Ketamine and its S-enantiomer esketamine (sold as Spravato) are now established treatments for treatment-resistant depression and acute suicidality. The standard safety pitch from sponsors and clinics: at single sub-anesthetic doses, in supervised settings, addiction risk is minimal.
A new case report in BJPsych Open documents a patient for whom that pitch failed catastrophically — and it raises questions the trial data are not yet powered to answer.
Research Highlights
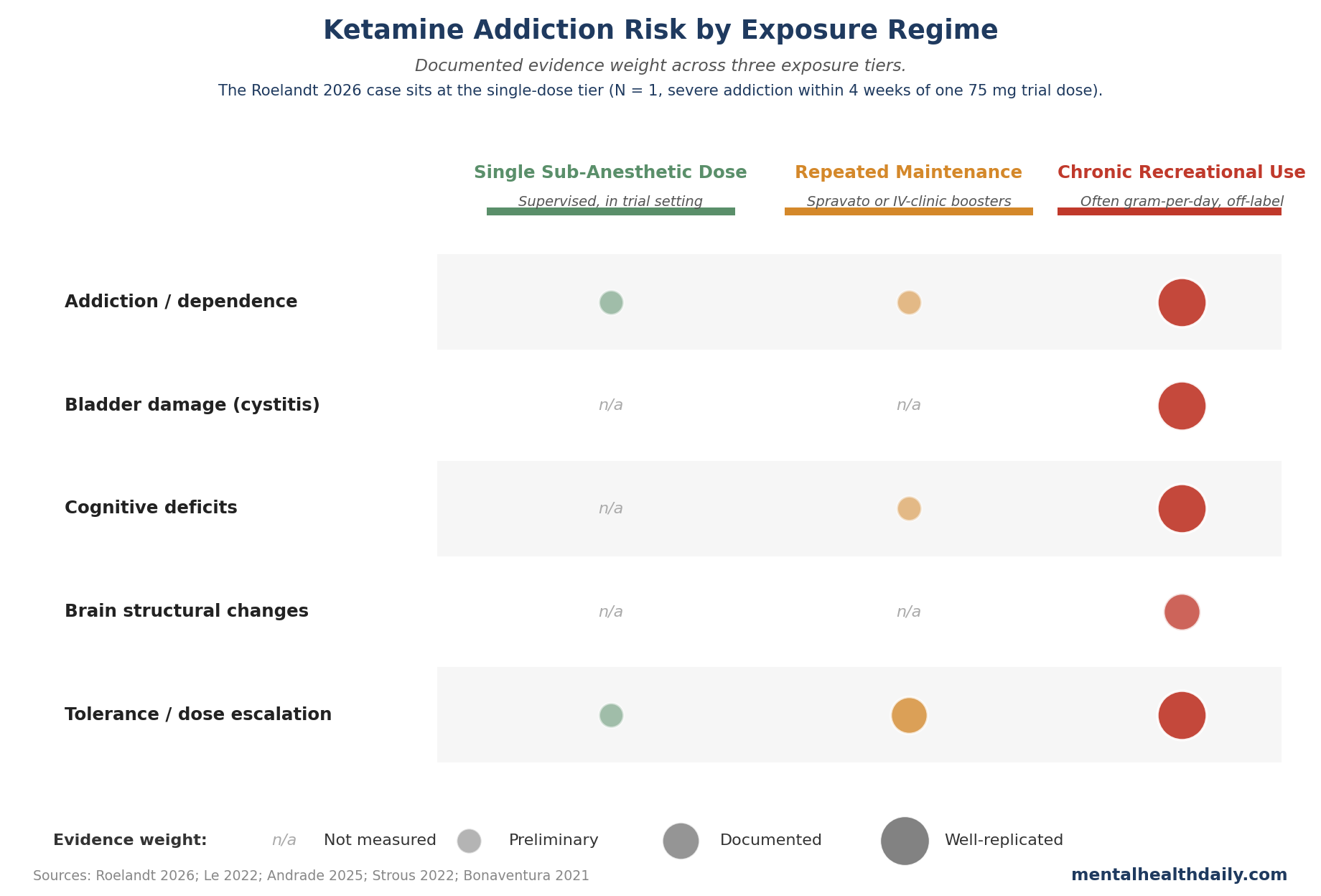
- One published case is not a base rate. Roelandt and colleagues (2026) describe a 25-year-old multimorbid patient who developed severe ketamine addiction after a single 75 mg intranasal sub-anesthetic dose given for acute suicidality. It is N = 1, but it is now the second published case after Bonnet 2015 of post-clinical-use ketamine addiction in a depressed patient with no prior addiction history.
