Electroconvulsive therapy carries more cultural baggage than perhaps any other treatment in modern psychiatry. Decades after the practice was modernized into its current form — brief-pulse stimulation under general anesthesia and muscle relaxation — the cultural memory still draws on mid-twentieth-century imagery and 1970s film depictions that bear little resemblance to current practice. Modern administrative-health-data studies, summarized in Kaster and colleagues’ 2026 review in the British Journal of Psychiatry, address the long-term risk and benefit questions clinical trials can’t answer. The picture they show is consistent and largely reassuring.1
Research Highlights
- ECT remains the most effective treatment for severe and treatment-resistant depression. The seminal 2003 UK ECT Review Group meta-analysis found a standardized mean difference of 0.91 favoring active over sham ECT — equivalent to roughly 9.7 points on the Hamilton Depression Rating Scale — and an SMD of 1.01 favoring ECT over pharmacotherapy. Network meta-analyses of brain-stimulation modalities consistently rank ECT first for symptom reduction.
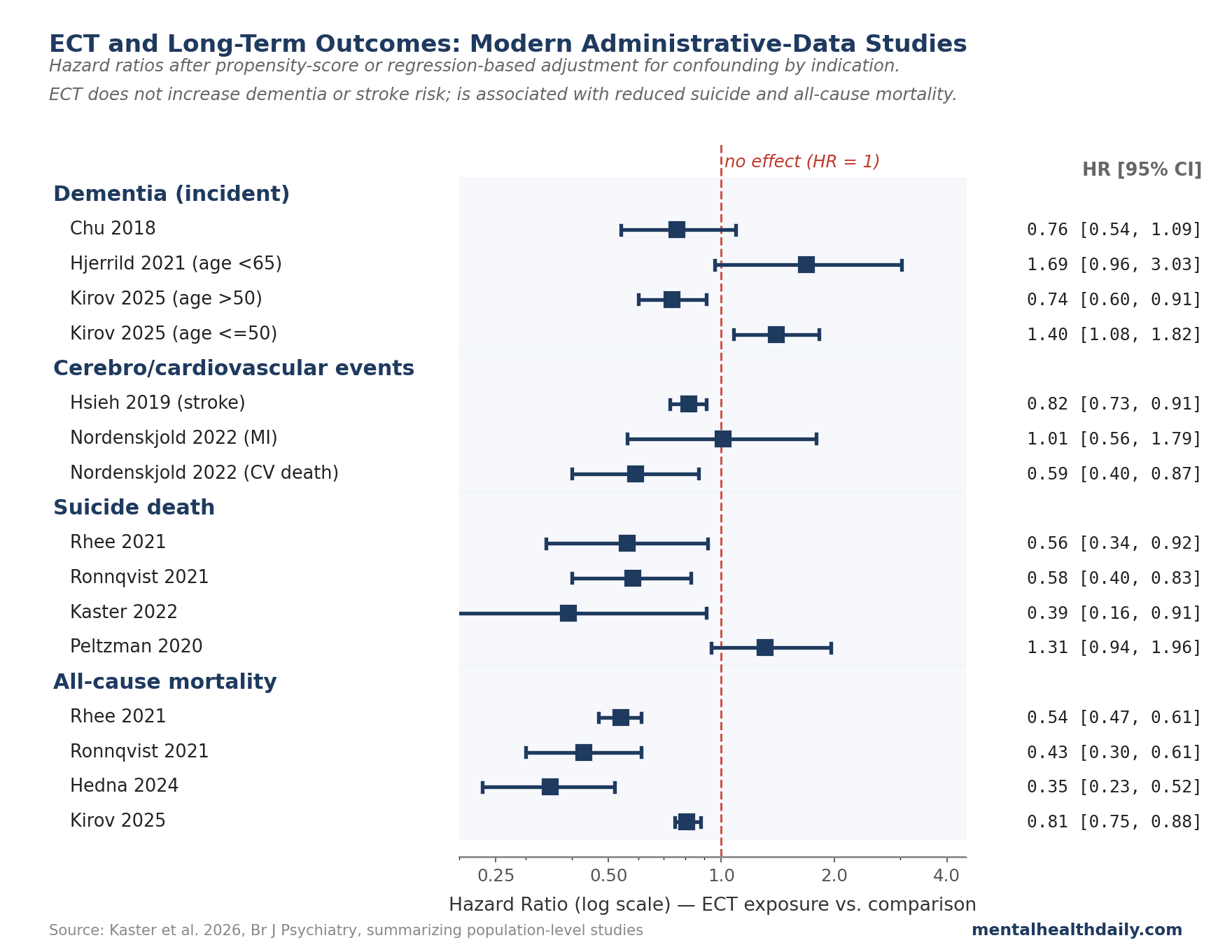
- Modern administrative-data studies refute the “ECT causes dementia” claim. Across five large population-level studies using propensity-score or regression-based confound adjustment, the association between ECT exposure and incident dementia in adults with mood disorders is null or favorable. The single subgroup signal (younger patients in one study) had few events and a small sample.
