A 2026 eLife computational study modeled classical psychedelics by increasing a network state variable from alpha = 0 to alpha = 1, shifting perception from bottom-up sensory inference toward top-down generative replay. The result is a useful but still unproven oneirogen model: hallucinations look less like random noise and more like wake-time replay of a learned visual world.1
Research Highlights
- Wake-time replay is the headline mechanism: Bredenberg et al. varied alpha from 0 to 1, where 0 represented Wake-like bottom-up control and 1 represented Sleep-like top-down control.1
- The model produced multiple hallucination markers: increasing alpha generated stimulus-conditioned and eyes-closed visual hallucinations, increased stimulus-conditioned variability, and increased synaptic plasticity across trained networks.1
- Table 1 makes the model testable: the oneirogen hypothesis supported 5 of 5 proposed predictions, while REBUS, DeepDream, and excitation-inhibition pattern models failed or left unaddressed several of them.1,3,4
- The simulations were broader than MNIST: Bredenberg et al. used MNIST and CIFAR10 in Wake-Sleep-trained networks, then tested richer hallucinations in VDVAE models trained on Tiny ImageNet and FFHQ-256.1
- Plasticity remains a calibrated claim: psilocybin and other psychedelics have experimental plasticity evidence, but this model does not prove that 5-HT2A drugs cause therapy gains through the same 512-image batch update logic used in the simulations.1,5,6
Classical psychedelics are drugs such as LSD, psilocybin, DMT, and mescaline that primarily act as agonists at the 5-HT2A serotonin receptor. That receptor sits in places that matter for cortical signaling: apical dendrites of pyramidal neurons, which receive higher-order top-down inputs, and parvalbumin interneurons, which can help gate bottom-up inputs.1
Bredenberg et al. used that anatomical clue to make a sharper computational proposal. If psychedelics increase the influence of apical, top-down inputs during wakefulness, the cortex may drift toward a state normally used for offline replay during sleep.
In that state, perception is no longer anchored as tightly to incoming sensory input; the network begins expressing its own learned generative model.
Alpha = 0 to Alpha = 1: Psychedelic Dose as a Wake-Sleep Shift
The Wake-Sleep algorithm is an older machine-learning method that trains 2 pathways in a generative model.2 In the Wake phase, bottom-up recognition pathways infer what sensory input means.
In the Sleep phase, top-down generative pathways create internal activity, and the recognition pathway learns from that generated content.
Bredenberg et al. mapped that algorithm onto cortical architecture. Basal dendrites stood in for bottom-up inference.
Apical dendrites stood in for top-down prediction. The alpha parameter controlled which side dominated: alpha = 0 meant Wake-like basal control, and alpha = 1 meant Sleep-like apical control.1
The psychedelic manipulation was simple and testable. Instead of adding random noise to the network, researchers increased alpha during wakefulness.
As alpha rose, top-down generative activity increasingly competed with, then dominated, bottom-up sensory input. Hallucinations emerged when a waking network was partially shifted toward its own Sleep-phase generative machinery.
Closed-Eye Hallucinations Matched Learned Visual Content
Model hallucinations were more than white noise. In MNIST-trained networks, increasing alpha made digit-like generative activity appear over the presented image.
Under eyes-closed conditions, where the input was a black image, the network generated content shaped by what it had learned during training.1
CIFAR10-trained networks produced the same qualitative pattern with natural-image structure instead of handwritten digits. In larger pretrained very deep variational autoencoder models (VDVAEs; generative image models with stacked latent layers), increasing alpha in the top 35 layers produced richer Tiny ImageNet textures and distorted FFHQ-256 face features.1
The dataset dependence is important. A model trained on digits hallucinates digit-like structure; a model trained on faces alters faces.
That is closer to how psychedelic visuals feel than a pure-noise account: the experience often has organized geometry, scene fragments, faces, memories, or learned symbolic content. Bredenberg et al. did not prove that human hallucinations are literally the same process, but the model explains why hallucinations can be internally generated and structured at the same time.
Plasticity Increased, But It Was Not Automatically Useful Learning
The model also predicted increased synaptic plasticity. Bredenberg et al. measured plasticity as relative changes in synaptic strength across network parameters, averaged over 512 stimulus presentations for computational efficiency.1
At intermediate modeled doses, plasticity rose at both apical and basal synapses whether or not alpha also gated the learning rule.
That finding lines up with adjacent biological evidence. Shao et al. reported rapid and persistent dendritic spine growth after psilocybin in frontal cortex in vivo, and Vargas et al. argued that psychedelic-induced plasticity depends on intracellular 5-HT2A receptor activation.5,6
Those studies make the plasticity part of the oneirogen hypothesis biologically plausible.
The calibration is just as important. Bredenberg et al. explicitly cautioned that plasticity inside the model should not be read as clean behavioral improvement.
In this framework, hallucinatory activity can pull a learning system away from a local optimum and increase weight change. That could help a rigid network explore new representations, but it could also be closer to noise than therapy.
The finding should not be flattened into “hallucinations heal the brain.”
