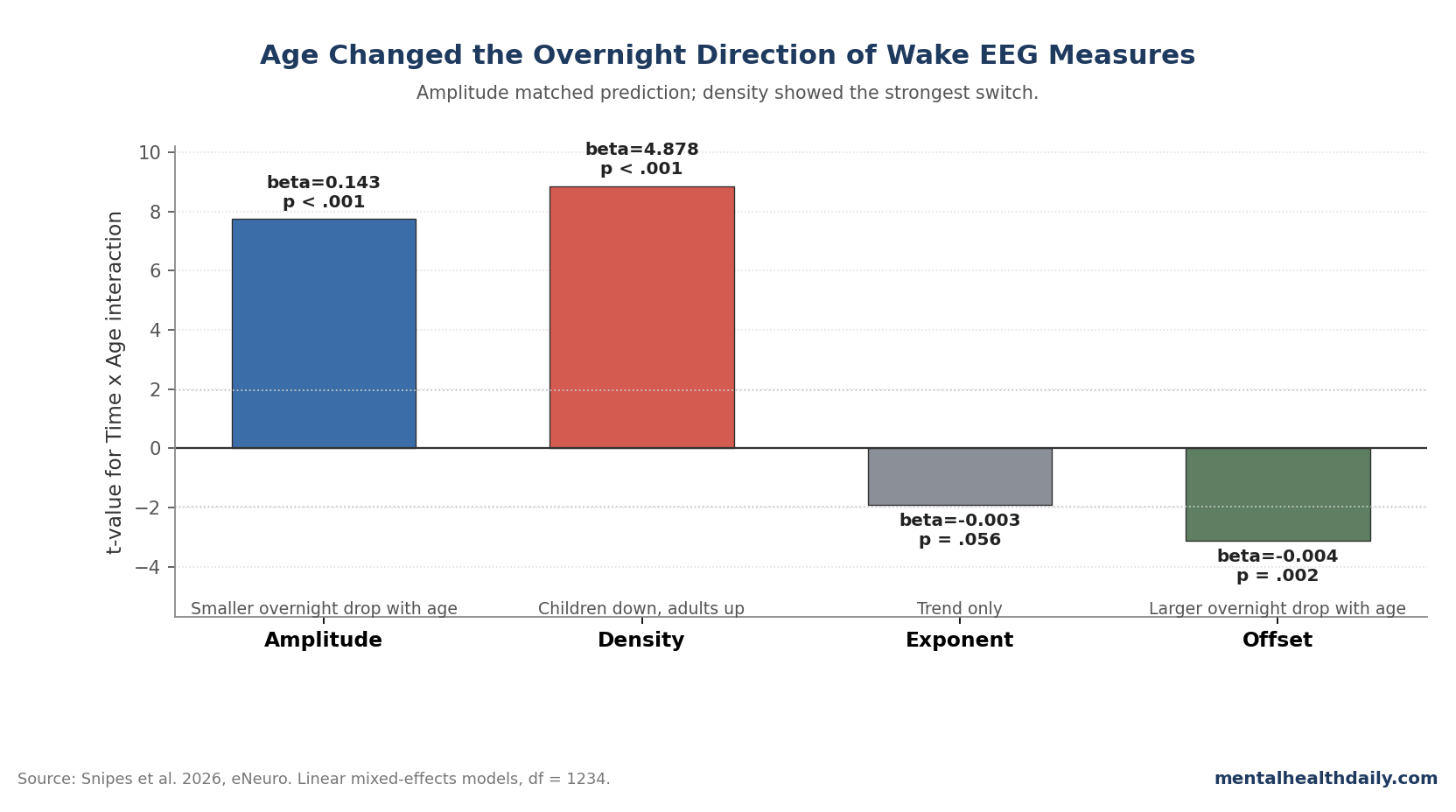
A 2026 eNeuro analysis of 163 people ages 3.5–24.7 found that wake EEG amplitude behaved like a sleep-pressure marker, but wake alpha-density changed in opposite directions across development: it decreased overnight in children and increased overnight in adolescents and adults.1
Research Highlights
- Alpha density flipped by age: In 163 participants ages 3.5–24.7, oscillation density showed a strong Time x Age interaction (β = 4.878, t = 8.85, p < .001), decreasing overnight in children under 15 and increasing in young adults.1
- Amplitude matched the main prediction: Wake oscillation amplitude decreased with age (β = −0.783, p < .001), decreased after sleep (β = −3.977, p < .001), and showed a Time x Age interaction (β = 0.143, p < .001).1
- ADHD did not survive testing: The expected ADHD amplitude difference was not significant (β = −0.574, p = .373), density was essentially null (β = −0.154, p = .992), and channel-level ADHD maps showed no FDR-corrected effect.1
- Aperiodic EEG was not interchangeable: Exponents became shallower with age (β = −0.027, p < .001) but steeper after sleep than before sleep (β = 0.099, p < .001), while offsets decreased with age (β = −0.067, p < .001) without a main overnight effect (p = .100).1
- Measurement timing matters: The dataset included 1,243 wake EEG recordings averaging 4.2 min each; spectral power tracked amplitude strongly (r = .90), but periodic power tracked density more closely (r = .85), meaning ordinary band-power summaries can blur the mechanism.1
The correction is narrower than “sleep affects children differently.” Snipes et al. did not run a sleep-deprivation experiment; they compared wake EEG in the evening before sleep and about 30 min after waking, then asked whether age, prior sleep, and ADHD changed the same EEG features.
That distinction matters for child psychiatry and developmental neuroscience because wake EEG is often treated as a trait-like brain measure. This paper shows why that can be risky: in children and adolescents, the same EEG recording can carry age, sleep/wake history, circadian timing, task, and clinical-group signal at once.
163 Participants Produced 1,243 High-Density Wake EEG Recordings