- ECT is associated with reduced suicide death and reduced all-cause mortality. Multiple 2025 systematic reviews report 30 to 50% reductions in suicide-related mortality after ECT exposure. All-cause mortality hazard ratios cluster between 0.4 and 0.8 across population-level studies.
- Cardiovascular risk after ECT is not elevated in most populations and may be lower for cardiovascular death. Small short-term increases in acute cardiac events appear in subgroups without prior comorbidity but don’t persist beyond 30 days. Stroke risk is not elevated after appropriate confound adjustment.
- The dementia-causation framing in particular is no longer well supported. Modern administrative cohorts find no overall dementia signal after appropriate adjustment, and a reduction in suicide and all-cause mortality. The supportive literature does carry known biases (treatment-by-indication confounding, short outcome windows, publication patterns favoring positive findings) — the picture is informative but not a wholesale endorsement of broad ECT use.
- Cognitive side effects remain a real and inadequately quantified concern. Transient anterograde amnesia and longer-tail retrograde amnesia for autobiographical memories are well-documented and an appropriate part of informed consent. They are distinct from the dementia question that the administrative data address.
The Efficacy Case: Established by RCTs Decades Ago
The foundational evidence for ECT efficacy is the UK ECT Review Group’s 2003 meta-analysis in The Lancet, which pooled data across the existing RCTs of active versus sham ECT and active ECT versus pharmacotherapy.2 Headline findings:
- Active vs. sham ECT: SMD 0.91 (favoring active) — approximately a 9.7-point reduction on the Hamilton Depression Rating Scale.
- Active ECT vs. pharmacotherapy: SMD 1.01 (favoring ECT) — ECT outperforming standard antidepressants in head-to-head trials.
- Remission rates in severe and treatment-resistant depression: typically 50 to 70% — substantially higher than typical pharmacotherapy remission rates of 30 to 40% after multiple medication trials.
(SMD — standardized mean difference — lets us compare studies that used different symptom scales. Rough benchmarks: 0.2 small, 0.5 moderate, 0.8 large. SMDs near 1.0 are unusually large for psychiatric treatments.)
A more recent network meta-analysis by Mutz and colleagues compared all forms of non-surgical brain stimulation for depression, including repetitive transcranial magnetic stimulation (rTMS), magnetic seizure therapy, and transcranial direct current stimulation. ECT outperformed every other modality on response and remission rates.3 Pagnin and colleagues’ earlier review of sham-controlled ECT trials reached the same conclusion via different methodology.9 The efficacy hierarchy is established and stable.
What RCTs cannot answer is whether the symptom-reduction benefits translate into hard outcomes years and decades later, and whether long-term harms emerge that short trials cannot detect. The largest reasonable RCT in the UK ECT Review Group was 12 weeks. The questions patients most want answered — “Will this cause dementia in 20 years? Will I die earlier? Will it really reduce my suicide risk?” — require population-scale longitudinal data.
ECT and Dementia: What Modern Cohorts Actually Show
The concern that ECT causes dementia is one of the most enduring stigmas around the treatment. The concern is biologically plausible-sounding given known cognitive side effects (anterograde and retrograde amnesia in the immediate post-treatment period), but the empirical record contradicts it.
Kaster’s review identified five large population-level studies looking at ECT and dementia. All five used statistical methods (propensity-score matching, regression adjustment) to compare ECT-exposed patients to similar non-exposed patients on the variables that matter — age, depression severity, comorbidities — rather than just lumping all comers together.1 The pattern across studies:
- Pre-adjustment, ECT-exposed patients had higher dementia incidence in several studies. This is what you’d expect: the indication for ECT (severe depression) is itself a known dementia risk factor, so unadjusted comparisons mostly capture confounding by indication.