- Single-dose addiction risk in supervised psychiatric trials is low but non-zero. A 2022 scoping review pooled 65 preclinical and clinical studies and found no systematic evidence of misuse or dependence in supervised treatment-resistant depression cohorts — with the caveat that most of those trials never measured abuse liability formally.
- S-ketamine appears more addictive than R-ketamine. Preclinical work in Bonaventura 2021 shows S-ketamine self-administers more reliably and at lower thresholds than R-ketamine. Spravato (esketamine, the S-enantiomer) is the FDA-approved option, so this matters for the clinic.
- The chronic-maintenance and clinic-pipeline blind spot is the bigger unknown. Phase 3 ketamine and esketamine trials run weeks to a few months; the explosion of off-label IV-ketamine clinics and ongoing Spravato maintenance has not been studied at the years-of-exposure horizon where dependence shows up most clearly. The case report flags this; the literature can’t yet answer it.
- Recreational ketamine harms are well-replicated and serious. 44–77% of chronic ketamine users develop lower urinary tract symptoms (ketamine cystitis); 8–30% develop upper urinary tract disease. These doses are far above clinical regimens, but they bound the worst case if therapeutic exposure escalates to recreational patterns — which is exactly what happened in the Roelandt case.
What the Roelandt Case Report Documents: One Patient, 75 mg, Then 1 g Daily
Roelandt and colleagues describe a 25-year-old woman enrolled in the KETA pilot study at University Medical Center Groningen.1 She had recurrent depression, obsessive–compulsive disorder, autism spectrum disorder, anorexia nervosa, and more than 10 prior suicide attempts. Her substance-use history was sporadic alcohol and cannabis only — no history of any addictive disorder.
The intervention: one 75 mg open-label dose of intranasal racemic ketamine. Symptom scores tracked the familiar pattern.
- MADRS (depression): 37 at baseline → 31 at one hour → 32 at one day → 36 at one week.1
- BSSI (suicidal ideation): 35 at baseline → 29 at one hour → 25 at one day → 31 at one week.1
(MADRS is the Montgomery–Åsberg Depression Rating Scale, where higher means worse; BSSI is the Beck Scale for Suicidal Ideation. Both showed a rapid drop and near-full rebound by day 7.)
The post-trial trajectory is what the case report exists to document. The patient said the ketamine experience pushed her chronic intrusive obsessional thoughts “to the side of her head.” About 4 weeks after the study dose, she contacted a drug dealer for the first time in her life and began intranasal ketamine self-administration. Within weeks she was using up to 1 g daily — roughly 13 times the trial dose, every day — cycling cocaine and 3-MMC (a synthetic cathinone stimulant) to counter ketamine’s sedating effects.1
She reported no urinary or cognitive adverse effects, but escalating tolerance, financial collapse, threatened eviction from supported housing, and a suicide attempt when supply was cut off followed. She quit, then relapsed into both ketamine and cocaine addiction.1
The authors are appropriately cautious. What this case cracks is the easy claim that controlled-setting ketamine carries no addiction risk in patients without prior substance abuse.
