A 2026 UK Biobank MRI preprint involving 71,214 adults found that chronic pain plus current depression had a structural brain profile that was more than chronic pain plus depression pasted together. The 1,377-person comorbid group showed widespread lower cortical volume, lower bilateral thalamic and hippocampal volume, lower left accumbens volume, and broad white-matter microstructure differences compared with people who had neither condition.1
Research Highlights
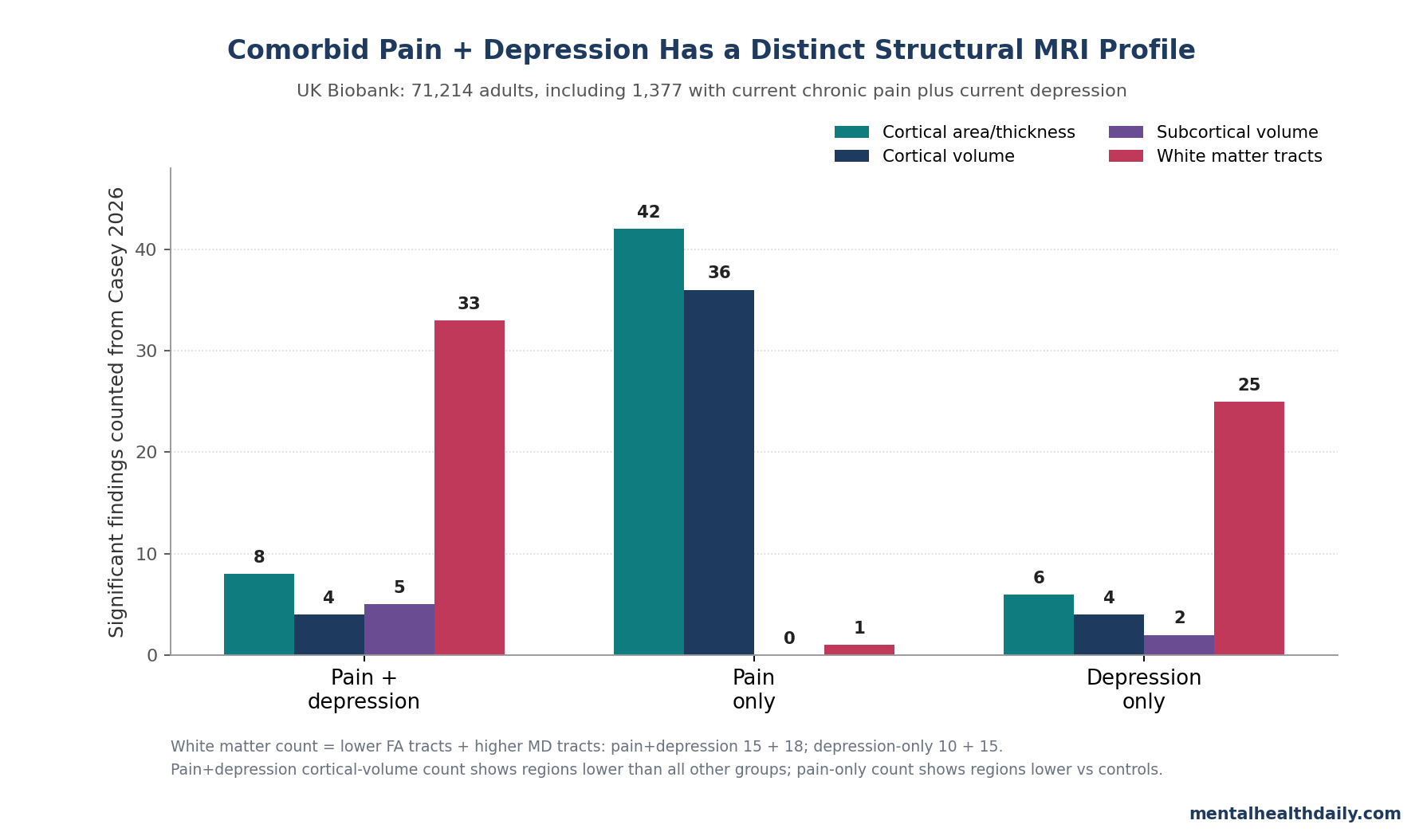
- 71,214 adults were analyzed: UK Biobank structural MRI data were split into controls (43,926), chronic pain only (24,981), current depression only (930), and comorbid chronic pain plus depression (1,377).1
- The comorbid group had a distinct cortical-volume pattern: left pars triangularis, left medial orbitofrontal cortex, right superior frontal cortex, and right posterior cingulate cortex volume were lower than in all 3 other groups, with β ranges down to −0.118.1
- Subcortical findings were not pain-only findings: chronic pain alone showed no significant subcortical volume differences vs. controls, while the comorbid group had lower thalamus, hippocampus, and left accumbens volumes (β = −0.048 to −0.088).1
- White matter separated depression-linked signal from pain-only signal: the comorbid group had lower fractional anisotropy in 15/27 tracts and higher mean diffusivity in 18/27 tracts; pain-only had only 1 significant tract finding.1
- The result is anatomical, not causal: the study was cross-sectional, PHQ-2 based, self-report pain based, and a medRxiv preprint; it can show group-level MRI differences, not whether pain or depression caused them.1
Chronic pain and depression travel together often enough that treating them as separate research boxes can mislead. A 2025 meta-analysis estimated that 39.3% of adults with chronic pain had clinically significant depressive symptoms, and older clinical reviews linked the combination to higher pain intensity, poorer function, and worse treatment response than either condition alone.2,3
The Casey et al. analysis asked the neuroimaging version of that clinical problem. If chronic pain studies include depressed pain patients, and depression studies include people with persistent pain, then a supposed “pain brain” or “depression brain” may partly be a comorbidity signal.
The UK Biobank sample was large enough to separate the 4 groups directly.
71,214 UK Biobank MRI Scans, Split Into 4 Current-Symptom Groups
