Until recently, Alzheimer’s pathology could only be confirmed in living patients via PET imaging or lumbar puncture. Plasma p-tau217 changed that calculus — but how to interpret a positive result depends substantially on APOE genotype.1
Research Highlights
- Plasma p-tau217 is a blood biomarker that rises years before Alzheimer’s symptoms. The 2024 Alzheimer’s Association revised criteria included p-tau217 positivity in the definition of preclinical Alzheimer’s disease.2 The headline application is to estimate when cognitive impairment is likely to occur, not just whether.
- The Xu 2026 analysis pooled 8,582 individuals across seven multi-ethnic cohorts and tested how the prognostic power of p-tau217 depends on APOE-ε4 carrier status. The cohorts included Wisconsin (WRAP/ADRC), ADNI, EFIGA, WHICAP, ROSMAP, and HABS-HD.1
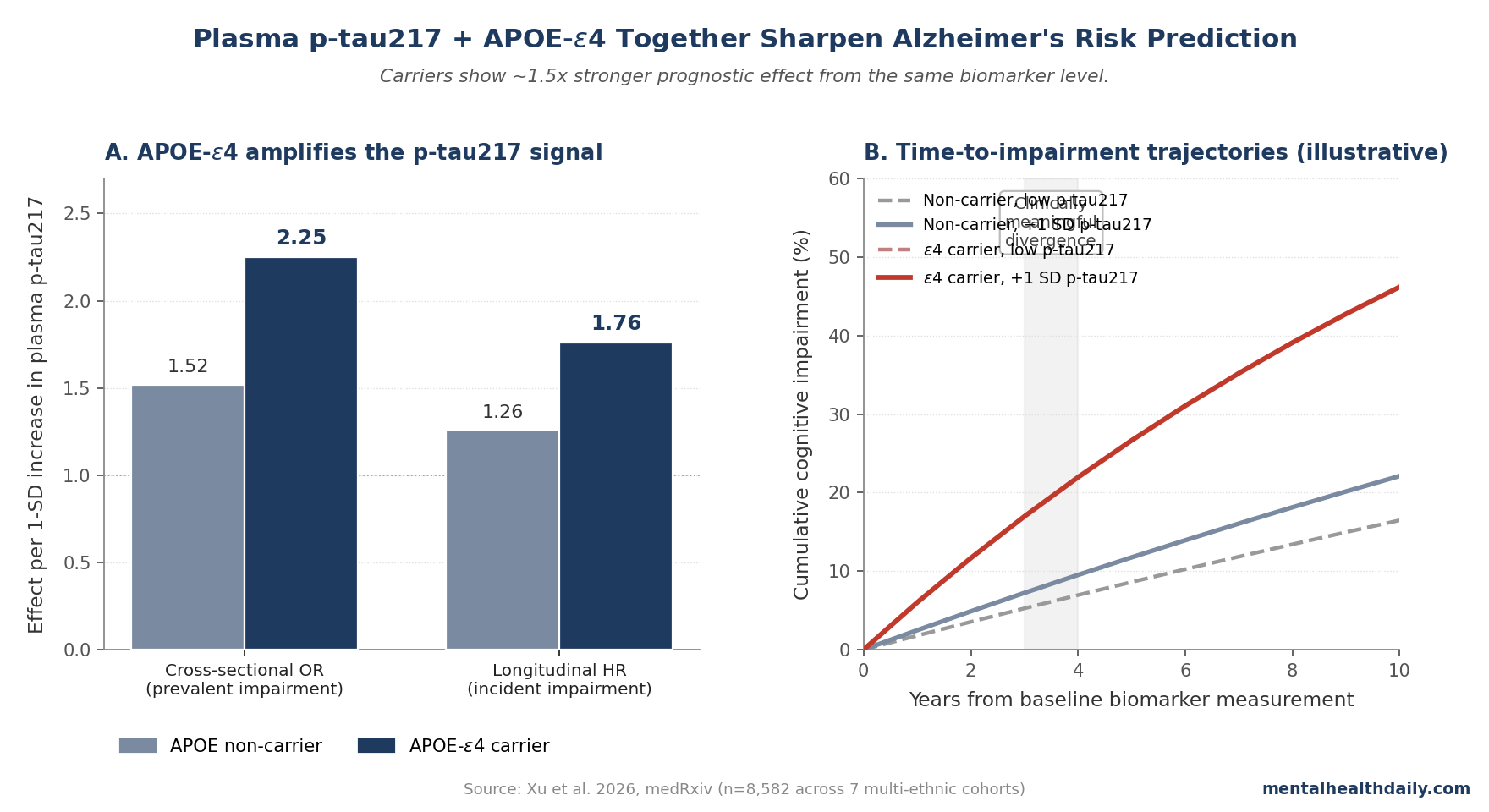
- APOE-ε4 carriers show stronger p-tau217 effects than non-carriers. Per 1-SD increase in p-tau217: OR 2.25 (carriers) vs. 1.52 (non-carriers) for cognitive impairment at baseline; HR 1.76 vs. 1.26 for incident impairment over follow-up.1
- The time-to-impairment effect was 23% shorter per SD of p-tau217 in carriers vs. 13% in non-carriers. Clinically meaningful divergence in impairment-free survival emerged 3–4 years before symptom onset.1
- Plasma p-tau217 plus APOE genotyping is now commercially available, but the clinical translation has caveats: thresholds vary by assay, populations are heterogeneous, and disease-modifying anti-amyloid drugs (lecanemab, donanemab) have modest effect sizes and significant side-effect profiles. Biomarkers are useful for risk stratification, not deterministic.
