A 2026 case report linked 1 mg nightly guanfacine modified-release to first-ever cessation of intrusive deliberate self-harm thoughts in a 16-year-old with attention-deficit/hyperactivity disorder (ADHD), autism spectrum disorder (ASD) traits, borderline personality disorder (BPD) traits, and post-traumatic stress disorder (PTSD). The same case became more interesting after the patient stopped taking guanfacine and the self-harm ideation returned, giving the paper a challenge-dechallenge pattern rather than a simple before-after anecdote.1
Research Highlights
- The headline signal is n=1 but specific: guanfacine modified-release was started at 1 mg nightly, briefly increased to 2 mg, then reduced to 1 mg because of drowsiness; on 1 mg, the patient reported that intrusive deliberate self-harm thoughts had stopped for the first time in his life.1
- The case had measurable complexity: the patient met 8 of 9 inattentive ADHD criteria and 8 of 9 hyperactivity/impulsivity criteria, scored 33/50 on the Autism-Spectrum Quotient, and had PTSD plus emerging BPD traits before guanfacine was tried.1
- The failed comparator matters: atomoxetine was prescribed before guanfacine and reached 75 mg at discharge, but the patient stopped it after limited benefit plus nausea and light-headedness.1
- Adjacent evidence is uneven: adult ADHD randomized data favor guanfacine over placebo on ADHD-RS-IV change (−11.55 vs. −7.27; P = .0005), while trauma and BPD self-injury evidence is still mostly open-label or case-report level.2,4,5
- Autism-spectrum hyperactivity has stronger trial support than self-harm outcomes: in 62 children with ASD and hyperactivity, guanfacine reduced ABC-Hyperactivity scores by 43.6% vs. 13.2% with placebo, but that trial did not test deliberate self-harm thoughts.3
Guanfacine is an α2A-adrenergic receptor agonist, meaning it stimulates a norepinephrine-sensitive receptor subtype that helps prefrontal cortex networks regulate attention, impulse control, and emotional arousal. That mechanism is why this case is worth reading carefully: the proposed target was not “suicidality” in the abstract, but mental restlessness that kept generating intrusive deliberate self-harm (DSH) thoughts.
The calibrated read is narrow. Guanfacine is not proven to prevent suicide, treat BPD, or replace trauma-focused care.
The case does suggest that, in some ADHD-trauma-dysregulation presentations, the clinically relevant symptom may be the racing internal pressure that makes self-harm thoughts harder to disengage from.
Yeung 2026: 1 mg Nightly After a 2 mg Sedation Problem
Yeung et al. described a 16-year-old transgender Maori male, biological female, with ADHD, ASD traits, emerging BPD traits, PTSD, and recurrent DSH behaviors.
The clinical chronology was unusually concrete for a case report: self-cutting began at age 11; a suicide attempt brought the patient to the emergency department in August 2022; self-harm led to another emergency presentation in February 2024; and a March 2024 youth inpatient admission involved intense DSH and suicidal ideation.1
ADHD was not a vague background label. The researchers used a clinical semi-structured diagnostic interview based on the Parental Account of Children’s Symptoms (PACS), and the patient met 8 of 9 inattentive criteria plus 8 of 9 hyperactivity/impulsivity criteria.
ASD traits were also quantified: the Autism-Spectrum Quotient (AQ), a self-report measure of autistic traits, was 33/50, a score the researchers interpreted as significant ASD symptom traits.1
Before guanfacine, treatment included atomoxetine 25 mg in the morning and quetiapine 25 mg at night, with discharge doses of atomoxetine 75 mg and quetiapine 50 mg. Atomoxetine is a non-stimulant ADHD medication that blocks norepinephrine reuptake; it increases norepinephrine availability rather than directly stimulating α2A receptors.
In this patient, atomoxetine had limited benefit and was stopped after intolerable nausea and light-headedness.1
Guanfacine modified-release was then started at 1 mg nightly on June 18 during a readmission for suicidal ideation. It was increased to 2 mg after 3 days, then reduced back to 1 mg after 2 days because of drowsiness and sedation.
At that lower dose, the patient reported that intrusive DSH and suicidal ideation had stopped for the first time in his life, alongside loss of “racing thoughts.”1
The Challenge-Dechallenge Pattern Is the Real Signal
Challenge-dechallenge means symptoms improve when an exposure starts and recur when the exposure is removed. It is weaker than a randomized trial, but stronger than a single time-point impression because the timeline asks whether the symptom moves with the intervention.
During active guanfacine treatment, acute healthcare use decreased, and the patient used the Patient Controlled Crisis Admission Program (PCAP), a planned 72-hour voluntary admission pathway for youth with BPD, in a more constructive way.