The researchers used the first UK Biobank imaging assessment and kept participants with complete structural MRI, covariates, and same-day pain/depression phenotype data. Current depression was defined by a Patient Health Questionnaire-2 (PHQ-2) score of 3 or higher; the PHQ-2 is a 2-item screen for depressed mood and loss of interest, not a full diagnostic interview.1,4
Chronic pain meant pain in at least 1 body site, or widespread pain, that interfered with usual activity and had lasted more than 3 months. Combining the pain and depression definitions created 4 groups:
- Controls: 43,926 people with neither current chronic pain nor current depression.
- Comorbid pain plus depression: 1,377 people, 1.9% of the MRI sample.
- Chronic pain only: 24,981 people, 35.1% of the MRI sample.
- Depression only: 930 people, 1.3% of the MRI sample.
The MRI outcomes covered cortical thickness, cortical surface area, cortical volume, subcortical regional volumes, and diffusion-tensor white-matter measures. Models adjusted for age, sex, body mass index, ethnic background, deprivation, smoking, alcohol use, imaging center, scanner head position, and intracranial volume where relevant.
The analysis was not a raw group comparison between sicker and healthier people; it was an adjusted group comparison, though residual confounding remains likely.
Comorbid Pain Plus Depression Had Lower Volume in 4 Cortical Regions Than All Other Groups
the most direct comorbidity-specific result was not “every pain and mood region shrank.” It was narrower and more useful: compared with all 3 other groups, the comorbid group had lower cortical volume in 4 regions — left pars triangularis, left medial orbitofrontal cortex, right superior frontal cortex, and right posterior cingulate cortex.1
Pars triangularis is part of the inferior frontal gyrus and participates in language, cognitive control, and reappraisal. Medial orbitofrontal cortex helps assign value to rewards, punishments, and pain relief.
Superior frontal cortex contributes to executive control and top-down regulation. Posterior cingulate cortex sits inside the default-mode network and has been linked to self-referential pain processing and catastrophizing.5
The effect ranges were small in standardized terms, but consistent after false-discovery-rate correction: left pars triangularis volume was lower across pairwise contrasts with β ranges from −0.106 to −0.063, left medial orbitofrontal cortex from −0.086 to −0.054, right superior frontal cortex from −0.086 to −0.058, and right posterior cingulate from −0.118 to −0.059.1
That pattern matters because those regions plausibly sit at the pain-mood interface: pain valuation, reward-related pain inhibition, negative-affect regulation, and internal attention to symptoms. The data do not prove those circuits caused the comorbid state.