- After adjustment for confounders, the association with dementia disappeared. Hjerrild et al. 2021 reported hazard ratios (HRs — the multiplier on the rate of the outcome over time, where 1.0 means no effect, above 1 means more risk, below 1 means less) of 1.08, 1.69, and 3.45 across age strata, none statistically significant. Chu et al. 2018 reported HR 0.76 (95% CI 0.54 to 1.09). Osler et al. 2018 reported HRs from 0.77 to 2.36 across age strata, only the oldest stratum borderline.1
- The single positive signal came from a younger subgroup in Kirov et al. 2025 (HR 1.40, 95% CI 1.08 to 1.82 in adults aged 50 or younger), but with small absolute numbers and an opposite signal in older patients (HR 0.74, 95% CI 0.60 to 0.91).4 The interpretation is genuinely uncertain.
The most defensible read: once the analyses properly account for the fact that ECT goes to sicker patients (which on its own raises dementia risk), the population-level data show no overall link between ECT and developing dementia. Brain-imaging studies have similarly failed to find structural damage that could be blamed on ECT.1
The cognitive side effects of ECT — transient anterograde amnesia, sometimes more lasting retrograde amnesia for autobiographical memories, subjective complaints of word-finding difficulty — are well-documented and an appropriate part of informed consent. Permanent neurodegeneration leading to dementia, however, is not what the data show.
Suicide Mortality: A Reduction in Observational Data
The clinical hypothesis that ECT reduces suicide death has historically been hard to test. ECT is preferentially given to the most acutely suicidal patients, who are themselves at higher baseline risk — confounding by indication that, without proper adjustment, would make ECT appear harmful even if it were lifesaving.
Modern propensity-score and regression-adjusted analyses largely overcome this. Four 2025 systematic reviews of population-level data converged on a consistent finding: ECT exposure is associated with a 30 to 50% reduction in suicide death.1 Individual studies in the population:
- Rhee et al. 2021 (older adults with mood disorders, n > 10,000): HR 0.56 (95% CI 0.34 to 0.92).5
- Ronnqvist et al. 2021 (Swedish in-patients with mood disorders): HR 0.58 (95% CI 0.40 to 0.83).6
- Kaster et al. 2022 (Ontario in-patients with mood disorders, propensity-score weighted): HR 0.39 (95% CI 0.16 to 0.91).7
- Peltzman et al. 2020 (Veterans Affairs, any psychiatric diagnosis): HR 1.31 (95% CI 0.94 to 1.96) — the one study that didn’t show a reduction, with results that crossed null.1
The convergence is meaningful. Different countries, different populations, different statistical adjustments — all yield estimates clustered in the 30 to 60% reduction range. The Peltzman 2020 outlier may reflect VA-specific case-mix differences, but the broader signal is robust.
For comparison, one of the same meta-analyses examined antidepressant treatment and found no significant association between antidepressants and suicide death.1 Worth flagging the asymmetry, with the same caveat: both estimates come from observational data subject to indication bias, but the directions of effect are opposite.
All-Cause Mortality: ECT Is Associated With Reduced Death
Beyond suicide, ECT exposure is consistently associated with reduced all-cause mortality across population-level studies.1 Hazard ratios cluster between approximately 0.4 and 0.8:
- Rhee 2021: HR 0.54 (95% CI 0.47 to 0.61)
- Ronnqvist 2021: HR 0.43 (95% CI 0.30 to 0.61)
- Hedna 2024: HR 0.35 (95% CI 0.23 to 0.52)
- Nordenskjöld 2022: HR 0.74 (95% CI 0.61 to 0.89)
- Osler 2022: HR 0.71 (95% CI 0.66 to 0.77)
- Kirov 2025: HR 0.81 (95% CI 0.75 to 0.88)
- Watts 2021 (Texas, 1998-2013, propensity-score matched): HR 0.87 (95% CI 0.79 to 1.11)10
The convergence across countries and decades is again notable. Confounding by indication would, if anything, push the apparent ECT mortality effect toward harm — the most severely ill patients receive ECT and have higher baseline mortality. That the adjusted hazard ratios consistently land below 1.0 is therefore informative: ECT may be lifesaving for the severely ill population that receives it.
The causal interpretation has limits. Even careful matching can’t account for things the data doesn’t capture — patient motivation, family support, willingness to follow through on treatment — which probably push both ECT acceptance and survival in the same direction. But the same pattern showing up across different countries, study designs, and statistical approaches makes the signal hard to write off as a quirk of any one analysis.
Cardiovascular and Cerebrovascular Events: Modest, Mostly Null
ECT involves a brief seizure under general anesthesia and produces transient sympathetic and parasympathetic activation. The concern that it might increase cardiovascular or cerebrovascular events is reasonable.
