A 2026 multimodal MRI analysis of 509 healthy adults found that fear acquisition, extinction learning, and renewal were not predicted by one generic “fear circuit” score: functional connectivity predicted acquisition, structural connectivity predicted extinction, and effective connectivity was the only renewal signal.1 The extinction result is the clinically interesting piece because exposure therapy depends on building new inhibitory learning, but the paper is still a laboratory prediction study rather than a treatment-response test.
Research Highlights
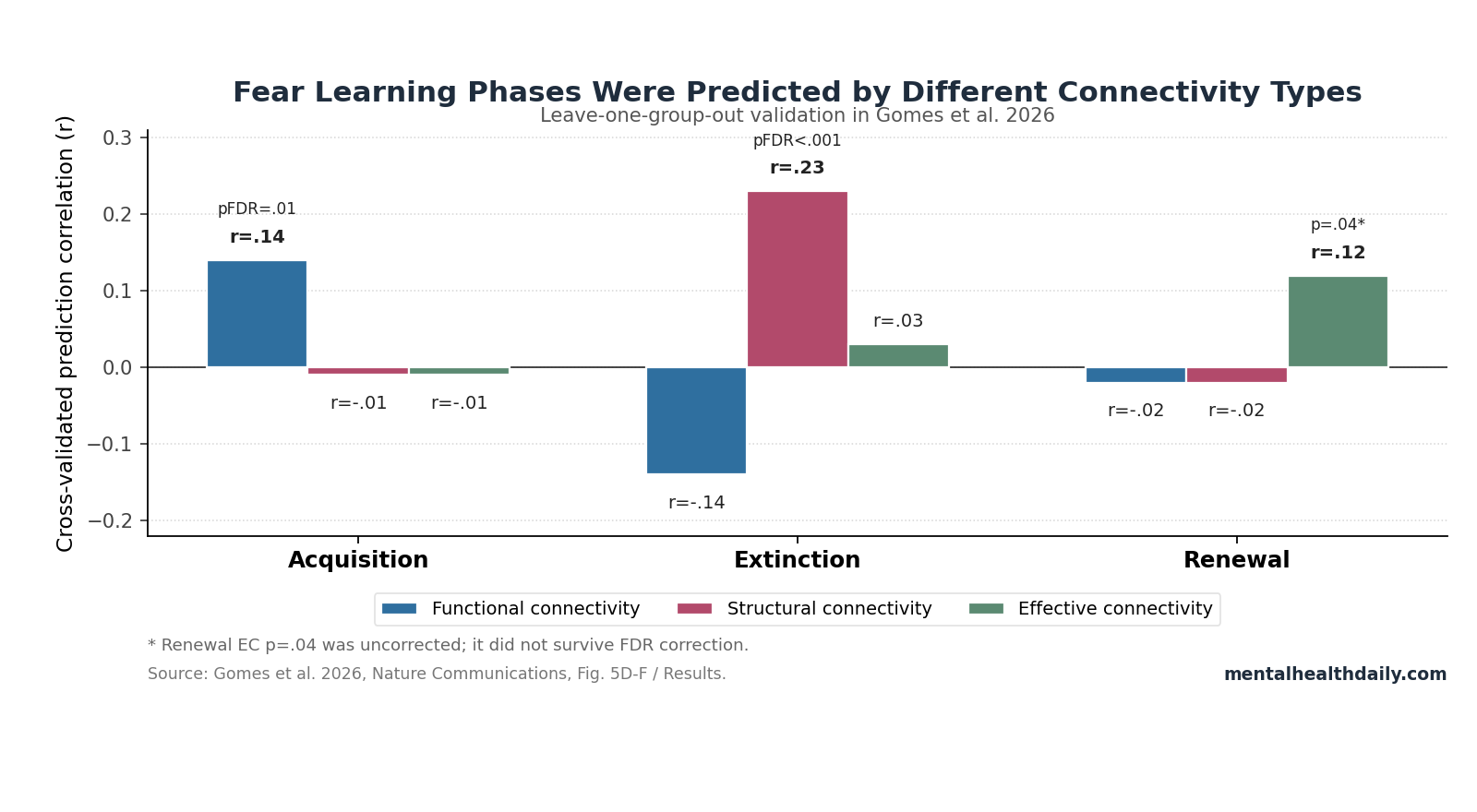
- Connectivity split by learning phase: Leave-one-group-out validation found acquisition predicted only by functional connectivity (r = .14, pFDR = .01), extinction predicted only by structural connectivity (r = .23, pFDR < .001), and renewal numerically best for effective connectivity (r = .12, p = .04 uncorrected).1
- The sample was large for this literature: Gomes et al. analyzed 509 resting-state fMRI datasets across 6 studies and 467 diffusion MRI datasets across 5 studies, using both fear-learning and cognitive predictive-learning tasks.1
- Extinction was not memory erasure: Extinction creates a second inhibitory trace; renewal occurs when context lets the older association reappear, a framework supported by 2021 extinction-mechanism work and human renewal studies.2,4
- Structural wiring carried the extinction signal: Several extinction predictors involved the dorsal anterior cingulate cortex (ACC), with ACC appearances exceeding other regions in the structural-connectivity resampling analysis (all z > 8, pFDR < .001).1
- Clinical translation is not ready: The study used healthy participants and laboratory tasks; renewal prediction came from only 2 studies, and the cross-validated effective-connectivity renewal signal did not survive FDR correction.1
Fear extinction means learning that a cue once paired with threat no longer predicts threat. In exposure therapy, the patient repeatedly encounters feared cues in safe conditions so the brain learns a competing safety association; the original fear memory can still return under stress, time, or a context shift.2,7
That non-erasure model matters for interpreting Gomes et al. The paper is not saying “the amygdala causes anxiety” or that one resting scan can choose a treatment. It is asking a narrower and more useful question: which baseline network properties help explain why some people acquire threat associations, extinguish them, or show renewal more strongly than others?