Snipes et al. assembled data from studies conducted between 2008 and 2021 at the University Children’s Hospital Zurich, plus a small Providence cohort of children ages 3.5–8.0. The final dataset included 163 participants, 38% female and 36% diagnosed with attention-deficit/hyperactivity disorder (ADHD).1
High-density EEG means the researchers recorded scalp electrical activity from 128 channels rather than a small clinical montage. Wake recordings were taken just before sleep and about 30 min after waking.
Across the pooled datasets, 115 participants had 2 sessions 1 week apart, and the analysis included 1,243 recordings with an average duration of 4.2 min.
The study separated ordinary EEG power into more specific components. Oscillation amplitude captures how large each rhythmic burst is, roughly indexing how synchronized the underlying neuronal population is.
Oscillation density captures how much of the recording is occupied by bursts, indexing how often that rhythm appears. Aperiodic activity is the background 1/f-like EEG slope and offset after rhythmic peaks are modeled out.
Those distinctions are not technical decoration. If amplitude and density move in different directions, a simple band-power number can hide the biology.
Snipes et al. tested this directly by comparing burst measures with spectral power, periodic power, aperiodic exponent, and aperiodic offset.
Wake Amplitude Behaved Like a Sleep Slow-Wave Marker
The researchers’ main stated hypothesis was amplitude-specific. Because sleep slow-wave activity falls with maturation and changes overnight as sleep pressure dissipates, wake oscillation amplitudes should also decrease with age and show larger overnight decreases in younger children.
the study’s discussion is explicit: “Our predictions were met on all accounts except for the sensitivity of wake amplitudes to ADHD.”1
The numbers support that partial confirmation. Across 1,234 model degrees of freedom, amplitude decreased with age (β = −0.783, t = −11.33, p < .001) and decreased in the morning after sleep (β = −3.977, t = −15.90, p < .001).
The Time x Age interaction was positive (β = 0.143, t = 7.74, p < .001), meaning the overnight amplitude drop became smaller as participants got older.