They do show that the comorbid group carried a measurable cortical-volume signature that was not present in the same way in either single-condition group.
Thalamus, Hippocampus, and Left Accumbens Were Lower Only in the Comorbid Profile
Subcortical volume sharpened the separation between chronic pain alone and the comorbid state. Compared with controls, the chronic pain-only group had no significant subcortical volume differences.
The comorbid group had lower bilateral thalamic volume, lower bilateral hippocampal volume, and lower left nucleus accumbens volume.1
The thalamus is a relay hub for sensory input, including pain-related signals, and also participates in cortico-striatal and cortico-limbic loops. In the comorbid group, thalamic volume was lower on the left (β = −0.048, pFDR = 0.038) and right (β = −0.060, pFDR = 0.007).
The depression-only group also had lower bilateral thalamic volume, with slightly larger standardized coefficients (β = −0.067 left; β = −0.066 right).1
The hippocampal and accumbens findings were more specific to comorbidity. The comorbid group had lower left hippocampal volume (β = −0.062, pFDR = 0.035), lower right hippocampal volume (β = −0.088, pFDR = 0.002), and lower left accumbens volume (β = −0.073, pFDR = 0.011).
Hippocampus links stress, memory, and mood regulation; nucleus accumbens links reward, motivation, and avoidance learning. A pain-depression syndrome with lower hippocampal and accumbens volume is therefore biologically plausible, but still only an association.
White-Matter Findings Looked More Like Depression Than Pain Alone
The diffusion-tensor part of the study may be the most underappreciated piece. Fractional anisotropy (FA) measures how directionally coherent water diffusion is inside white-matter tracts; lower FA is often interpreted as weaker microstructural integrity.
Mean diffusivity (MD) measures overall diffusion magnitude; higher MD can indicate less restricted water movement and is often read as another sign of altered microstructure.1
Comorbid pain plus depression showed lower FA in 15 of 27 white-matter tracts and higher MD in 18 of 27 tracts compared with controls. The largest FA effects were in the inferior longitudinal fasciculus, a temporal-occipital tract involved in visual-emotional processing (left β = −0.104; right β = −0.116).
The largest MD effects were in the uncinate fasciculus (left β = 0.122; right β = 0.137), a tract linking orbitofrontal regions with the amygdala and anterior temporal lobe.1
Depression-only participants showed a similar broad white-matter pattern: lower FA in 10 of 27 tracts and higher MD in 15 of 27. Chronic pain-only participants did not.
Their only significant diffusion finding was higher FA in the right acoustic radiation (β = 0.022, pFDR = 0.025). The white-matter profile therefore does not support a simple story in which chronic pain alone drives the tract-level signal.
It looks closer to depression and comorbid pain-depression than to pain-only.
Pain-Only Was Broadly Cortical; Depression-Only Was More Region-Specific
The single-condition groups still mattered because they show what the comorbid pattern is being compared against. Chronic pain-only participants had widespread lower cortical surface area in 42 of 66 hemisphere-specific regional measures and lower cortical volume in 36 of 66 regions.
The largest surface-area effects were in the postcentral gyrus, which contains primary somatosensory cortex (left β = −0.043; right β = −0.042).1
That pain-only pattern fits adjacent UK Biobank chronic-pain work. Bhatt et al. found that pain widespreadness and pain type shaped brain-structure variability, supporting the idea that “chronic pain” is not 1 neuroanatomical category.6
Casey et al. add that once current depression is separated out, chronic pain alone still has a broad cortical footprint, but not the same subcortical and white-matter profile as the comorbid group.
Depression-only participants showed no significant cortical surface-area differences vs. controls, but they had lower cortical thickness in the right entorhinal cortex and several left-hemisphere regions, plus lower volume in 4 cortical regions.
The depression-only pattern partly lined up with earlier UK Biobank depression imaging reports by Harris et al. and Shen et al., especially on white matter, while diverging in some cortical and subcortical details because Casey et al. used a current-symptom PHQ-2 phenotype rather than lifetime depression definitions.7,8
Why Separating Comorbidity Changes the Interpretation
Many older neuroimaging studies tried to find a “pain signature” or “depression signature” by comparing cases with controls. That design becomes unstable when the conditions commonly co-occur.