Bonnet 2015 and 65 Pooled Studies: What the Prior Literature Shows
The first published account of ketamine addiction after intended clinical use is Bonnet 2015: a 50-year-old anesthetic nurse with major depression who began self-injecting 50 mg of racemic ketamine intramuscularly, then escalated over years to a daily addiction.2 Bonnet’s ketamine was diverted from hospital supply rather than administered in a trial, but the pattern was the same: rapid relief, craving, and dose escalation in a depressed patient with no prior addiction history. Roelandt 2026 is the next published instance — this time in a patient who received a fully supervised single trial dose. Two cases 11 years apart anchor a narrow but documented signal: supervised clinical use does not fully neutralize the molecule’s abuse liability.
Le and colleagues (2022) ran the most comprehensive scoping review of ketamine abuse liability to date, pooling 65 preclinical and clinical studies.3 Their summary: in supervised treatment-resistant-depression cohorts, single or repeated ketamine administrations did not produce systematic evidence of misuse, dependence, diversion, or gateway activity.
The caveat: most included trials never measured abuse liability with validated instruments. They tracked vital signs, mood, and dissociation, not craving, drug liking, or off-protocol acquisition. That probably underreports outcomes of the kind Roelandt documents.3
Chubbs and colleagues (2022) tried to fix this gap, surveying 62 outpatients on sublingual or intranasal ketamine in real-world clinics with structured drug-liking and craving scales.4 Patients reported meaningful drug liking; the authors stopped short of dependence. It’s the only real-world dataset with structured abuse-liability measurement, and it isn’t large enough to settle the question.
Population-level addiction rates after supervised clinical ketamine exposure are not pinned down. The best-available estimate is “low” — but the evidence base systematically under-measures the outcome it would need to be confident.
S-Ketamine Self-Administers More Reliably Than R-Ketamine
The Roelandt patient received racemic ketamine — the 50/50 mixture of the R- and S-enantiomers used in most older psychiatric trials and still common in IV-ketamine clinics. Spravato is the S-enantiomer alone. The two are not interchangeable on abuse liability.
Bonaventura and colleagues’ (2021) preclinical work directly compared the enantiomers in self-administration paradigms: S-ketamine self-administered at lower thresholds and supported dose escalation, while R-ketamine did not.6 S-ketamine has higher NMDA-receptor affinity and stronger psychotomimetic effects — both track abuse liability across dissociative drug classes.
The animal data don’t show that Spravato is more addictive in patients than racemic IV ketamine; they show that the higher-abuse-liability enantiomer became the regulatory pathway. Long-horizon Spravato data on craving, drug liking, and off-protocol use will fill in the rest.
Where Chronic Maintenance Risk Stands Uncharacterized
Spravato maintenance is twice-weekly for the first month, then weekly to every-other-week indefinitely. IV-ketamine clinics typically run 6 initial infusions over 2 to 3 weeks plus boosters every 2 to 4 weeks. Ongoing exposure is the norm because the antidepressant effect is short-lived.
Phase 3 esketamine trials ran 4 to 16 weeks; even open-label extensions cap out around a year. Cohorts on chronic maintenance ketamine for 3, 5, or 10 years have not been studied prospectively for abuse outcomes — they barely existed when the regulatory packages closed.
Strous and colleagues (2022) reviewed brain changes in long-term ketamine users (predominantly recreational) and found consistent white-matter and cortical changes plus cognitive decrements at heavy exposure.7 Whether anything similar happens at clinical-exposure levels is unknown. The dose gap between recreational use (grams per day) and clinical maintenance (tens of milligrams per session) is large; the in-between zone has not been measured at scale.
The clinical read: single-dose risk is roughly characterizable. Multi-year maintenance risk in patients receiving clinic-controlled doses is not. The Phase 3 evidence base is what got Spravato approved; it is not what would be needed to fully characterize maintenance-era safety.
Recreational Ketamine Bladder and Cognitive Harms at the Worst Case
Roelandt’s patient escalated to recreational dose ranges within weeks. The literature on recreational ketamine is clearer than the clinical literature and bounds the worst case.
