Chronic insomnia affects 10–20% of adults. The standard medical response is a benzodiazepine-receptor agonist — zolpidem, zopiclone, eszopiclone — despite every major guideline recommending psychological treatment as first-line. The gap between guideline and prescribing pad is large, and the popular framing of CBT-I as a labor-intensive, therapist-only intervention is a meaningful part of why.
Research Highlights
- CBT-I works, and it’s the first-line treatment for chronic insomnia. Across the strongest meta-analyses, CBT-I (cognitive behavioral therapy for insomnia) produces moderate-to-large reductions on every standard sleep outcome: time to fall asleep cut by ~19 minutes, time awake at night cut by ~26 minutes, sleep efficiency up ~10 percentage points.
- Sleep hygiene alone is not CBT-I, and it isn’t effective for chronic insomnia. The two are routinely conflated. Sleep hygiene is the small “avoid caffeine, keep your room dark” handout. The active ingredients of CBT-I are sleep restriction and stimulus control — behavior changes that compress and re-anchor sleep. European guidelines explicitly do not recommend sleep hygiene as a stand-alone treatment.
- CBT-I matches or beats sleep medications long-term. Short-term, hypnotics work faster. By 3–6 months, CBT-I produces equal or better outcomes — and the gains hold after treatment stops, while medication gains require continued use.
- Self-help versions of CBT-I work. A 2026 Norwegian RCT found that a CBT-I self-help book reduced daily hypnotic use from 25.4% to 18.6% in primary-care patients prescribed sleep medications, and dropped the proportion meeting criteria for anxiety from 32.1% to 23.2%. A pamphlet of sleep hygiene advice did neither.
- The “CBT-I is too complicated and inaccessible” framing is mostly wrong. A 187-page book mailed to patients beat the standard sleep-hygiene handout in a real-world primary-care population. Digital and app-delivered CBT-I produce similar effects in head-to-head trials. The bottleneck is awareness, not feasibility.
What CBT-I Is (and Isn’t)
CBT-I is a multi-component protocol, typically delivered over 4 to 8 sessions. The two components that carry most of the clinical effect are sleep restriction therapy and stimulus control therapy. Cognitive techniques and relaxation training are usually included; sleep hygiene psychoeducation is sometimes included but isn’t the active ingredient.1
- Sleep restriction therapy compresses time-in-bed down to the hours the patient is currently sleeping, then expands it back gradually as sleep efficiency (sleep duration divided by time in bed) climbs above ~85%. It is counterintuitive — patients with insomnia typically extend their time in bed hoping to capture more sleep, which perpetuates the problem by reinforcing the bed-as-wakefulness association and lowering sleep pressure.
- Stimulus control therapy re-trains the bed-sleep association: leave the bedroom if you haven’t fallen asleep within ~20 minutes, return only when sleepy, no reading or phone use in bed, no daytime napping. Both protocols are behavioral, not pharmacological. Both are short-term uncomfortable.
Sleep hygiene, by contrast, is the standard handout: avoid caffeine and alcohol before bed, keep the room dark and cool, exercise (but not late), don’t go to bed hungry. Each item is sensible, but as a stand-alone intervention sleep hygiene fails to treat chronic insomnia. The most recent European Insomnia Guideline (Riemann et al. 2023) says this directly: sleep hygiene alone is not evidence-based treatment.2 Conflating CBT-I with sleep hygiene — common in primary care — sets patients up for the wrong intervention.
The Headline Numbers Across Meta-Analyses
The cleanest synthesis is Trauer and colleagues’ 2015 meta-analysis in Annals of Internal Medicine, pooling 20 RCTs of multi-component CBT-I in adults with chronic insomnia (n = 1,162).3 Pooled effects at post-treatment:
- Sleep onset latency (time to fall asleep): reduced by 19.03 minutes (95% CI 14.12 to 23.93).
- Wake after sleep onset (WASO — time awake during the night): reduced by 26.00 minutes (95% CI 15.48 to 36.52).
- Sleep efficiency: increased by 9.91 percentage points (95% CI 8.09 to 11.73).
- Total sleep time: increased modestly, around 8 minutes — the smallest effect of the four standard outcomes.