If a chronic-pain sample includes many depressed participants, and a depression sample includes many people with persistent pain, overlapping brain findings can be mistaken for shared neurobiology when they may partly reflect an unmodeled comorbid subgroup.
Yu et al. recently reported shared cortical thinning clusters across major depression, anxiety, and chronic pain, including right insula, left anterior cingulate cortex, left inferior frontal gyrus, and left middle temporal gyrus.9 That finding may be real shared biology.
Casey et al. do not erase it. They add a necessary caution: shared-looking anatomy can also come from comorbidity mixing inside case-control samples.
The direct comorbidity literature is smaller and messier. Gustin et al. and Ma et al. reported lower volumes in regions such as thalamus, posterior cingulate cortex, and prefrontal areas among chronic-pain patients with depressive symptoms, which lines up with parts of the Casey profile.10,11
But Casey et al. found lower hippocampal volume where some smaller studies reported increases. Pain type, pain location, medication exposure, symptom severity, and sample size can all move these estimates.
The Result Supports Integrated Care Logic, Not MRI-Based Diagnosis
The practical read is not that brain scans should diagnose comorbid chronic pain and depression. The group differences are too small, overlapping, and confounded for that.
The stronger implication is conceptual: comorbid pain-depression deserves to be treated as a real clinical category, not a nuisance variable.
Roughan et al. linked comorbid chronic pain and depression to shared risk factors and differential antidepressant effectiveness, and Bair et al. summarized the clinical burden: worse symptoms, poorer function, and more difficult treatment courses.3,12 Casey et al. supply the neuroanatomical companion to that clinical observation.
The comorbid group differs in regions tied to pain modulation, self-referential distress, reward, memory-stress circuitry, and fronto-limbic connectivity.
For treatment thinking, that supports integrated pain-depression care: antidepressants with analgesic evidence when appropriate, psychological treatments adapted for pain and mood together, graded activity or rehabilitation that accounts for depressive inhibition, sleep treatment, and care models that do not make patients bounce between pain clinics and psychiatry as if the problems are unrelated. The MRI data do not tell clinicians which treatment to pick.
They do make the “treat one and hope the other follows” model look biologically thin.
Limitations of This UK Biobank MRI Analysis
The biggest limitation is temporal ordering. A cross-sectional scan cannot show whether brain differences preceded chronic pain and depression, resulted from persistent symptoms, reflected medication or medical comorbidity, or came from shared upstream vulnerability.
The researchers explicitly call for longitudinal imaging to test whether structural differences predict later comorbidity or track symptom persistence.1
Severity is not cleanly separable from comorbidity. People with both chronic pain and depression often have higher total symptom burden than people with either condition alone. Some “comorbidity-specific” MRI signal may therefore be severity signal.
The phenotypes were broad. PHQ-2 screening is useful at population scale, but it is not a structured psychiatric diagnosis. Chronic pain combined local pain and widespread pain, even though pain location and distribution can have different neurobiology.
UK Biobank is not the general population. Imaging participants are healthier, more affluent, and less severely ill on average than the source population. That healthy-volunteer bias can attenuate effect sizes and limit generalizability.
The paper was a preprint. The PDF used here was posted to medRxiv and had not been certified by peer review. The sample size and methods are serious, but the result should still be read as pre-publication evidence.
Questions About Chronic Pain, Depression, and Brain Structure
What was the main finding in the Casey 2026 UK Biobank analysis?
The main finding was a distinct MRI profile in 1,377 people with both current chronic pain and current depression. Compared with controls, they had lower cortical volume across multiple regions, lower bilateral thalamic and hippocampal volume, lower left accumbens volume, and broad white-matter microstructure differences.
Compared with all 3 other groups, they also had lower volume in 4 specific cortical regions.
Did chronic pain alone show the same brain pattern?
No. Chronic pain-only participants had widespread lower cortical surface area and cortical volume, including 42 of 66 surface-area regions and 36 of 66 cortical-volume regions, but they did not show significant subcortical volume differences vs. controls.
They also had only 1 significant diffusion finding, and it was higher FA in the right acoustic radiation, not the broad lower-FA/higher-MD profile seen in comorbidity.
Does this prove chronic pain and depression damage the brain?
No. The study shows adjusted group-level structural MRI differences.