Functional Connectivity Predicted Acquisition, Not Extinction
Functional connectivity means regions fluctuate together during resting-state functional magnetic resonance imaging (fMRI). It is correlational, not directional: 2 regions can move together without proving which region drives the other.
In Gomes et al., acquisition was the phase where functional connectivity carried the clearest whole-sample prediction signal. The acquisition model selected connections involving cerebellar nuclei, hippocampus, prefrontal cortex, amygdala, and ACC, with ACC and hippocampus appearing most reliably in resampling.
Leave-one-group-out validation kept the effect modest but significant: functional connectivity predicted acquisition at r = .14, pFDR = .01, while effective and structural connectivity were both r = −.01 with pFDR = 1.00.1
Plain English: resting co-activation patterns helped predict who learned the initial association better. Older human and animal work already placed ACC, hippocampus, amygdala, and prefrontal regions inside acquisition and appraisal circuits; Gomes et al. add that this functional co-activation profile belongs mainly to acquisition, while extinction depends more on structural wiring.
Structural Connectivity Was the Strongest Extinction Predictor
Structural connectivity means white-matter pathway strength estimated from diffusion MRI. It is closer to anatomical wiring than moment-to-moment fMRI co-activation, although tractography estimates still infer pathways rather than measuring axons directly.
Extinction learning reversed the modality pattern. Across all paradigms, Gomes et al. found no significant functional- or effective-connectivity predictors of extinction.
Structural connections carried the extinction result instead, especially connections involving ACC: right hippocampus to right ACC, left cerebellar nuclei to right ACC, and bilateral amygdala to ACC were repeatedly selected. In resampling, ACC had the largest number of appearances in significant structural-connectivity extinction predictors, with pairwise tests against other regions at z > 8 and pFDR < .001.1
Leave-one-group-out validation put the extinction result in direct numerical form: structural connectivity predicted extinction at r = .23, pFDR < .001, while effective connectivity was only r = .03 (pFDR = .23) and functional connectivity was r = −.14 (pFDR = 1.00). The cross-modality comparison also showed structural-connectivity prediction performance for extinction was greater than for acquisition or renewal (z > 3.2, pFDR < .01).1
That is the main correction to a loose headline. Resting connectivity did predict extinction learning, but the strongest extinction signal was structural.
The practical interpretation is closer to “baseline wiring in the extinction network may constrain how easily a person builds inhibitory safety learning” than to “resting fMRI activity predicts exposure therapy response.”
Effective Connectivity Carried the Tentative Renewal Signal
Effective connectivity means a model-based estimate of directed influence between regions. Gomes et al. used spectral dynamic causal modeling, a method that estimates whether one region’s resting activity tends to increase or decrease activity in another region.
Renewal is the return of a response after extinction when the cue is encountered in the acquisition context again. Only 2 of the 6 studies tested renewal, so the renewal evidence is weaker by design.
In the combined renewal analysis, functional and structural connectivity did not significantly predict individual differences. Effective connectivity did, mainly through hippocampal disinhibition by amygdala and prefrontal cortex, plus greater prefrontal inhibition of ACC.1
Renewal evidence was weaker than extinction evidence: effective connectivity was the only modality with a positive renewal signal in leave-one-group-out validation (r = .12, p = .04), but that p-value was uncorrected. Functional and structural connectivity were both r = −.02, with pFDR = .63 and 1.00.
The result points toward a renewal mechanism, but it should not be sold as a stable clinical predictor yet.1
Fear Extinction Is a Network Problem, Not an Amygdala Story
The older “amygdala equals fear” shortcut is too crude for this paper. Gomes et al. analyzed a network that included amygdala, hippocampus, dorsal ACC, ventromedial prefrontal cortex, and cerebellar nuclei.
Each region has a different job in the learning system:
- Amygdala: tags stimulus-outcome relevance and participates in fear-expression and salience processing.