The total-sleep-time finding is worth flagging. CBT-I doesn’t dramatically lengthen sleep; it compresses and consolidates the sleep that’s already happening. Patients fall asleep faster, wake less, and report higher subjective quality — even if total sleep barely budges. The disconnect between “feels much better” and “added 10 minutes” reflects the role sleep efficiency, not duration, plays in subjective rest.
van Straten and colleagues’ 2018 meta-analysis in Sleep Medicine Reviews pooled 87 RCTs across multiple delivery formats — in-person, group, internet, self-help.4
Pooled effect sizes were large by convention: SMDs (standardized mean differences, a way to compare studies that used different scales) of 0.98 for insomnia severity, 0.71 for sleep efficiency, and similar effects for sleep quality. In plain terms, an SMD of ~1.0 means the average treated patient is roughly a full standard deviation better off than the average control patient — a large clinical effect.
Heterogeneity was substantial (I² routinely above 70%, meaning trials disagreed on the magnitude of the benefit), but the direction of effect was robust across every analysis.
Wu and colleagues’ 2015 meta-analysis in JAMA Internal Medicine focused on CBT-I for insomnia comorbid with another psychiatric or medical condition (37 RCTs).5 Effects on insomnia outcomes were comparable to the primary-insomnia literature, with notable secondary benefits on the comorbid condition.
The convergence across reviews is what matters. Different inclusion criteria, different statistical adjustments, and different decades all yield effect sizes that are clinically meaningful and well-replicated.
CBT-I vs. Sleep Medications: Different Time Horizons, Different Winners
Hypnotics — typically zolpidem (Ambien), zopiclone (Imovane), eszopiclone (Lunesta), or older benzodiazepines — act faster. Within the first 1–2 weeks, their effect on sleep latency and wake time matches or exceeds CBT-I.6 By 4–8 weeks, the gap closes. By 3–6 months, CBT-I matches or beats medication on most outcomes — and the durability gap widens further afterward.
Mitchell and colleagues’ 2012 systematic review compared CBT-I head-to-head with pharmacotherapy across 5 RCTs.7 Short-term effects were comparable between treatments. Long-term effects favored CBT-I — benefits persisted after treatment ended, while medication benefits required continued use, and discontinuation often led to rebound insomnia.
The clinical picture this paints is straightforward. For acute, situational insomnia, hypnotics are the faster tool. For chronic insomnia, the durability data favor CBT-I. The European Insomnia Guideline’s stepped-care framework reflects this: short-term hypnotic use is acceptable, but chronic prescribing isn’t, and CBT-I is the recommended first-line intervention regardless of severity.2
Self-Help CBT-I: A 187-Page Book Beats the Standard Handout
A new RCT by Bjorvatn and colleagues in the Scandinavian Journal of Primary Health Care sharpens this picture.8
172 Norwegian primary-care patients who had been prescribed zopiclone or zolpidem in the prior 6 months were randomized to receive either a sheet of standard sleep hygiene advice or a 187-page CBT-I self-help book.
Mean age 64; 69% female; 83% met criteria for insomnia at baseline; 27% reported anxiety; 27% reported depression. These are everyday primary-care patients, not self-referred volunteers from a sleep clinic.
Outcomes at 4–5 month follow-up:
- Daily hypnotic use: dropped from 25.4% to 18.6% in the book group; increased from 21.2% to 22.7% in the sleep hygiene group. The standard between-group test was near-significant in the primary model (p = 0.077); a sensitivity analysis using a model better suited to small cell counts found an odds ratio (the multiplier on the odds of the outcome) of 6.69 (95% CI 2.30 to 19.47, p = 0.001) favoring the book.8
- Anxiety: the proportion meeting threshold on the PHQ-4 (a brief 4-item screen for anxiety and depression) dropped from 32.1% to 23.2% in the book group; increased from 27.0% to 31.7% in the sleep hygiene group. The corresponding odds ratio was 17.13 (95% CI 1.33 to 221.24, p = 0.030) — large point estimate but a wide confidence interval, so treat the magnitude with caution.8
- Insomnia severity (ISI — the standard self-report scale): improved significantly in both groups (Cohen’s d — a standardized effect-size where roughly 0.2 is small, 0.5 medium, 0.8 large — was 0.28 in the sleep hygiene group, 0.38 in the book group). The between-group interaction was not significant.
- Sleep duration: increased significantly only in the book group.
- Depression: unchanged in both groups.