This tracks the sleep slow-wave literature. Jaramillo et al. found overnight slow-wave slope changes across development, with stronger plasticity-linked sleep EEG changes in younger participants.2
Snipes et al. 2023 also found that wake oscillation amplitude changed with time awake in adults, supporting the idea that wake amplitude can carry sleep-pressure information.3
Amplitude still should not be treated as a clean substitute for sleep slow-wave activity. In adults, Snipes et al. reported that the overnight decrease in wake amplitudes was near 0 µV, unlike sleep slow-wave slopes or amplitudes.
They proposed one plausible reason: evening recordings occurred in the wake maintenance zone, the circadian window before habitual bedtime when alertness can rise and EEG sleep-pressure markers can be counteracted.
Alpha Density Decreased Overnight in Children and Increased in Adults
The sharper finding is density. Oscillation density decreased with age overall (β = −3.540, t = −2.11, p = .035) and decreased in the morning after sleep overall (β = −77.506, t = −10.39, p < .001), but the Time x Age interaction was stronger than either headline would suggest (β = 4.878, t = 8.85, p < .001).
The age correlation with overnight density change was r = .57.
Plain English: children and adults did more than show different sizes of the same overnight density effect. Density moved in different directions.
The paper states that oscillation densities “decreased overnight in children under 15 and increased in young adults.”1
The frequency-specific analysis makes the reversal more concrete. Theta (4–7 Hz) was rare overall, most prevalent in the youngest children, and shifted more frontally with age.
Alpha (8–11 Hz) began as a midline occipital cluster in young children, added central peaks around ages 7–10, and became a broader occipital-parietal pattern across adolescence and adulthood. Low beta (12–16 Hz) was nearly absent in the youngest group and decreased overnight across ages.
For alpha density, the developmental switch was the point. In 7–10-year-olds, the main occipital alpha spot decreased overnight.
By ages 10–14, central alpha began increasing overnight while the occipital decrease weakened. In adolescents and adults, overnight alpha-density increases spread across the scalp and peaked over occipital-parietal regions.
That does not establish sleep is “bad” for children’s alpha rhythms. It means the same frequency label can refer to different developmental mixtures of rhythm source, circadian phase, sleep inertia, and function.
Cellier et al. likewise reported substantial theta and alpha reorganization from ages 3 to 24, and Tröndle et al. showed why separating periodic alpha from aperiodic background signal changes the interpretation of alpha maturation.4,5
ADHD Did Not Leave a Reliable Wake-EEG Signature After Good-Sleep Screening
The ADHD result is easy to overread in both directions. Snipes et al. included 58 participants with ADHD, which is large for a high-density pediatric EEG dataset, and the study expected lower wake amplitudes because earlier work found lower sleep slow-wave activity in ADHD.1
That prediction failed in this dataset. Amplitude was not significantly different in ADHD (β = −0.574, t = −0.89, p = .373).
Density was not different either (β = −0.154, t = −0.01, p = .992). Exponent (β = 0.028, p = .284) and offset (β = 0.021, p = .579) were also not significant, and the topographic channel analysis found no FDR-corrected ADHD effects.
The most useful interpretation is not that ADHD has no EEG correlates. The study screened participants for habitual good sleep quality, and the discussion notes that roughly 40–55% of children with ADHD report sleep deficits in other literature.
If prior ADHD EEG differences partly reflected poorer sleep, daytime sleepiness, medication mixture, or resting-task differences, a good-sleep screened sample could reduce those apparent group differences.
For diagnostic use, this is a caution flag. A wake EEG marker that changes with time of recording, age, task, and sleep quality is not an intrinsic ADHD marker unless those factors are controlled.
Fattinger et al. had already shown that theta waves in children’s waking EEG can resemble local aspects of sleep during wakefulness; Snipes et al. extend that caution to burst amplitude, density, and aperiodic measures across development.6
Aperiodic EEG Carried Developmental Signal Without a Clean Sleep-Pressure Readout
Aperiodic EEG is the non-rhythmic background slope of the signal. In simplified terms, a steeper exponent means power drops off faster at higher frequencies, while the offset reflects overall background power.
These measures have become popular because they can separate broadband background activity from named rhythms such as theta or alpha.
Snipes et al. found strong developmental effects. Exponents became shallower with age (β = −0.027, t = −9.23, p < .001), and offsets decreased even more strongly with age (β = −0.067, t = −16.59, p < .001).
