A 2026 retrospective case series of 22 adults with treatment-resistant depression and comorbid PTSD found that trauma re-experiencing during intranasal esketamine disappeared over later sessions in 72.7% of patients, while 27.3% stopped esketamine because the flashbacks persisted.1
Research Highlights
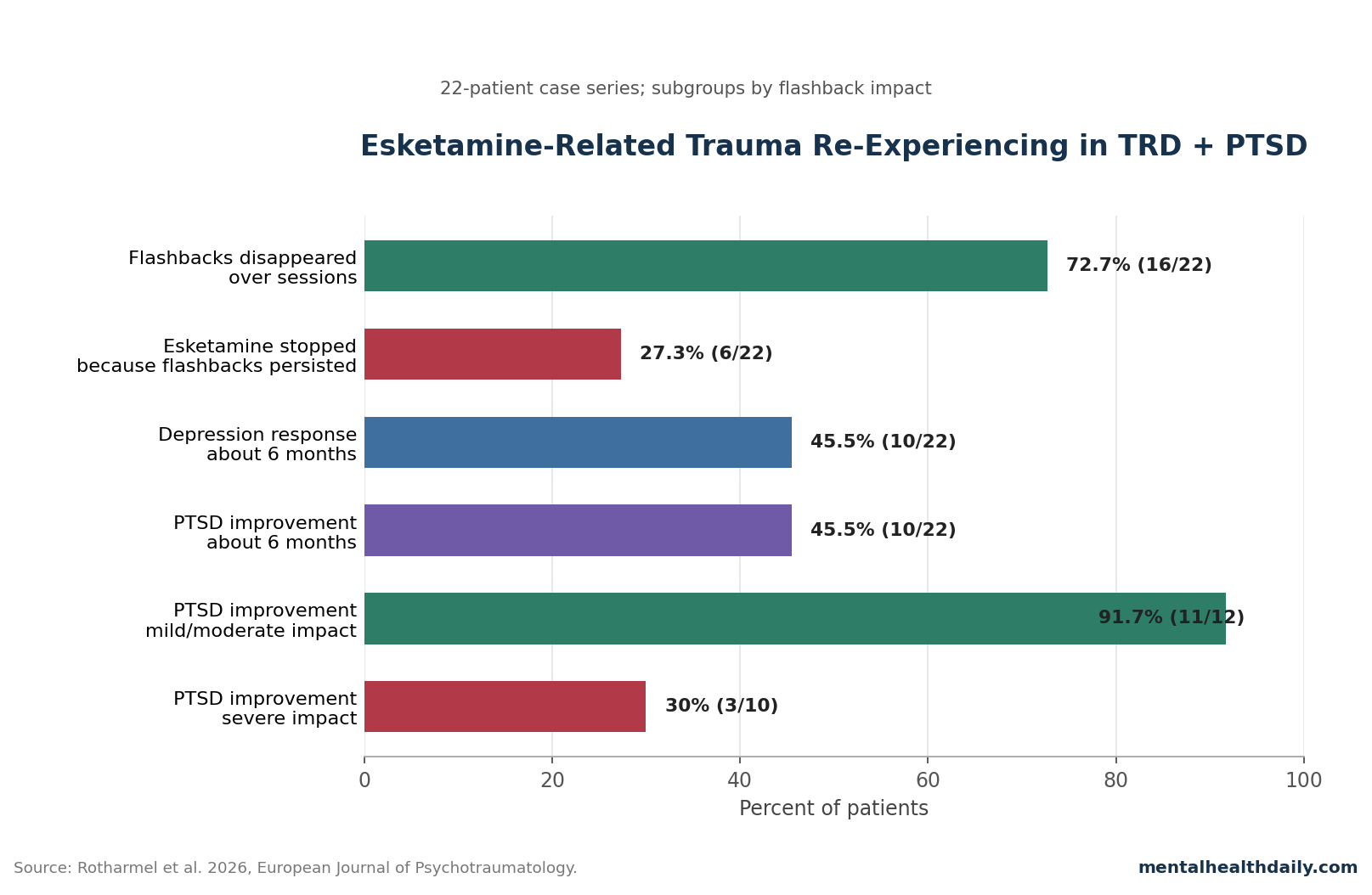
- Flashbacks usually faded, but not always: In 16 of 22 patients (72.7%), esketamine-related trauma re-experiencing disappeared as sessions continued; in 6 of 22 (27.3%), treatment stopped because flashbacks persisted.1
- The first episodes came early: The first flashback appeared at a mean of session 4.4, with 40.9% during session 1 and 77.3% during the first 5 esketamine sessions.1
- Clinical outcomes were not uniformly bad: About 6 months after treatment start, depression response was 45.5%, depression remission was 22.7%, PTSD improvement was 45.5%, and PTSD remission was 18.2%.1
- Severity of the flashback experience mattered: PTSD improvement occurred in 91.7% of patients whose flashback impact was mild or moderate, vs. 30% when the impact was severe (p = .006).1
- The design cannot estimate prevalence: The 22-person series included only patients already identified as having at least 1 esketamine-related trauma re-experiencing episode, so it cannot tell clinics how often this happens among all TRD/PTSD patients receiving esketamine.1
Esketamine is the S-enantiomer of ketamine, delivered as a supervised nasal spray for treatment-resistant depression. Trauma re-experiencing means involuntary trauma-memory reliving during the drug session: flashback-like images, emotions, body sensations, or autobiographical scenes that feel tied to a past traumatic event.