Andrade’s 2025 review of ketamine-associated uropathy reports lower urinary tract symptoms (urgency, frequency, painful voiding, hematuria) in 44–77% of regular ketamine users and upper urinary tract disease in 8–30%.8 Severe ketamine cystitis can require bladder reconstruction. These prevalence figures come from heavy users at daily doses in the gram range — the regime Roelandt’s patient reached within weeks.
UK epidemiology shows scale. Ketamine recreational prevalence was around 1.7% in 2008/2009 with lifetime use of 4%, and recreational use roughly doubled between 2016 and 2025.1 A Global Drug Survey self-treatment study estimated yearly use as high as 33 g for males and 29 g for females — orders of magnitude above any therapeutic regimen.9
Bladder, cognitive, and dependence harms at this exposure level are well-replicated. Whether supervised clinical exposure escalates into this zone is what 2 case reports cannot answer.
Limitations of the Roelandt Case Report
N = 1. A case report establishes that a phenomenon can occur; it does not establish how often. “How rare is post-clinical-use ketamine addiction?” is not answerable from this paper or from Bonnet 2015.
This patient’s profile is unusually high-risk. Four concurrent psychiatric diagnoses, more than 10 prior suicide attempts, intrusive thoughts ketamine specifically suppressed, fragile housing, and emerging features of borderline personality disorder — a known risk factor for impulsive substance use. Generalizing from this profile to a stable patient with single-diagnosis treatment-resistant depression is a long jump.
Causal attribution is hard. Whether the trial dose caused the addictive trajectory or introduced her to a substance she would have encountered anyway is unanswerable from one observation.
Selection toward dramatic cases. Case reports skew toward what gets written up; patients without addiction emergence — numerically the vast majority — rarely do. Two published cases inform what can happen, not the per-exposure base rate.
Practical Monitoring Adjustments for Ketamine and Spravato
This case doesn’t change whether ketamine should remain available for treatment-resistant depression and acute suicidality — the antisuicidal effect signal is strong enough to justify the option for patients who have exhausted standard treatments.5 It does change what reasonable monitoring should look like. Four adjustments are defensible from the available evidence.
- Screen current vulnerability, not just past history. The Roelandt patient had no prior addiction history; current vulnerability indicators (active intrusive thoughts ketamine could suppress, isolation, financial precarity, comorbid impulse-related disorders) might have surfaced more risk than the past-history checkbox.
- Follow up over weeks, not days. Roelandt’s team extended the KETA RCT follow-up from 7 days to 4 weeks specifically because of this case. The transition from supervised dose to recreational acquisition took roughly 4 weeks here. A 7-day follow-up would have missed it entirely.
- Use structured craving and drug-liking instruments. The Ketamine Side Effect Tool (KSET) and similar scales exist; they are not routine in trials or in clinics. Direct measurement of post-dose craving catches what passive vital-sign monitoring does not.
- Loop in the primary psychiatrist actively. Ketamine clinics often run parallel to a patient’s primary mental-health team. The treating clinician needs to know what was administered, when, and what to watch for — particularly if the patient is at high baseline risk.
For patients considering ketamine for depression or suicidality: the molecule has well-documented abuse liability outside controlled settings, and a small documented signal of post-clinical-use abuse inside them. The benefit-risk math still favors treatment for many patients with treatment-resistant depression or acute suicidality, especially after other options have failed. It doesn’t support the casual “ketamine is safer than alcohol” framing some clinics market with.
Common Questions About Ketamine and Addiction Risk
Is ketamine addictive at therapeutic doses?
Low but non-zero risk. Pooled reviews of supervised single sub-anesthetic doses in treatment-resistant depression cohorts found no systematic evidence of dependence.3 Two published case reports (Bonnet 2015, Roelandt 2026) document severe addiction after intended clinical exposure in patients with no prior substance-use disorder.1,2 Population-level rates remain unestimated because most trials never measured abuse liability with validated instruments.
Is Spravato (esketamine) more addictive than racemic ketamine?