Those patterns agree with developmental aperiodic EEG work by Favaro et al. and Hill et al., where wake and sleep aperiodic activity changed across childhood and adolescence.7,8
The overnight pattern was less sleep-pressure-like. Exponents became steeper after sleep (β = 0.099, t = 5.51, p < .001), but the Time x Age interaction only trended (β = −0.003, t = −1.91, p = .056).
Offsets had no significant main overnight effect (β = 0.029, p = .100), even though the Time x Age interaction was significant (β = −0.004, t = −3.11, p = .002).
That pattern is why the paper lands on amplitude rather than aperiodic exponent or offset as the closest wake analog to sleep slow-wave activity. Aperiodic markers carried developmental information, but they did not reproduce the full expected combination of age decline, overnight change, and larger overnight change in younger children.
Simple Band Power Would Blur Amplitude and Density
Classical EEG analysis often compresses signals into delta, theta, alpha, beta, and gamma power. Snipes et al. used a more granular approach because power can rise when bursts get larger, when bursts become more frequent, or when the background signal changes.
The correlations show the split. Ordinary power was most related to amplitude (r = .90; mixed-effects t = 39.3), while periodic power was most related to density (r = .85; mixed-effects t = 46.9).
The differences in those correlation strengths were statistically significant (p < .001).1
the direction can diverge. In 14–18-year-olds, oscillation densities increased overnight while periodic power decreased, likely because amplitude decreases pulled the power summary downward.
If a study only reports band power, it can miss whether the real change is burst size, burst occurrence, background activity, or some mixture.
The Snipes et al. 2022 “theta paradox” paper is useful here because theta can reflect sleep pressure and cognitive control depending on source, task, and timing.9 The 2026 paper makes a parallel point for development: frequency labels are not enough when the source, density, amplitude, and time-of-day context are moving together.
Pediatric EEG Studies Need Sleep History and Clock Time Built In
The most defensible clinical implication is methodological. Wake EEG in a child or teenager is more than a snapshot of trait brain function.
It is also a state-dependent readout shaped by how long the person has been awake, when the recording happened, how much sleep they had, and where they are in development.
That does not make EEG useless. It raises the standard for interpretation.
Pediatric EEG studies that compare ADHD vs control groups, anxiety vs control groups, medicated vs unmedicated groups, or intervention vs baseline should record and model sleep history rather than treating it as noise.
Useful minimum controls would include recent sleep duration, subjective sleepiness, time since waking, clock time, medication timing, task type, and age as more than a nuisance covariate. the study’s dataset was rich enough to show age-by-time interactions; many clinical EEG datasets are not.
For mental-health readers, the takeaway is calibration. Sleep matters for attention, mood, and behavior, but this paper does not prove that changing sleep schedules will normalize a child’s EEG or improve symptoms.
It shows that wake EEG biomarkers can be misleading when sleep/wake history and developmental stage are not part of the design.
Limits: Circadian Timing, Sparse Younger Data, and Mixed Tasks
The main limitation is built into the design: evening-to-morning change cannot separate sleep homeostasis from circadian rhythm. A true separation would require more intensive protocols, such as repeated wake recordings across the day, shifted sleep windows, controlled sleep restriction, or sleep deprivation.
Those designs are harder to justify in children.
The youngest age range is also thin. Only 11 participants came from the 3.5–8.0-year-old dataset, and Snipes et al. note that early childhood may involve nonlinear EEG changes as synaptic growth gives way to pruning.
A linear age model across 3.5–24.7 years may miss sharper bends around early childhood.
The pooled design adds power but also heterogeneity. Participants came from several datasets with different tasks, sessions, medication histories, and recording contexts.
The core amplitude and density results remained significant in more uniform subsets, but a purpose-built longitudinal design would be cleaner.
Finally, the study measured EEG, not school grades, depression, anxiety, executive function, or treatment response. The results are strongest as a warning about EEG interpretation.
Behavioral and clinical consequences still need direct testing.