Rothärmel et al. did not show that esketamine commonly causes flashbacks in PTSD, because the study deliberately selected patients in whom clinicians had already noticed at least 1 episode. The sharper finding is narrower and more useful: when trauma memories emerged during esketamine, most episodes faded across later sessions, a substantial minority forced discontinuation, and response looked more likely when the flashback was emotionally tolerable rather than overwhelming.1
22 TRD/PTSD Patients Had Flashbacks During Medically Monitored Esketamine
The case series came from 11 French psychiatric departments and included adults ages 21-62 with moderate-to-severe treatment-resistant depression (TRD) and clinician-diagnosed comorbid PTSD. TRD meant insufficient remission after at least 2 adequate antidepressant trials for unipolar depression, or insufficient remission after guideline-level mood-stabilizing treatment for bipolar depression.1
All 22 patients had at least 1 trauma re-experiencing episode during intranasal esketamine. That inclusion criterion is the hinge of the whole paper: the denominator is not “all people with PTSD who received esketamine.” It is “patients with TRD/PTSD whose treating team identified a trauma-memory episode during esketamine.”
The sample was clinically severe. Mean current depressive-episode duration was 35.7 months, the mean Maudsley staging level was 8.4, 86.4% had complex PTSD, 63.6% had an anxiety disorder, 40.9% had an eating disorder, 22.7% had alcohol use disorder, and 77.3% had previous suicide attempts.1
Monitoring context: Esketamine was given in full or partial hospitalization settings, with blood tests and electrocardiogram screening before initiation and at least 120 minutes of post-dose monitoring. Dosing followed standard supervised-esketamine schedules: 56 or 84 mg twice weekly during weeks 1-4, then weekly during weeks 5-8, then every 1-2 weeks thereafter when clinically appropriate.1
40.9% Had a Flashback in Session 1 and 77.3% by Session 5
Flashbacks appeared early. The first esketamine-related flashback occurred at a mean of the 4.4th session, with a median of 2.5 sessions and a wide range from session 1 to session 22.
Nine patients (40.9%) had the first flashback during the first esketamine session, and 17 patients (77.3%) had it within the first 5 sessions.1
Symptoms were still high during the week when flashbacks occurred. Mean Montgomery-Åsberg Depression Rating Scale (MADRS) score was 29.6 ± 9.8 across all 22 patients, and mean PTSD Checklist for DSM-5 (PCL-5) score was 52.9 ± 15.0 among the 9 patients with available PCL-5 data.
The mean Clinical Global Impression-Suicide Scale score was 2.2 ± 1.6 among 20 patients.1
Flashbacks were more than ordinary dissociation with a different label. Standard esketamine dissociation can involve altered perception, derealization, time distortion, or floating sensations.
In this series, the episodes were trauma-content specific: rape, physical violence, discovery of a dead person, delivery after pregnancy denial, or forced abortion.1
The trauma-memory episodes also exposed information clinicians did not already have. In 8 of 22 patients (36.4%), the flashback revealed the trauma to the medical team; in 6 of 22 (27.3%), the episode involved events the patient reported having forgotten.
Episodes began within minutes after esketamine administration, peaked during drug effects, resolved within 2 hours, and did not occur outside esketamine sessions.1
72.7% Resolved, 27.3% Stopped Esketamine, and PTSD Response Split by Flashback Intensity
Rothärmel et al. reported 2 clinically different pathways. In 16 patients (72.7%), trauma re-experiencing disappeared as esketamine sessions progressed.