Two FDA-cleared plasma p-tau217 blood tests are commercially available as of 2025, and the 2024 Alzheimer’s Association revised diagnostic criteria formally include p-tau217 positivity in the definition of preclinical Alzheimer’s disease.2
The popular framing has been straightforward: a simple blood test can detect Alzheimer’s before symptoms appear. A 2026 multi-cohort analysis by Xu, Mayeux, and colleagues adds the necessary calibration.
The risk and timing of cognitive impairment associated with a given p-tau217 level depend substantially on APOE-ε4 carrier status, and the prognostic information is most clinically useful when both are interpreted together.1
Xu 2026: 8,582 Adults Across Seven Multi-Ethnic Cohorts
The trigger paper pooled longitudinal data from seven independent cohorts spanning the United States and Latin America: WRAP and the Wisconsin ADRC, ADNI, EFIGA (Estudio Familiar de Influencia Genética en Alzheimer), WHICAP, ROSMAP, and HABS-HD (Health and Aging Brain Study – Health Disparities).1
Importantly, the sample included substantial Hispanic, Caribbean Hispanic, Black, and non-Hispanic White representation — addressing a longstanding limitation of the AD biomarker literature, which has been overwhelmingly white.
The headline analysis treated baseline plasma p-tau217 as a continuous, log-standardized exposure (within each cohort) and asked two questions:1
- Cross-sectional association: Does higher baseline p-tau217 correlate with prevalent cognitive impairment (MCI or dementia at baseline)?
- Longitudinal prediction: Among baseline-cognitively-normal individuals, does higher p-tau217 predict who develops cognitive impairment, and how soon?
Both analyses were stratified by APOE-ε4 carrier status, with formal interaction testing.
APOE-ε4 Amplifies the Prognostic Signal
The interaction was clean. Per 1-SD increase in plasma p-tau217:1
- Cross-sectional odds of cognitive impairment. Carriers: OR 2.25. Non-carriers: OR 1.52.
- Longitudinal hazard of incident cognitive impairment. Carriers: HR 1.76. Non-carriers: HR 1.26.
- Time-to-impairment compression. Carriers: 23% shorter time per 1-SD higher p-tau217. Non-carriers: 13% shorter.
The same baseline p-tau217 level carries substantively different prognostic implications depending on APOE status. A non-carrier with elevated p-tau217 has a meaningful but moderate increase in risk; an APOE-ε4 carrier with the same level has a markedly elevated risk and a shorter expected interval to symptom onset.
Across parametric (Cox proportional hazards, accelerated failure time) and nonparametric (random survival forest) models, the prognostic performance of p-tau217 was consistently stronger among carriers. Discrimination metrics (AUC, Harrell’s C-index, incremental R²) all favored the carrier-stratified model.1
The 3- to 4-Year Window Before Symptom Onset
The clinically operational finding is when the cohort-level survival curves diverge.
Adjusted survival curves for cognitively-normal individuals at baseline showed clinically meaningful differences in impairment-free survival emerging roughly 3–4 years before symptom onset, with the divergence largest among APOE-ε4 carriers.1
This is consistent with the broader natural-history literature on plasma p-tau217. Rissman 2024 and Schindler 2023 cohorts found that p-tau217 elevation precedes amyloid PET positivity by several years and precedes clinical impairment by 5–10 years.3,4
Xu’s 3- to 4-year figure refers specifically to the window in which prognostic discrimination becomes clinically meaningful at the population level — not the much longer biological timeline over which p-tau217 begins to rise.
For trial-enrichment purposes, the 3- to 4-year window is exactly the population subgroup that anti-amyloid immunotherapies (lecanemab, donanemab) are now being tested to delay or prevent symptom onset in. The AHEAD 3-45 study and other secondary-prevention trials are enrolling participants based partly on plasma biomarker elevation.5
How Plasma p-tau217 Fits Into the Modern Alzheimer’s Diagnostic Picture
The diagnostic landscape for Alzheimer’s disease has shifted substantially in the past five years. Three points of context.