The administrative-data evidence is reassuring overall, with some nuance. For stroke and cerebrovascular events, Hsieh 2019 reported HR 0.82 (95% CI 0.73 to 0.91) — lower stroke risk after ECT. A sensitivity analysis using competing-risk methods produced a non-significant subdistribution HR of 0.86, suggesting the apparent reduction may partly reflect competing risk from mortality.1 No study found increased cerebrovascular risk after appropriate adjustment.
For cardiovascular events, the picture is more mixed. Most studies find no overall increase.
Osler 2022 reported a transient elevation in acute cardiac events in patients without prior comorbidity (HR 3.72, 95% CI 1.86 to 7.43), but the increase was based on small numbers and did not persist beyond 30 days.1 Nordenskjöld 2022 found no increase in myocardial infarction (HR 1.01) and a reduction in cardiovascular death (HR 0.59).1
The defensible synthesis: peri-ECT cardiovascular monitoring is appropriately part of standard practice, with a small early-window risk in some subgroups, but no sustained increase in cardiovascular events or death. Most populations show no elevated risk; some show reduced cardiovascular mortality, likely tied to the broader all-cause mortality reduction.
Where the Modern ECT Administrative Data Have Limits
The administrative-data literature, despite its strengths, has limitations that should be understood by anyone considering ECT.
The “ECT goes to the sickest patients” problem can’t be fully solved. ECT is reserved for severe and treatment-resistant cases, and severity is a hard thing to fully measure in administrative records. Even with careful matching, some leftover effect of “these patients were sicker than the comparison group” probably remains. That leftover bias would push the apparent results in the direction of “ECT looks worse than it really is,” which makes the consistently favorable findings more credible — but the exact magnitudes shouldn’t be read as precise.
Cognitive side effects are inadequately captured in administrative data. The most clinically important short-to-medium-term concern about ECT is the cognitive effect — particularly retrograde amnesia for autobiographical memories. Administrative data cannot quantify this nuance. Detailed neuropsychological-assessment trials remain the appropriate evidence base for the cognitive question; administrative data answer the dementia question, which is different.
Bilateral vs. unilateral electrode placement and dose parameters vary across studies. Modern ultra-brief right-unilateral protocols produce fewer cognitive side effects than older bilateral protocols. The pooled population-level studies don’t uniformly distinguish protocol types, so estimates average across heterogeneous practice.
Generalizability to outpatient ECT and continuation/maintenance ECT is limited. Most administrative studies focus on inpatient acute-course ECT. The expanding outpatient and maintenance ECT populations have less complete population-level evidence.
The dissenting literature exists and should not be ignored. Read and Bentall’s 2010 review argued that ECT efficacy is overstated and risks understated.8 Subsequent critiques have raised similar concerns. The modern administrative-data evidence largely answers the long-term-harm questions these critiques raised, but the underlying ethical concerns about informed consent and patient experience remain valid — and inform why ECT remains reserved for severe and treatment-resistant cases rather than first-line use.
Where ECT Sits in Modern Practice
ECT has well-established efficacy that exceeds first-line antidepressants and other brain-stimulation modalities. The risk profile, on the long-term-outcomes administrative data, is favorable: no overall dementia risk, no overall stroke risk, reduced suicide mortality, reduced all-cause mortality. The cognitive side effects are well-documented, transient for most patients, and the proper subject of informed consent.
- For severe or treatment-resistant depression with active suicidality, ECT is among the most evidence-supported interventions. The combination of high efficacy and reduced suicide-mortality association makes it particularly appropriate for acutely high-risk presentations.
- For depression with psychotic features or catatonia, ECT is often the most effective option. Response rates in these specific presentations are higher than for general depression, and pharmacological alternatives often work poorly.
- For treatment-resistant mania and life-threatening catatonia, ECT can be lifesaving. These are the historical core indications.
- Right-unilateral ultra-brief protocols are now standard for cognitive-side-effect minimization. Patients evaluating ECT should ask about electrode placement and pulse width; modern protocols are notably easier on memory than older bilateral approaches.
- The dementia-causation specifically is not what the modern data show, but informed declination is still informed. Patients and families weighing ECT have legitimate reasons beyond the dementia question: cognitive side effects on autobiographical memory, the historical and ongoing concerns about coercion and informed consent in some settings, and the publication-bias caveats on the supportive literature. The takeaway isn’t “decline is irrational” — it’s that the specific claim “ECT causes long-term brain damage / dementia” is not supported by modern cohort data.
Quick Answers on Modern ECT
Does ECT cause brain damage or dementia?
No, on the available evidence. Imaging studies do not find structural neural damage; population-level administrative-data studies, after appropriate adjustment for confounding by indication, do not find an overall increased risk of incident dementia.1,4 Cognitive side effects are well-documented and short-term but distinct from neurodegeneration.