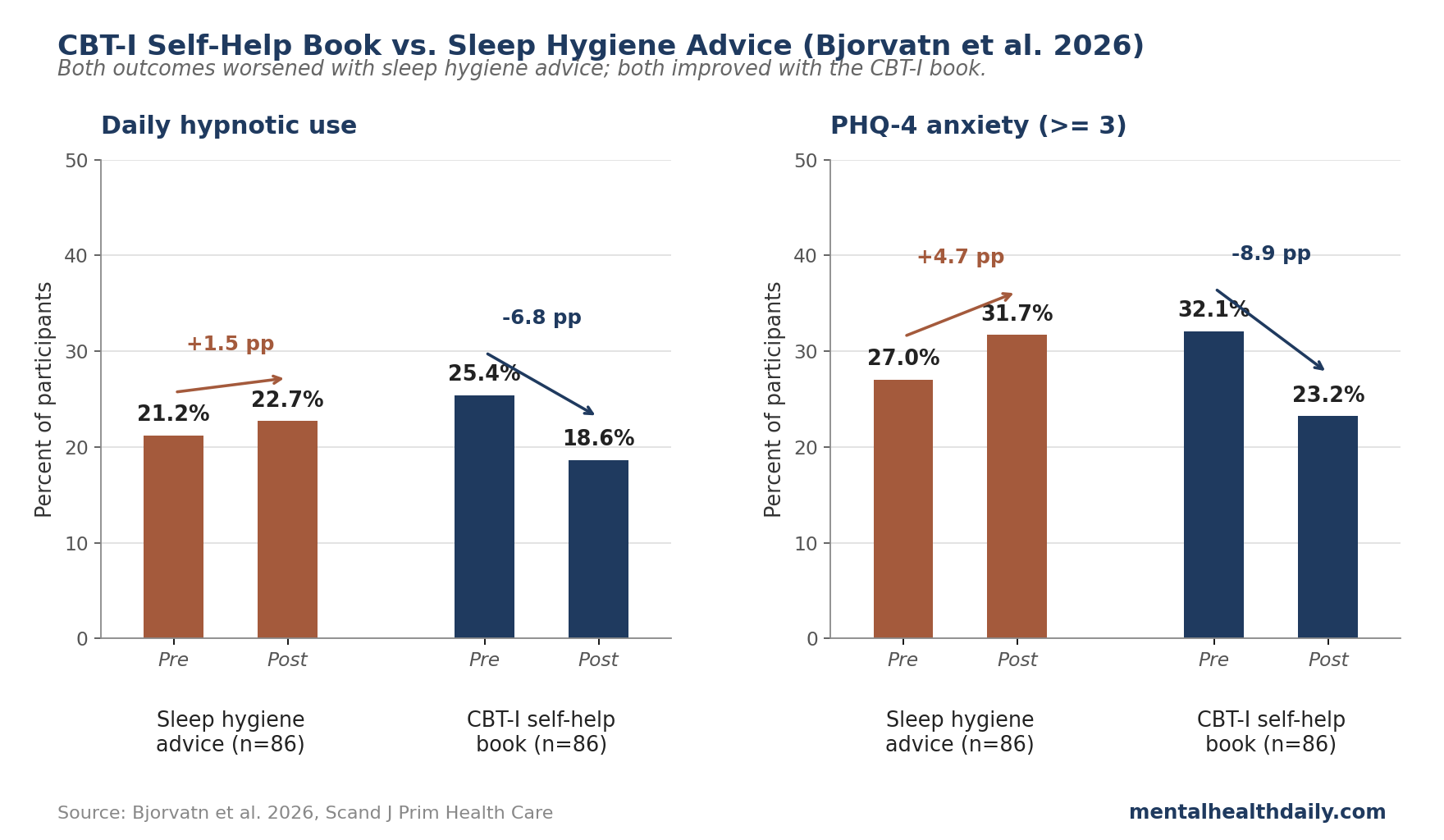
The contrast is the point. Sleep hygiene advice didn’t help — on hypnotic use and anxiety, the standard handout was associated with a small worsening. The book, which costs less than a single doctor’s visit and requires no clinician time, produced clinically meaningful improvements in a population that’s typically resistant to deprescribing.
The practical read: if the standard primary-care response to insomnia is a hypnotic prescription plus a sleep-hygiene handout, the handout is doing nothing — and the prescription is locking in a problem CBT-I would solve durably. A book, an app, or a brief structured digital program is a better baseline than either.