After late-August nonadherence and self-discontinuation, the patient deteriorated, had a September polypharmacy overdose involving 25 guanfacine tablets, spent 2 days in the emergency department, and had 2 October emergency presentations for hallucinations and suicidal ideation requiring admission for risk containment.1
The sequence still does not prove causality. Adolescents with complex trauma, neurodevelopmental symptoms, and self-harm histories can improve or deteriorate for many reasons: anniversary reactions, family stress, inpatient containment, psychotherapy intensity, sleep, substance exposure, and engagement with services can all move risk.
But the return of DSH ideation after guanfacine discontinuation makes the case harder to dismiss as mere spontaneous improvement.
Guanfacine Targets Regulation Rather Than Stimulation
Stimulants such as methylphenidate and amphetamine increase catecholamine signaling, especially dopamine and norepinephrine, across multiple circuits. They are often the most effective ADHD medications, but in patients with trauma hyperarousal, insomnia, agitation, or substance-use risk, clinicians may hesitate to push activation higher.
Prefrontal regulation: α2A receptor stimulation in prefrontal cortex can strengthen network signaling for working memory, response inhibition, and top-down emotional control. Arnsten’s translational work describes this as a prefrontal-cortical mechanism rather than a broad activating stimulant effect, which is why guanfacine can make sense when the target is internal overdrive rather than low motivation alone.6
In the Yeung case, the proposed mediator was mental restlessness: a subjective state of constant internal activity, racing thoughts, and cognitive agitation that can resemble worry or obsessional intrusion.
The researchers argued that this restlessness may have carried the DSH thoughts forward. Guanfacine did not need to be framed as an anti-suicide drug for the case to be clinically interesting; it may have reduced the restlessness that made self-harm ideation persistent.
Adjacent Evidence Supports Symptoms, Not a Broad Self-Harm Claim
The surrounding guanfacine literature splits into stronger and weaker zones. For ADHD symptoms, adult randomized evidence exists.
Iwanami et al. tested guanfacine extended-release in adults with ADHD and found greater ADHD-RS-IV total-score reduction at week 10 with guanfacine than placebo: −11.55 vs. −7.27, P = .0005, effect size 0.52.2 That supports guanfacine as a real ADHD medication, more than a sedating adjunct.
For ASD-linked hyperactivity, the evidence is also controlled. Scahill et al. randomized 62 children with ASD plus hyperactivity, impulsiveness, and distractibility to extended-release guanfacine or placebo.
Aberrant Behavior Checklist-Hyperactivity scores fell 43.6% with guanfacine compared with 13.2% with placebo, with an effect size of 1.67.3 That trial supports hyperactivity and impulsivity reduction in ASD, but it does not test DSH ideation, BPD traits, or trauma memories.
For trauma symptoms, the evidence becomes thinner. Connor et al. ran an 8-week open-label pilot in 19 children and adolescents aged 6–18 years with traumatic-stress symptoms, using guanfacine extended-release 1–4 mg in the evening.
The average daily dose was 1.19 mg ± 0.35 mg, and parent-reported UCLA PTSD Reaction Index clusters for reexperiencing, avoidance, and overarousal improved.4 The design is hypothesis-generating because everyone knew the treatment and there was no placebo group.
BPD and self-injury evidence: Mori et al. reported 2 adult BPD cases, one with comorbid ADHD, in which guanfacine 4–6 mg/day was associated with improvements in impulsivity, irritability, mood instability, aggression, and self-injury.5
Yeung et al. extended that case-report territory by shifting the proposed pathway from outward impulsivity alone to internal mental restlessness and intrusive DSH ideation.
