desmethylclomipramine to clomipramine ratio predicts more effectiveness and fewer adverse effects in major depressive disorder. European Neuropsychopharmacology. 2024;89:56–57. doi:10.1016/j.euroneuro.2024.09.002
- Evans LE, Bett JH, Cox JR, Dubois JP, Van Hees T. The bioavailability of oral and parenteral chlorimipramine (Anafranil). Progress in Neuro-Psychopharmacology. 1980;4(3):293–302. doi:10.1016/0364-7722(80)90050-8
- Pollock BG, Perel JM, Nathan RS, Kupfer DJ. Acute antidepressant effect following pulse loading with intravenous and oral clomipramine. Archives of General Psychiatry. 1989;46(1):29–35. doi:10.1001/archpsyc.1989.01810010031005
- Sallee FR, Vrindavanam NS, Deas-Nesmith D, Carson SW, Sethuraman G. Pulse intravenous clomipramine for depressed adolescents: Double-blind, controlled trial. American Journal of Psychiatry. 1997;154(5):668–673. doi:10.1176/ajp.154.5.668
- Koran LM, Sallee FR, Pallanti S. Rapid benefit of intravenous pulse loading of clomipramine in obsessive-compulsive disorder. American Journal of Psychiatry. 1997;154(3):396–401. doi:10.1176/ajp.154.3.396
- Koran LM, Aboujaoude E, Ward H, et al. Pulse-loaded intravenous clomipramine in treatment-resistant obsessive-compulsive disorder. Journal of Clinical Psychopharmacology. 2006;26(1):79–83. doi:10.1097/01.jcp.0000195112.24769.b3
- Fallon BA, Liebowitz MR, Campeas R, et al. Intravenous clomipramine for obsessive-compulsive disorder refractory to oral clomipramine: A placebo-controlled study. Archives of General Psychiatry. 1998;55(10):918–924. doi:10.1001/archpsyc.55.10.918
- Cipriani A, Furukawa TA, Salanti G, et al. Comparative efficacy and acceptability of 21 antidepressant drugs for the acute treatment of adults with major depressive disorder: A systematic review and network meta-analysis. Lancet. 2018;391(10128):1357–1366. doi:10.1016/S0140-6736(17)32802-7