Why CBT-I Works: The Behavioral Mechanism
The mechanism isn’t mysterious. Chronic insomnia is sustained by two reinforcement loops that sleep restriction and stimulus control directly target:
- Time-in-bed extension lowers sleep pressure. A patient sleeping 5 hours but spending 8 hours in bed has a sleep efficiency of ~62%, far below the ~85% threshold associated with consolidated sleep. The extra 3 hours dilute sleep pressure across the night, fragment what sleep does occur, and worsen subjective sleep quality. Sleep restriction directly fixes this by re-establishing a tight bed-sleep ratio.1
- Conditioned arousal in the bedroom. Repeated nights of lying awake in bed condition the bedroom as a wake cue rather than a sleep cue. Stimulus control breaks this conditioning by removing the patient from the bedroom whenever sleep doesn’t come quickly — deconditioning the wake association and reconditioning the sleep association.
These mechanisms explain the headline pattern: large reductions in sleep latency and WASO, modest changes in total sleep time, and substantial improvements in subjective sleep quality. Patients aren’t sleeping much more — they’re sleeping much more efficiently.
Where the CBT-I Literature Has Gaps
Several limitations cut across the CBT-I literature.
Heterogeneity is high. Trauer 2015 and van Straten 2018 both report I² statistics above 70% on most outcomes — meaning trials disagree on the magnitude of the benefit beyond what sampling noise alone would produce.3,4
Pooled effect sizes average over genuine underlying differences in protocol intensity, patient selection, and outcome assessment. A “moderate” pooled effect can hide trials where the intervention worked dramatically and others where it barely moved the needle.
Self-help dropout is substantial. Online and app-delivered CBT-I show dropout rates that can exceed 50% in some programs.8
The Bjorvatn book trial had a 73% follow-up rate, but follow-up isn’t the same as completion; the book asks for sustained behavioral effort that not everyone delivers. Intent-to-treat estimates (which count everyone who started, not just finishers) are typically smaller than per-protocol estimates — the right direction if you want a number that maps to ordinary clinical use.
Severe psychiatric comorbidity is underrepresented. Most efficacy trials excluded patients with active psychosis, severe bipolar disorder, or current substance dependence.
The Bjorvatn trial included community patients with meaningful comorbidity but didn’t enrich for severe psychiatric illness. CBT-I in treatment-resistant schizophrenia, where insomnia rates approach 80%, is currently being formally tested in the COSTS trial (NCT06749444) — results aren’t in yet.9
Older adults and patients on concurrent medications are understudied for adverse effects. Sleep restriction transiently worsens daytime sleepiness during the protocol’s first 1–2 weeks, which raises fall risk in elderly patients and driving-safety concerns more broadly. Most trials don’t systematically track these.
How to Get CBT-I Without Going to a Sleep Specialist
CBT-I has essentially no medical downside for the vast majority of adults, and the upside on chronic insomnia is large and durable. The calibration above is about effect size variability across studies, not about whether to do it.
- Self-help options work. A book like the Bjorvatn study used, or an evidence-based digital program (Sleepio, SHUTi, CBT-I Coach app from the VA), produces meaningful improvements without therapist contact. Start here if access to a sleep specialist is limited.
- The active ingredients are sleep restriction and stimulus control — commit to those. Sleep hygiene tweaks alone won’t move chronic insomnia. The discomfort of compressed time-in-bed in week one is the protocol working, not failing.
- Track sleep efficiency, not sleep duration. Calculate hours asleep divided by hours in bed. Adjust bedtime to keep the ratio above 85% — that’s the lever, not lying in bed longer hoping to bank more sleep.
- Use hypnotics short-term if needed, but plan an off-ramp. Acute and situational insomnia is a reasonable hypnotic indication. Chronic daily hypnotic use isn’t — tolerance, dependence, and rebound insomnia on discontinuation are well-documented. CBT-I plus a deprescribing taper is the best-evidenced exit.
- Escalate to in-person CBT-I if self-help doesn’t help in 4–6 weeks. European guidelines recommend a stepped-care approach: self-help first, individualized therapy if needed.2 Most patients respond to the lowest tier; a minority need clinician-delivered protocols.
Quick Answers on CBT-I
How long does CBT-I take to work?
Sleep latency and wake time typically improve within 2–4 weeks of starting sleep restriction. The protocol itself usually runs 4–8 weeks. Sleep medications produce faster relief in the first 1–2 weeks; CBT-I catches up by ~4 weeks and matches or exceeds medication thereafter.7
Is CBT-I as effective as sleep medication?