How effective is ECT compared to antidepressants?
More effective in head-to-head trials. The UK ECT Review Group reported SMD 1.01 favoring ECT over pharmacotherapy — a large effect.2 ECT also outperforms rTMS and other brain-stimulation modalities in network meta-analyses.3
What are the side effects of ECT?
The most common are transient anterograde amnesia (difficulty forming new memories around the time of treatment), sometimes more lasting retrograde amnesia (loss of autobiographical memories from before treatment), headache, muscle soreness, and the standard risks of brief general anesthesia. Most cognitive effects resolve within weeks; a minority of patients report persistent memory gaps. Modern right-unilateral ultra-brief protocols reduce these effects relative to older bilateral protocols.
Is ECT used in current practice?
Yes — routinely in major academic and community hospitals. Modern ECT involves brief-pulse stimulation under general anesthesia and muscle relaxation. The mid-twentieth-century practice depicted in cultural memory and 1970s film bears little resemblance to the current procedure.
Can ECT be done as an outpatient?
Yes. Acute courses (typically 6 to 12 sessions over 2 to 4 weeks) are increasingly outpatient. Maintenance ECT (less frequent sessions to prevent relapse) is also outpatient.
Why is ECT still controversial?
Several reasons, some legitimate. Mid-twentieth-century practice was cruder and used without anesthesia; that historical memory persists, and arguably should. Cultural depictions (notably One Flew Over the Cuckoo’s Nest) anchored a coercive image that maps to documented mid-century abuses. Cognitive side effects on autobiographical memory are personally salient and not fully captured in administrative data. A dissenting clinical literature (Read & Bentall and others) continues to argue ECT efficacy is overstated and harms understated — a critique modern cohort data partly answer on the dementia question but not entirely on the cognitive-effect question.8 The most defensible read: dementia causation is not supported, the suicide and mortality signals are informative, and the cognitive and consent concerns remain substantive.
Who should consider ECT?
Patients with severe or treatment-resistant depression (particularly with suicidality, psychotic features, or catatonia), treatment-resistant mania, or life-threatening catatonia. ECT is not first-line for mild-to-moderate depression; the risk-benefit profile favors trying medication and psychotherapy first. For severe presentations where standard treatments have failed, ECT is among the most evidence-supported options available.
References
- Electroconvulsive therapy: improved understanding of long-term risks and benefits from advances in administrative health data. Kaster TS et al. The British Journal of Psychiatry. 2026;1-6. doi:10.1192/bjp.2026.10613
- Efficacy and safety of electroconvulsive therapy in depressive disorders: a systematic review and meta-analysis. UK ECT Review Group. The Lancet. 2003;361(9360):799-808. doi:10.1016/S0140-6736(03)12705-5
- Comparative efficacy and acceptability of non-surgical brain stimulation for the acute treatment of major depressive episodes in adults: systematic review and network meta-analysis. Mutz J et al. BMJ. 2019;364:l1079. doi:10.1136/bmj.l1079
- Electroconvulsive therapy and the risk of subsequent dementia: a population-based cohort study. Kirov G et al. The British Journal of Psychiatry. 2025;226(2):78-85. doi:10.1192/bjp.2024.215
- Association of Electroconvulsive Therapy With Mortality and Suicide Among Patients With Depression. Rhee TG et al. JAMA Network Open. 2021;4(3):e213164.
- Electroconvulsive Therapy and the Risk of Suicide in Hospitalized Patients With Major Depressive Disorder. Ronnqvist I et al. JAMA Network Open. 2021;4(7):e2116589. doi:10.1001/jamanetworkopen.2021.16589
- Differential Effects of Electroconvulsive Therapy on All-Cause and Suicide Mortality. Kaster TS et al. The American Journal of Psychiatry. 2022;179(8):568-576. doi:10.1176/appi.ajp.21111100
- The effectiveness of electroconvulsive therapy: a literature review. Read J, Bentall R. Epidemiologia e Psichiatria Sociale. 2010;19(4):333-347. doi:10.1017/S1121189X00000671
- Electroconvulsive therapy for depression: a review of the quality of ECT versus sham ECT trials and meta-analyses. Pagnin D et al. Journal of ECT. 2004;20(1):13-20. doi:10.1097/00124509-200403000-00004
- Electroconvulsive Therapy and All-Cause Mortality in Texas, 1998-2013. Watts BV et al. The Journal of ECT. 2021;37(3):192-199. doi:10.1097/YCT.0000000000000750