Five Predictions Separate Oneirogen From REBUS and DeepDream
REBUS (Relaxed Beliefs Under Psychedelics) is the dominant predictive-coding account of psychedelic action. It proposes that psychedelics reduce the precision-weighting of high-level priors, allowing more bottom-up information to update rigid beliefs.3
Bredenberg et al. made a different first-order claim: the hallucination state is driven by increased top-down influence.
That disagreement is not a minor wording issue. If REBUS predicts more bottom-up flow and the oneirogen model predicts more top-down replay, experiments should be able to separate the models.
Bredenberg et al. summarized the test in 5 predictions: neuronal variability should increase, pairwise across-stimulus correlations should be preserved, silencing apical dendrites should reduce variability more after psychedelic administration, silencing higher-order areas should affect lower-order activity more after psychedelic administration, and the drug effect should use circuitry responsible for generative replay.1
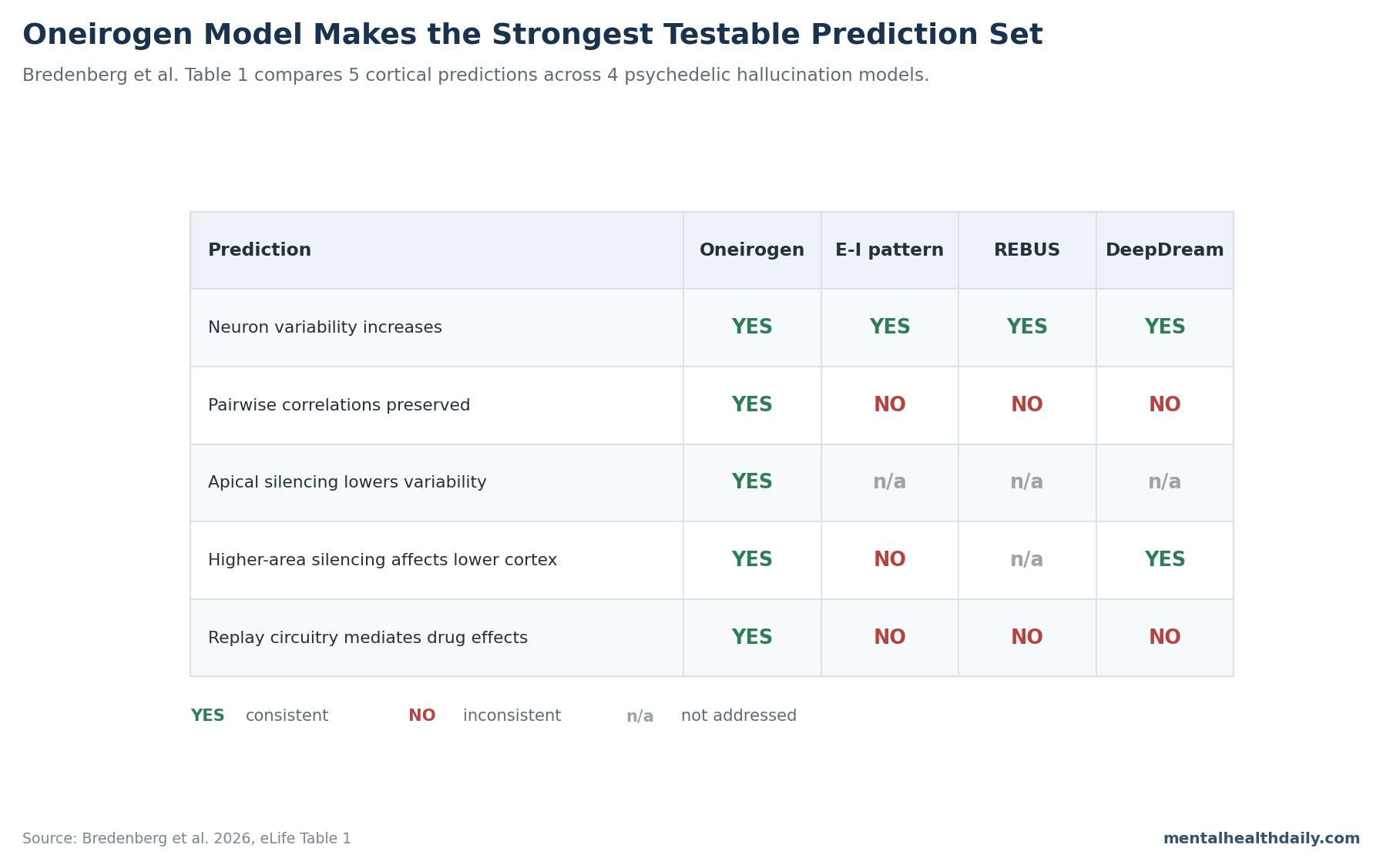
DeepDream-style models help with a different part of the problem. Suzuki et al. showed that top-down generative algorithms can create realistic hallucination-like visual phenomenology.4
Bredenberg et al. agreed with that direction of travel, but added a sleep-replay learning mechanism. DeepDream can make visuals look trippy; oneirogen tries to explain why a 5-HT2A drug might recruit a replay-like cortical state in the first place.