The Case-Report Table Shows Why Overclaiming Would Be Wrong
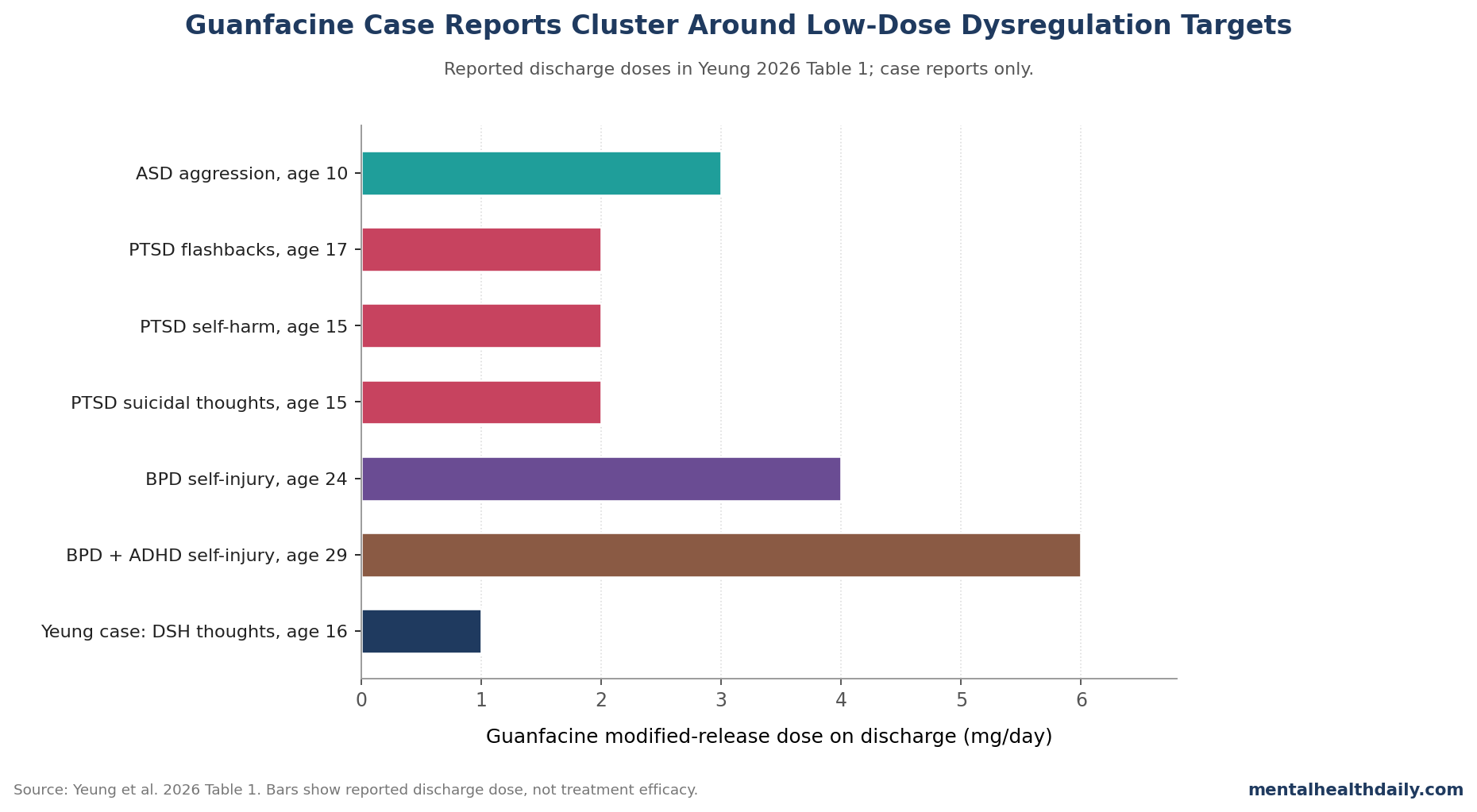
Yeung et al.’s own Table 1 is useful because it refuses to look like a guideline. Across 7 case rows, ages ranged from 10 to 29 years, discharge doses ranged from 1 mg/day to 6 mg/day, and reported symptom targets differed by diagnosis: disruptive behavior in ASD, intrusive images and poor sleep in PTSD, self-injury and aggression in BPD, and intrusive self-harm thoughts in the Yeung case.1
That heterogeneity is not a nuisance detail. It is the central limitation.
The table supports a family resemblance across dysregulation states, not a single validated indication. Guanfacine may help when prefrontal regulation, hyperarousal, impulsivity, and intrusive cognition overlap; it does not follow that every self-harm presentation is a guanfacine-responsive state.
Evidence-strength note: for deliberate self-harm thoughts specifically, the current evidence is case-report level. The strongest data in this evidence base concerns ADHD and ASD hyperactivity; the most clinically provocative data concern DSH ideation, PTSD-related intrusions, and BPD-linked self-injury, but those sit lower on the evidence ladder.
Dosing and Monitoring Are Part of the Interpretation
The Yeung case should also be read as a tolerability lesson. The patient did not need a high dose for the reported mental-restlessness effect.
Guanfacine was started at 1 mg nightly, increased to 2 mg after 3 days, and then reduced to 1 mg after drowsiness and sedation. The published table lists the final dose as 1 mg nocte, meaning at night.1
Common adverse effects follow from the drug’s pharmacology. Somnolence, fatigue, lowered blood pressure, and bradycardia are not side issues; guanfacine began as an antihypertensive.
Blood pressure and heart rate should be checked at baseline and after dose changes. Abrupt discontinuation can produce rebound hypertension, so stopping should usually be tapered rather than improvised.
The overdose episode in the Yeung case matters too. A later polypharmacy overdose involved 25 guanfacine tablets.
That does not erase the earlier treatment signal, but it changes the practical risk calculation. In a patient with DSH behaviors, medication quantity, family supervision, blister packing, dispensing interval, and crisis-plan access are part of the prescription, not administrative afterthoughts.