In 6 patients (27.3%), esketamine was stopped because flashbacks persisted; discontinuation occurred after an average of 6 ± 3 sessions.1
The discontinuation group still contained a partial depression signal. Among the 6 patients who stopped, 3 had a partial depression response without PTSD response, and 3 had no response for either depression or PTSD.
Three patients wanted to keep working on trauma through cognitive behavioral therapy (CBT) or eye movement desensitization and reprocessing (EMDR) after stopping esketamine.1
Across the full sample, depression response was 45.5% (10 of 22), depression remission was 22.7% (5 of 22), PTSD improvement was 45.5% (10 of 22), and PTSD remission was 18.2% (4 of 22), assessed around 6 months after treatment start. Depression and PTSD outcomes tracked together: response rates were associated at p = .002, and remission rates at p < .001.1
Flashback emotional intensity was the most clinically interesting split. PTSD improvement occurred in 91.7% of patients whose initial flashback impact was mild or moderate, compared with 30% of patients whose impact was severe (p = .006).
The paper did not prove that moderate flashbacks caused improvement. It did suggest a dose-of-distress problem familiar from exposure therapy: tolerable activation in a safe setting may support processing, while severe activation can become destabilizing.1
Ketamine/PTSD Evidence Is Positive in Some Small Trials and Negative in a Larger Military RCT
The Rothärmel case series sits inside an uneven ketamine/PTSD literature. A 2022 open-label pilot by the same French group tested intranasal esketamine in 11 patients with TRD and comorbid chronic PTSD, reporting improvements in both depression and PTSD symptoms over a 6-month treatment window.
That earlier study made the 2026 flashback series plausible: clinicians had already been seeing both symptom improvement and trauma-content emergence in the same comorbid population.2
Repeated intravenous ketamine has stronger placebo-controlled PTSD data in at least 1 small civilian trial. Feder et al. randomized 30 patients with chronic PTSD to 6 ketamine or midazolam infusions over 2 weeks.
At week 2, ketamine was associated with a 11.88-point lower Clinician-Administered PTSD Scale for DSM-5 (CAPS-5) score than midazolam, with an effect size of d = 1.13; 67% of ketamine patients responded vs. 20% of midazolam patients.3
A larger military trial was less encouraging. Abdallah et al. randomized 158 veterans and service members with antidepressant-resistant PTSD symptoms to 8 infusions of placebo, low-dose ketamine (0.2 mg/kg), or standard-dose ketamine (0.5 mg/kg).
The trial failed to show a significant dose-related effect on PTSD symptoms, even though ketamine produced dose-related dissociative and psychotomimetic effects that returned to baseline within 2 hours.4
That mixed landscape matters for interpretation. The 2026 case series should not be treated as proof that esketamine treats PTSD.
It is better read as a clinical-process paper: when esketamine is used for TRD in people who also have PTSD, trauma memories can come forward, and the treatment setting determines whether that event becomes unsupported distress or a therapeutically usable opening.
Trauma-Aware Support Is the Difference Between Side Effect and Processing Opportunity
The paper’s most practical claim is not that flashbacks are good. It is that flashbacks require preparation.
Patients should know before treatment that trauma re-experiencing can happen, clinicians should ask about trauma history directly, and the team monitoring esketamine should know how to ground a patient who is reliving trauma content.1
Rothärmel et al. described several mechanisms that could make esketamine a double-edged tool in PTSD. NMDA receptor antagonism means esketamine blocks N-methyl-D-aspartate glutamate receptors, producing downstream glutamate release, altered perception, and increased synaptic plasticity.
In trauma terms, that altered state may loosen access to autobiographical memory, reduce avoidance, and change how fear memories reconsolidate after they are reactivated.1
Adjacent mechanistic work points in the same direction without proving the case-series mechanism. Duek et al. randomized 27 people with PTSD to ketamine or midazolam after retrieval of the traumatic memory, followed by 4 days of brief exposure therapy.
PTSD symptoms improved in both groups, but ketamine recipients showed lower amygdala reactivation to trauma memories and marginally lower hippocampal reactivation after treatment.5
The clinical fork is intensity plus support: a tolerable flashback in a monitored, trauma-aware setting may create a window for exposure, grounding, and integration; an overwhelming flashback in a medication-only setting can become another aversive experience. The 91.7% vs. 30% PTSD-improvement split by mild/moderate vs. severe flashback impact is not causal proof, but it is too clinically direct to bury.1
What This Case Series Cannot Tell Clinics
Several limits should stay attached to every clinical reading of the paper.