Network Variability Rose While Correlation Structure Stayed Mostly Intact
Entropic-brain accounts are not discarded here. Bredenberg et al. found that stimulus-conditioned variability increased as alpha rose, and that result fits the broader idea that psychedelic states involve more variable brain activity.1,7
The model adds a more specific mechanism: variability increases because top-down activity is less constrained by the particular sensory stimulus being presented.
The network-level result was more selective. Pairwise correlation structure and effective dimensionality were largely preserved across modeled psychedelic doses in trained Wake-Sleep networks.
Untrained networks and noise-based hallucination controls showed larger changes. That difference matters because it says the oneirogen model is more than “turn up entropy.” It predicts extra variability without necessarily throwing local network structure into a totally different regime.
Bredenberg et al. also tested causal influence inside the model. When alpha was high, silencing apical dendritic activity reduced across-stimulus variability more than it did at baseline, and silencing the deepest network layer produced a larger reduction in stimulus-layer variability.
Those are the kinds of predictions an animal or circuit study could actually attack.
Evidence Is Strongest for a Model, Not for Psychedelic Therapy Claims
The evidence strength sits in a narrow lane. eLife assessed the work as useful and the evidence as convincing for a computational model that reproduces some hallucination features, while noting uncertainty about whether it applies specifically to 5-HT2A hallucinogens.1 That is a fair read.
What the model supports: a biologically motivated mechanism by which increasing top-down/apical influence can generate structured hallucinations, increased variability, and increased plasticity-like weight changes in trained networks.
What the model does not support by itself: a clinical claim that stronger hallucinations produce better antidepressant, PTSD, or addiction outcomes. The model focuses on cortical perception and plasticity.
It does not include subcortical serotonergic systems, mood-regulation pathways, psychotherapy context, trauma memory reconsolidation, or the messy clinical details that decide whether a psychedelic session helps or harms.
The simplest clinical translation is therefore cautious. If the oneirogen model is right, psychedelic intensity may be a marker that top-down replay machinery is active.
That does not make intensity the treatment target. The more plausible target is what gets stabilized after the experience: which representations are reweighted, which memories are revisited, and which behavioral routines are rebuilt during the post-dose window.
Questions About Replay-Based Psychedelic Models
Are psychedelic hallucinations just dreams while awake?
No. The oneirogen model says psychedelic hallucinations may recruit replay-like cortical dynamics that resemble sleep in a computational sense.
It does not say a waking psychedelic state is identical to REM sleep, slow-wave sleep, or ordinary dreaming.1
Does this contradict REBUS?
Partly. REBUS emphasizes relaxed high-level priors and more bottom-up updating, while Bredenberg et al. emphasize increased top-down/apical influence during hallucinations.
Both could be relevant at different timescales, but the acute hallucination mechanism points in a different direction.1,3
What experiment would most directly test the model?
A strong test would combine psychedelic administration with compartment-specific or pathway-specific intervention: apical dendrite silencing, higher-order cortical silencing, or comparable recordings that can show whether top-down replay circuitry actually drives the increased variability predicted by the model.1
Should psychedelic therapy aim for stronger hallucinations?
The model does not justify that conclusion. It makes hallucinations mechanistically interesting, but it also warns that increased plasticity can be noisy and is not automatically beneficial learning.
Therapy claims still need clinical outcome data, more than a compelling cortical model.
References
- Bredenberg C, Normandin F, Richards B, Lajoie G. Modeling the hallucinatory effects of classical psychedelics in terms of replay-dependent plasticity mechanisms. eLife. 2026;14:RP105968. doi:10.7554/eLife.105968
- Hinton GE, Dayan P, Frey BJ, Neal RM. The “wake-sleep” algorithm for unsupervised neural networks. Science. 1995;268(5214):1158–1161. doi:10.1126/science.7761831
- Carhart-Harris RL, Friston KJ. REBUS and the anarchic brain: toward a unified model of the brain action of psychedelics. Pharmacological Reviews. 2019;71(3):316–344. doi:10.1124/pr.118.017160
- Suzuki K, Roseboom W, Schwartzman DJ, Seth AK. A deep-dream virtual reality platform for studying altered perceptual phenomenology. Scientific Reports. 2017;7:15982. doi:10.1038/s41598-017-16316-2
- Shao LX, Liao C, Gregg I, Davoudian PA, Savalia NK, Delagarza K, Kwan AC. Psilocybin induces rapid and persistent growth of dendritic spines in frontal cortex in vivo. Neuron. 2021;109:2535–2544. doi:10.1016/j.neuron.2021.06.008
- Vargas MV, Dunlap LE, Dong C, et al. Psychedelics promote neuroplasticity through the activation of intracellular 5-HT2A receptors. Science. 2023;379:700–706. doi:10.1126/science.adf0435
- Carhart-Harris RL, Leech R, Hellyer PJ, et al. The entropic brain: a theory of conscious states informed by neuroimaging research with psychedelic drugs. Frontiers in Human Neuroscience. 2014;8:20. doi:10.3389/fnhum.2014.00020