Preclinical evidence suggests modestly more. Bonaventura and colleagues showed S-ketamine self-administered more reliably in animals than R-ketamine.6 Clinical head-to-head abuse-liability data in humans are limited. Spravato carries an FDA Risk Evaluation and Mitigation Strategy (REMS) requirement for in-clinic supervised dosing, which is the regulatory acknowledgment of this concern.
What does ketamine cystitis look like?
Lower urinary tract symptoms (urgency, frequency, painful voiding, hematuria) develop in 44–77% of heavy regular ketamine users; 8–30% develop upper urinary tract disease including hydronephrosis.8 Severe cases require bladder reconstruction. These rates are from recreational users at gram-per-day exposure levels, far above any clinical regimen.
Are IV-ketamine clinics safe long-term?
No good answer yet. Phase 3 trials and even open-label extensions cap at roughly one year. The cohorts on multi-year maintenance ketamine are too new and too small to have produced prospective abuse-outcome data. The safety profile at the years-of-exposure horizon is not characterized, and patients should weigh that uncertainty alongside the genuine benefits.
Should a patient with a past substance-use disorder avoid ketamine for depression?
Most current ketamine and esketamine protocols list active substance use as a contraindication and past substance-use disorder as a relative risk factor. The Roelandt case complicates the inverse claim — that no prior history equals safety — but does not overturn the framework. Decisions belong with the treating psychiatrist, weighing symptom severity, alternatives exhausted, and current vulnerability indicators alongside past history.
References
- Ketamine addiction following a single sub-anaesthetic ketamine treatment for acute suicidality in a psychiatrically multimorbid patient: case report. Roelandt GHJ, Strous JFM, Kamphuis J, Schoevers RA, Marijnissen RM. BJPsych Open. 2026;12:e110. doi:10.1192/bjo.2026.11012
- Long-Term Ketamine Self-Injections in Major Depressive Disorder: Focus on Tolerance in Ketamine’s Antidepressant Response and the Development of Ketamine Addiction. Bonnet U. Journal of Psychoactive Drugs. 2015;47(4):276-285. doi:10.1080/02791072.2015.1072653
- The abuse liability of ketamine: A scoping review of preclinical and clinical studies. Le TT, Cordero IP, Jawad MY et al. Journal of Psychiatric Research. 2022;151:476-496. doi:10.1016/j.jpsychires.2022.04.035
- A survey of drug liking and cravings in patients using sublingual or intranasal ketamine for treatment resistant depression: a preliminary evaluation of real world addictive potential. Chubbs B, Wang J, Archer S et al. Frontiers in Psychiatry. 2022;13:1016439. doi:10.3389/fpsyt.2022.1016439
- The effect of a single dose of intravenous ketamine on suicidal ideation: a systematic review and individual participant data meta-analysis. Wilkinson ST, Ballard ED, Bloch MH et al. American Journal of Psychiatry. 2018;175(2):150-158. doi:10.1176/appi.ajp.2017.17040472
- Pharmacological and behavioral divergence of ketamine enantiomers: implications for abuse liability. Bonaventura J, Lam S, Carlton M et al. Molecular Psychiatry. 2021;26(11):6704-6722. doi:10.1038/s41380-021-01093-2
- Brain Changes Associated With Long-Term Ketamine Abuse, A Systematic Review. Strous JFM, Weeland CJ, van der Draai FA et al. Frontiers in Neuroanatomy. 2022;16:795231. doi:10.3389/fnana.2022.795231
- Ketamine-Associated Uropathy During Therapeutic and Nontherapeutic Use: Prevalence, Clinical Features, Mechanisms, and Strategies for Risk Reduction. Andrade C. Journal of Clinical Psychiatry. 2025;86:25f16083. doi:10.4088/JCP.25f16083
- Self-treatment of psychiatric conditions using ketamine: patterns, characteristics, and retrospective insights. Smith G, Piatkowski T, Ferris J et al. Journal of Psychopharmacology. 2025. doi:10.1177/02698811251378509