Short-term, no — medication acts faster. Long-term, yes, and for durability, CBT-I wins. Mitchell 2012’s head-to-head review found comparable short-term effects and persistent CBT-I benefits after treatment ended, while medication benefits required continued use.7 European guidelines recommend CBT-I as first-line for chronic insomnia regardless.2
Does an app or book work as well as a therapist?
Self-help CBT-I produces smaller effect sizes than therapist-delivered CBT-I, but the difference is modest, and access trade-offs typically favor the self-help version. van Straten 2018’s pooled analysis across delivery formats found large effects across the board.4 The Bjorvatn 2026 RCT showed a CBT-I book outperformed sleep hygiene in actual primary-care patients on hypnotics.8
Is sleep hygiene the same thing as CBT-I?
No, and the conflation is doing substantial damage. Sleep hygiene is the small handout of basic advice (caffeine, alcohol, room temperature, etc.). CBT-I is a structured protocol whose active ingredients are sleep restriction and stimulus control. The European Insomnia Guideline explicitly does not recommend sleep hygiene as a stand-alone treatment.2
Can I do CBT-I if I’m taking sleep medication?
Yes — in fact, CBT-I plus a planned deprescribing taper is one of the best-evidenced exits from chronic hypnotic use. The Bjorvatn 2026 trial enrolled patients prescribed zopiclone or zolpidem in the prior 6 months and showed the self-help book reduced daily hypnotic use during the intervention.8 Don’t stop hypnotics abruptly — talk to your prescriber about a taper schedule alongside CBT-I.
Does CBT-I help if my insomnia is caused by depression or anxiety?
Yes. Wu et al. 2015’s meta-analysis of CBT-I for comorbid insomnia found effects on insomnia outcomes comparable to those in primary insomnia, with secondary benefits on the comorbid condition.5 Hertenstein and colleagues’ 2022 review confirms CBT-I improves comorbid depression and anxiety symptoms, not just sleep.10 The Bjorvatn 2026 RCT also found anxiety improvement specifically in the book group.8
References
- Cognitive behavioral therapy for chronic insomnia: a systematic review and meta-analysis. Mitchell MD et al. BMC Family Practice. 2012;13:40. doi:10.1186/1471-2296-13-40
- The European Insomnia Guideline: an update on the diagnosis and treatment of insomnia 2023. Riemann D et al. Journal of Sleep Research. 2023;32(6):e14035. doi:10.1111/jsr.14035
- Cognitive Behavioral Therapy for Chronic Insomnia: A Systematic Review and Meta-analysis. Trauer JM et al. Annals of Internal Medicine. 2015;163(3):191-204. doi:10.7326/M14-2841
- Cognitive and behavioral therapies in the treatment of insomnia: A meta-analysis. van Straten A et al. Sleep Medicine Reviews. 2018;38:3-16. doi:10.1016/j.smrv.2017.02.001
- Cognitive Behavioral Therapy for Insomnia Comorbid With Psychiatric and Medical Conditions: A Meta-analysis. Wu JQ et al. JAMA Internal Medicine. 2015;175(9):1461-1472. doi:10.1001/jamainternmed.2015.3006
- Direct Comparison of Sleep Restriction Therapy with Sleep Medication: A Randomized Controlled Trial. Jacobs GD et al. Archives of Internal Medicine. 2004;164(17):1888-1896. doi:10.1001/archinte.164.17.1888
- Comparative effectiveness of cognitive behavioral therapy for insomnia: a systematic review. Mitchell MD et al. BMC Family Practice. 2012;13:40 (head-to-head comparisons section). doi:10.1186/1471-2296-13-40
- A randomized controlled trial comparing sleep hygiene advice with a self-help book focusing on cognitive behavioral therapy for insomnia: a study among patients with prescribed hypnotics from the GP. Bjorvatn B et al. Scandinavian Journal of Primary Health Care. 2026;44(1):1-10. doi:10.1080/02813432.2025.2525423
- Cognitive behavioral therapy for insomnia vs. standard cognitive behavioral therapy for sleep and circadian disturbances in treatment-resistant schizophrenia: study protocol for the randomized controlled trial (COSTS). Johansen JF et al. Trials. 2026;27:309. doi:10.1186/s13063-026-09482-0
- Cognitive behavioral therapy for insomnia in patients with mental disorders and comorbid insomnia: A systematic review and meta-analysis. Hertenstein E et al. Sleep Medicine Reviews. 2022;62:101597. doi:10.1016/j.smrv.2022.101597