Where Guanfacine Fits for ADHD-PTSD-BPD-ASD Comorbidity
A defensible clinical sequence is narrower than the current hype would be. If ADHD symptoms are dominant and the patient can tolerate activation, stimulants usually remain the evidence-backed first move.
If stimulants are risky or poorly tolerated, atomoxetine or an α2A agonist becomes more plausible. If the presentation is dominated by internal restlessness, hyperarousal, sleep disruption, impulsive escalation, and intrusive self-harm thoughts, guanfacine becomes mechanistically coherent.
That sequence does not make guanfacine a stand-alone treatment for PTSD or BPD. Trauma-focused psychotherapy, environmental stabilization, safety planning, family work, sleep repair, and careful management of access to lethal means remain central.
Guanfacine is best understood as a possible regulator of the internal pressure that can make those interventions easier to use.
The Yeung case fits that role. Atomoxetine was poorly tolerated.
The patient’s ADHD and ASD traits gave guanfacine an established neurodevelopmental rationale. PTSD and BPD traits added the hyperarousal and dysregulation context.
Intrusive DSH ideation supplied the symptom target. A 1 mg nightly dose then coincided with a specific change the patient could name: the racing thoughts and self-harm thoughts stopped.
Questions About Guanfacine for Self-Harm Thoughts and Complex ADHD
Did the Yeung case prove that guanfacine prevents suicide?
No. It was a single case report, and the outcome was intrusive DSH and suicidal ideation moving with guanfacine exposure, not a controlled suicide-prevention endpoint.
The case supports a hypothesis about mental restlessness and self-harm ideation; it does not estimate suicide-attempt prevention.
Why was atomoxetine failure relevant in this case?
Atomoxetine had been the first non-stimulant ADHD strategy and reached 75 mg at discharge, but the patient stopped it after limited benefit plus nausea and light-headedness. That makes the guanfacine response less like a generic improvement after any ADHD medication and more like a potentially different mechanism.
Should guanfacine be considered for BPD-related self-injury?
Only with tight calibration. Mori et al. reported 2 adult BPD cases with self-injury improvement on 4–6 mg/day, and Yeung et al. reported 1 adolescent complex-comorbidity case on 1 mg nightly.
That is enough to justify research and careful off-label consideration in selected cases, not enough to make guanfacine a routine BPD treatment.
What kind of patient matches the Yeung hypothesis best?
The best match is not “anyone with self-harm thoughts.” It is a patient whose self-harm ideation appears tied to ADHD-like mental restlessness, trauma hyperarousal, impulsive escalation, sleep disruption, or ASD-linked dysregulation, especially when stimulants or atomoxetine are poorly tolerated.
What would make the guanfacine self-harm signal more convincing?
A useful next study would measure mental restlessness repeatedly, randomize guanfacine against placebo or another non-stimulant ADHD medication, track DSH ideation and behavior separately, and report whether improvement in restlessness statistically mediates the self-harm outcome.
References
- Yeung JT, Wan P, Chen W. Guanfacine as a treatment option for persistent mental restlessness and deliberate self-harm thoughts in a patient with ADHD, ASD, BPD, and PTSD: case report. Clin Psychopharmacol Neurosci. 2026;24(2):419-424. doi:10.9758/cpn.25.1352
- Iwanami A, Saito K, Fujiwara M, Okutsu D, Ichikawa H. Efficacy and safety of guanfacine extended-release in the treatment of attention-deficit/hyperactivity disorder in adults: results of a randomized, double-blind, placebo-controlled study. J Clin Psychiatry. 2020;81(3):19m12979. doi:10.4088/JCP.19m12979
- Scahill L, McCracken JT, King BH, et al. Extended-release guanfacine for hyperactivity in children with autism spectrum disorder. Am J Psychiatry. 2015;172(12):1197-1206. doi:10.1176/appi.ajp.2015.15010055
- Connor DF, Grasso DJ, Slivinsky MD, Pearson GS, Banga A. An open-label study of guanfacine extended release for traumatic stress related symptoms in children and adolescents. J Child Adolesc Psychopharmacol. 2013;23(4):244-251. doi:10.1089/cap.2012.0119
- Mori H, Ochi S, Hashida H, Iga JI, Ueno SI. Efficacy of guanfacine for self-injurious and aggressive behaviors through the reduction of impulsivity in borderline personality disorder: two case reports and a literature review. Clin Psychopharmacol Neurosci. 2025;23(3):520-525. doi:10.9758/cpn.24.1263
- Arnsten AFT. Guanfacine’s mechanism of action in treating prefrontal cortical disorders: successful translation across species. Neurobiol Learn Mem. 2020;176:107327. doi:10.1016/j.nlm.2020.107327