It does not prove irreversible damage, causation, or diagnostic separability. Brain structure can reflect many things: symptom burden, medication exposure, inflammation, sleep, activity level, vascular risk, early-life risk, or pre-existing vulnerability.
Why does the hippocampus matter in this study?
The hippocampus is involved in memory, stress regulation, and mood circuitry. The comorbid group had lower left and right hippocampal volume (β = −0.062 and −0.088), while the pain-only and depression-only groups did not show the same bilateral hippocampal finding in the reported control contrasts.
That makes hippocampal volume part of the comorbidity-specific signal, not a generic chronic-pain result.
What would make the result more clinically actionable?
Longitudinal MRI with repeated pain and depression measurement would matter most. The next useful evidence would show whether the cortical, subcortical, and white-matter differences appear before comorbid symptoms, worsen with persistent symptoms, improve with integrated treatment, or simply mark people with higher baseline vulnerability.
Should treatment change because of this MRI study?
Not by itself. The study supports integrated care logic but does not test a treatment.
Clinicians still have to use symptom severity, pain mechanism, functional impairment, medication history, sleep, substance use, and patient preference. The result mainly argues against treating pain and depression as unrelated problems when they co-occur.
References
- Casey H, Adams MJ, McIntosh AM, et al. Structural brain characteristics of current co-occurring chronic pain and depression: a cross-sectional analysis of UK Biobank. medRxiv. 2026. doi:10.64898/2026.04.02.26350033
- Aaron RV, Ravyts SG, Carnahan ND, et al. Prevalence of depression and anxiety among adults with chronic pain: a systematic review and meta-analysis. JAMA Network Open. 2025;8(3):e250268. doi:10.1001/jamanetworkopen.2025.0268
- Bair MJ, Robinson RL, Katon W, Kroenke K. Depression and pain comorbidity: a literature review. Archives of Internal Medicine. 2003;163(20):2433–2445. doi:10.1001/archinte.163.20.2433
- Kroenke K, Spitzer RL, Williams JB. The Patient Health Questionnaire-2: validity of a two-item depression screener. Medical Care. 2003;41(11):1284–1292. doi:10.1097/01.MLR.0000093487.78664.3C
- Lee J, Protsenko E, Lazaridou A, et al. Encoding of self-referential pain catastrophizing in the posterior cingulate cortex in fibromyalgia. Arthritis & Rheumatology. 2018;70(8):1308–1318. doi:10.1002/art.40507
- Bhatt RR, Haddad E, Zhu AH, et al. Mapping brain structure variability in chronic pain: the role of widespreadness and pain type and its mediating relationship with suicide attempt. Biological Psychiatry. 2024;95(5):473–481. doi:10.1016/j.biopsych.2023.07.016
- Harris MA, Cox SR, de Nooij L, et al. Structural neuroimaging measures and lifetime depression across levels of phenotyping in UK Biobank. Translational Psychiatry. 2022;12(1):157. doi:10.1038/s41398-022-01926-w
- Shen X, Reus LM, Cox SR, et al. Subcortical volume and white matter integrity abnormalities in major depressive disorder: findings from UK Biobank imaging data. Scientific Reports. 2017;7(1):5547. doi:10.1038/s41598-017-05507-6
- Yu W, Tao B, Zhu F, et al. Shared cortical characteristics in major depressive disorder, anxiety disorder, and chronic pain: a structural MRI meta-analysis study. Translational Psychiatry. 2025;15(1):430. doi:10.1038/s41398-025-03424-1
- Gustin SM, Peck CC, Macey PM, Murray GM, Henderson LA. Unraveling the effects of plasticity and pain on personality. The Journal of Pain. 2013;14(12):1642–1652. doi:10.1016/j.jpain.2013.08.005
- Ma T, Ji YY, Yan LF, et al. Gray matter volume abnormality in chronic pain patients with depressive symptoms: a systematic review and meta-analysis of voxel-based morphometry studies. Frontiers in Neuroscience. 2022;16:826759. doi:10.3389/fnins.2022.826759
- Roughan WH, Campos AI, Garcia-Marin LM, et al. Comorbid chronic pain and depression: shared risk factors and differential antidepressant effectiveness. Frontiers in Psychiatry. 2021;12:643609. doi:10.3389/fpsyt.2021.643609