- No prevalence estimate: Because the case series included only patients with identified esketamine-related flashbacks, it cannot estimate how often flashbacks occur among all esketamine-treated TRD/PTSD patients.1
- No causal test: The study cannot show whether flashbacks caused improvement, marked patients who were already likely to improve, or simply appeared alongside symptom changes driven by esketamine’s antidepressant effects.1
- No structured PTSD diagnosis for everyone: PTSD diagnoses came from treating clinicians using DSM-5 and ICD-11 criteria, and only 9 patients had PCL-5 scores available during the flashback week.1
- No uniform PTSD outcome timing: Improvement and remission were based partly on clinical judgment, with follow-up timing ranging from 1 to 15 months.1
Those limits do not erase the observation. They keep the observation in its proper lane.
Esketamine-session flashbacks are real enough to plan for, early enough to discuss before dose 1, and consequential enough that severe episodes may stop treatment. They are not yet measured well enough to be sold as a predictable therapeutic mechanism.
Questions About Esketamine Flashbacks in PTSD
Does this mean PTSD patients should avoid esketamine for depression?
No categorical avoidance follows from this case series. The paper argues for trauma screening, explicit consent, and trauma-aware monitoring, not for excluding every patient with PTSD.
The discontinuation rate was real at 27.3%, but 72.7% had flashbacks fade over sessions and 45.5% had depression response.1
Can the paper tell a clinic how common esketamine flashbacks are?
No. The study selected 22 patients who already had at least 1 identified trauma re-experiencing episode.
A prevalence study would need to start with all TRD/PTSD patients receiving esketamine and prospectively track flashbacks using a standard definition.1
Was trauma re-experiencing a bad sign when it happened?
It depended on severity and whether treatment could continue safely. Mild or moderate flashback impact was associated with 91.7% PTSD improvement, while severe impact was associated with 30% improvement and sometimes led to stopping esketamine.
The association is clinically useful but not proof that flashbacks caused recovery.1
How should esketamine clinics change consent for TRD patients with PTSD?
Consent should name the possibility of trauma re-experiencing, explain that episodes may appear early, and describe what staff will do if flashbacks occur. The case series also supports asking directly about trauma history before treatment, because 36.4% of cases revealed trauma information that the team had not previously known.1
Is this the same thing as ketamine-assisted psychotherapy?
No. Standard intranasal esketamine for TRD is a medication protocol with monitoring.
Ketamine-assisted psychotherapy deliberately pairs the altered state with psychotherapy. The Rothärmel series suggests standard esketamine clinics treating PTSD-comorbid patients may need some trauma-informed elements from psychotherapy models, especially grounding, support during re-experiencing, and post-session integration.1,5
References
- Rothärmel M, Mekaoui L, Kazour F, et al. Trauma re-experiencing episodes during esketamine treatment in patients with treatment-resistant depression and comorbid PTSD: a retrospective case series. European Journal of Psychotraumatology. 2026. doi:10.1080/20008066.2025.2609425
- Rothärmel M, Benosman C, El-Hage W, et al. Efficacy and safety of intranasal esketamine in patients with treatment-resistant depression and comorbid chronic post-traumatic stress disorder: open-label single-arm pilot study. Frontiers in Psychiatry. 2022;13:865466. doi:10.3389/fpsyt.2022.865466
- Feder A, Costi S, Rutter SB, et al. A randomized controlled trial of repeated ketamine administration for chronic posttraumatic stress disorder. American Journal of Psychiatry. 2021;178(2):193–202. doi:10.1176/appi.ajp.2020.20050596
- Abdallah CG, Roache JD, Gueorguieva R, et al. Dose-related effects of ketamine for antidepressant-resistant symptoms of posttraumatic stress disorder in veterans and active duty military: a double-blind, randomized, placebo-controlled multi-center clinical trial. Neuropsychopharmacology. 2022;47(8):1574–1581. doi:10.1038/s41386-022-01266-9
- Duek O, Korem N, Li Y, et al. Long term structural and functional neural changes following a single infusion of ketamine in PTSD. Neuropsychopharmacology. 2023;48(11):1648–1658. doi:10.1038/s41386-023-01606-3
- Samalin L, Rothärmel M, Mekaoui L, et al. Esketamine nasal spray in patients with treatment-resistant depression: the real-world experience in the French cohort early-access programme. International Journal of Psychiatry in Clinical Practice. 2022;26(4):352–362. doi:10.1080/13651501.2022.2030757
- Feder A, Brown O, Rutter SB, et al. Combining ketamine infusions and written exposure therapy for chronic PTSD: an open-label trial. Journal of Clinical Psychiatry. 2025;86(2):24m15622. doi:10.4088/JCP.24m15622