- Hippocampus: carries contextual information, which is central for renewal and extinction recall.3,4
- Ventromedial prefrontal cortex: supports safety learning and regulation of amygdala output during extinction recall.3
- Dorsal ACC: appears repeatedly in appraisal, conflict, and extinction-network coordination; in Gomes et al., it was the central structural hub for extinction prediction.1,5
- Cerebellar nuclei: contribute prediction-error timing and associative-learning signals rather than only motor coordination.6
Human fMRI meta-analysis also supports a distributed view. Fullana et al. found fear extinction involved a broad network rather than a single fear center, and older extinction-recall work by Milad et al. linked successful recall to coordinated ventromedial prefrontal and hippocampal activity.3,5
Exposure Therapy Should Not Be Reduced to One Brain Scan
Exposure-based cognitive-behavioral therapy uses the same core learning principle: repeated safe exposure builds inhibitory learning that can suppress the old threat association. Craske et al. framed modern exposure therapy around inhibitory learning and inhibitory regulation, emphasizing expectancy violation, varied contexts, and relapse prevention rather than simple fear reduction inside one session.7
Gomes et al. fit that framework because the study separates extinction from acquisition. A person can acquire threat associations efficiently without having the same neural profile for extinguishing them; in the paper, acquisition and extinction were correlated at only r = .16, sharing roughly 3% of variance.1
This helps explain why “learns fast” is not the same as “extinguishes fast.”
Clinical overreach would be a mistake. The study did not enroll patients starting exposure therapy, did not randomize people to different treatment intensities, and did not test whether a therapist should change protocol based on ACC-amygdala tractography.
Clinical translation: structural features of the extinction network are plausible predictors to test in future treatment studies, especially when combined with symptom course, avoidance behavior, task-based learning, and relapse outcomes.
What the Gomes Study Can and Cannot Support
What it supports: resting-state network properties can explain some individual differences in laboratory acquisition, extinction, and renewal. The phase-specific pattern is the key result, with functional connectivity strongest for acquisition, structural connectivity strongest for extinction, and effective connectivity most relevant to renewal.
What it does not support: a clinical test for who will or will not respond to exposure therapy. Even the validated correlations were modest; the paper’s limitations section notes that generalizability improvements were sometimes numerically small, often around .03 in the cross-paradigm MSE-difference analyses, and that LOGO/task-fMRI/simulation estimates ranged roughly .07–.23.1
Why the direction still matters: if extinction depends more on structural wiring than momentary functional coupling, treatment-response research should not rely only on resting fMRI co-activation maps. Diffusion MRI, task learning curves, context-dependent renewal measures, and longitudinal clinical outcomes may need to be modeled together.
Questions About Brain Connectivity and Fear Extinction
Did Gomes et al. prove that resting brain scans can predict exposure therapy response?
No. The study involved healthy adults in laboratory fear-learning and predictive-learning tasks.
It supports a research hypothesis about who acquires, extinguishes, or renews learned associations more strongly; it does not validate a treatment-selection biomarker.
Which connectivity type best predicted extinction learning?
Structural connectivity. In leave-one-group-out validation, structural connectivity predicted extinction at r = .23 with pFDR < .001, while functional connectivity and effective connectivity did not generalize for extinction.1
Why did renewal look weaker than acquisition or extinction?
Renewal was measured in only 2 studies. Effective connectivity was the only positive cross-validated renewal signal (r = .12, p = .04 uncorrected), but it did not survive the same FDR-corrected standard as the acquisition and extinction results.1
Does extinction erase fear memories?
No. Extinction usually builds a competing inhibitory association.
The older association can return through renewal, reinstatement, or spontaneous recovery, which is why exposure therapy often emphasizes varied practice contexts and relapse-prevention planning.2,7
What should future clinical studies test?
A strong next step would enroll patients before exposure therapy, measure structural and functional connectivity plus task extinction and renewal, and test whether baseline ACC-amygdala-hippocampal network features predict symptom change and relapse beyond ordinary clinical variables.
References
- Gomes CA et al. Predicting individual differences of fear and cognitive learning and extinction. Nature Communications. 2026;17:3780. doi:10.1038/s41467-026-71830-0
- Bouton ME, Maren S, McNally GP. Behavioral and neurobiological mechanisms of Pavlovian and instrumental extinction learning. Physiological Reviews. 2021;101(2):611-681. doi:10.1152/physrev.00016.2020
- Milad MR et al. Recall of fear extinction in humans activates the ventromedial prefrontal cortex and hippocampus in concert. Biological Psychiatry. 2007;62(5):446-454. doi:10.1016/j.biopsych.2006.10.011
- Lissek S et al. Hippocampal activation during extinction learning predicts occurrence of the renewal effect in extinction recall. NeuroImage. 2013;81:131-143. doi:10.1016/j.neuroimage.2013.05.025
- Fullana MA et al. Fear extinction in the human brain: a meta-analysis of fMRI studies in healthy participants. Neuroscience & Biobehavioral Reviews. 2018;88:16-25. doi:10.1016/j.neubiorev.2018.03.002
- Doubliez A et al. The cerebellum and fear extinction: evidence from rodent and human studies. Frontiers in Systems Neuroscience. 2023;17:1166166. doi:10.3389/fnsys.2023.1166166
- Craske MG et al. Optimizing exposure therapy for anxiety disorders: an inhibitory learning and inhibitory regulation approach. Verhaltenstherapie. 2018;28(1):24-31. doi:10.1159/000485415