Questions About Wake EEG, Sleep, and Development
Did Snipes et al. test sleep deprivation in children?
No. The study compared evening wake EEG before sleep with morning wake EEG about 30 min after waking.
That design can detect overnight change, but it cannot cleanly separate sleep pressure from circadian timing, sleep inertia, or morning melatonin differences.
Why does alpha density decreasing in children but increasing in adults matter?
It means alpha density is not a single lifespan-invariant marker. A decrease in a 7-year-old and an increase in a young adult may both follow sleep, but they likely reflect different developmental rhythm sources, circadian phase, or functional states.
Does the child alpha-density decrease mean sleep harms children’s waking brain rhythms?
No. The paper did not test harm or performance.
It measured how often alpha bursts appeared in wake EEG before and after sleep. The safer reading is that children’s morning EEG is developmentally and state dependent, not that sleep produces a bad neural pattern.
Can wake EEG amplitude replace sleep slow-wave activity as a maturation marker?
Not yet. Amplitude followed the predicted age and overnight pattern, but the authors emphasized that wake amplitude is affected by circadian and state factors.
It may carry related information, but sleep slow-wave activity remains the cleaner sleep-pressure measure.
Did the ADHD null result prove ADHD has no EEG signature?
No. It showed that this good-sleep screened, mixed-medication sample did not have a reliable wake EEG difference after correction.
The result argues against using these wake measures as intrinsic ADHD markers without stronger control of sleep quality, medication, task, age, and recording time.
How should pediatric EEG studies use this finding?
They should treat sleep/wake history and clock time as design variables, not afterthoughts. At minimum, studies should record recent sleep, time since waking, recording time, task, medication timing, and age, then model whether group differences survive those controls.
References
- Snipes S, Jaramillo V, Krugliakova E, Volk C, Furrer M, Studler M, et al. The interaction between sleep and development on wake EEG oscillations. eNeuro. 2026. doi:10.1523/ENEURO.0384-25.2026
- Jaramillo V, Volk C, Maric A, Furrer M, Fattinger S, Kurth S, et al. Characterization of overnight slow-wave slope changes across development in an age-, amplitude-, and region-dependent manner. Sleep. 2020;43(9):zsaa038. doi:10.1093/sleep/zsaa038
- Snipes S, Meier E, Meissner SN, Landolt H-P, Huber R. How and when EEG reflects changes in neuronal connectivity due to time awake. iScience. 2023;26:107138. doi:10.1016/j.isci.2023.107138
- Cellier D, Riddle J, Petersen I, Hwang K. The development of theta and alpha neural oscillations from ages 3 to 24 years. Developmental Cognitive Neuroscience. 2021;50:100969. doi:10.1016/j.dcn.2021.100969
- Tröndle M, Popov T, Dziemian S, Langer N. Decomposing the role of alpha oscillations during brain maturation. eLife. 2022;11:e77571. doi:10.7554/eLife.77571
- Fattinger S, Kurth S, Ringli M, Jenni OG, Huber R. Theta waves in children’s waking electroencephalogram resemble local aspects of sleep during wakefulness. Scientific Reports. 2017;7:11187. doi:10.1038/s41598-017-11577-3
- Favaro J, Colombo MA, Mikulan E, Sartori S, Nosadini M, Pelizza MF, et al. The maturation of aperiodic EEG activity across development reveals a progressive differentiation of wakefulness from sleep. NeuroImage. 2023;277:120264. doi:10.1016/j.neuroimage.2023.120264
- Hill AT, Clark GM, Bigelow FJ, Lum JAG, Enticott PG. Periodic and aperiodic neural activity displays age-dependent changes across early-to-middle childhood. Developmental Cognitive Neuroscience. 2022;54:101076. doi:10.1016/j.dcn.2022.101076
- Snipes S, Krugliakova E, Meier E, Huber R. The theta paradox: 4–8 Hz EEG oscillations reflect both sleep pressure and cognitive control. Journal of Neuroscience. 2022;42(45):8569–8586. doi:10.1523/JNEUROSCI.1063-22.2022