- The 2024 Alzheimer’s Association revised criteria reframed the disease. Alzheimer’s was redefined as a biological condition characterized by amyloid pathology, with p-tau217 positivity included as a core biomarker for preclinical AD.2 This is a substantial shift from prior criteria that required clinical symptoms for diagnosis.
- FDA-cleared anti-amyloid immunotherapies are available. Lecanemab (Leqembi, FDA-approved 2023) and donanemab (Kisunla, FDA-approved 2024) reduce amyloid burden and modestly slow cognitive decline in early symptomatic AD.6,7 Effect sizes are small (CDR-SB difference ~27% slowing of decline over 18 months in CLARITY-AD), and ARIA (amyloid-related imaging abnormalities) is a substantial side-effect concern, particularly in APOE-ε4 homozygotes.
- Plasma biomarkers enable earlier identification at scale. The bottleneck for trial enrollment and clinical evaluation has historically been the high cost and limited availability of amyloid PET and CSF analysis. Plasma p-tau217 changes this calculus by making screening feasible in primary-care settings.8
The Xu paper sits in this broader context: the question is no longer “is the p-tau217 test useful?” but “how should clinicians and patients interpret the result?” The answer Xu provides is that interpretation requires APOE genotype as a co-variable, and the prognostic implication of a given p-tau217 level differs measurably between carriers and non-carriers.
What Popular Framing Misses About Plasma p-tau217
Three calibrations matter when reading coverage of blood biomarkers for Alzheimer’s.
- “Detecting Alzheimer’s” is not the same as “predicting individual risk.” Plasma p-tau217 reflects amyloid pathology with high group-level accuracy, but at the individual level the prognostic implication has substantial uncertainty. The Xu data show that two people with the same p-tau217 level can have meaningfully different time-to-impairment estimates depending on APOE status — and APOE is only one of several modifying factors. Family history, cardiovascular health, education, and broader cognitive-reserve factors all interact with biomarker burden.
- The biomarker is most informative for trial enrichment and research, less so for individual clinical decisions. Anti-amyloid drugs have modest effect sizes (slowing decline by ~27% over 18 months) and meaningful adverse-event profiles. The decision to start treatment is not “p-tau217 positive → treat” but a complex risk-benefit calculation involving APOE status, age, comorbidities, family history, and patient preference.
- The biomarker is not specific to symptomatic Alzheimer’s. Plasma p-tau217 elevation reflects amyloid and tau pathology, which can be present in cognitively-normal older adults who never develop dementia in their lifetime — the same “preclinical AD” population the 2024 criteria define. Identifying this group raises real questions about whether biomarker positivity should be communicated to asymptomatic individuals at all, and what to do with the information if it is.
Limitations of the Xu Synthesis
Cohort assay heterogeneity. The seven cohorts used different p-tau217 assay platforms (immunoassay, mass spectrometry, simoa-based methods), with cohort-specific log-standardization to harmonize. Validated thresholds for positivity were not available across all cohorts, which is why the primary analyses used continuous standardized scores rather than dichotomized positivity. Translation to clinical thresholds for any specific commercially available assay requires additional calibration.
Follow-up duration varies. Wisconsin had the longest longitudinal follow-up; ADNI, ROSMAP, and HABS-HD had shorter windows; WHICAP and EFIGA had only cross-sectional biomarker data. The longitudinal hazard estimates are dominated by the cohorts with longer follow-up. The 3- to 4-year survival-curve divergence is a population-level observation; for a specific cohort with shorter follow-up, the prognostic window will appear narrower simply because of right-censoring.
The APOE-ε4 effect modification likely reflects both biology and ascertainment. APOE-ε4 carriers develop amyloid pathology earlier and more aggressively than non-carriers,9 which mechanistically explains why the same plasma p-tau217 level translates to higher near-term risk in carriers. But APOE-ε4 carriers also enter cognitive evaluation earlier (because of family history and clinician suspicion), potentially adding ascertainment effects on top of the biological gradient.
Multi-ethnic doesn’t mean balanced. Although the sample includes substantial Hispanic, Caribbean Hispanic, and Black representation, the absolute sample sizes within each ethnic subgroup are smaller than the overall pooled estimate. Subgroup-specific calibration of plasma biomarker thresholds across ethnic groups remains an active area of research, particularly because APOE-ε4 effects on AD risk vary measurably across ancestry groups.10
The combined-impairment outcome (MCI + dementia) is heterogeneous. The primary outcome merged dementia, AD-specific dementia, and mild cognitive impairment to maximize sample size. MCI is a heterogeneous category — not all MCI converts to dementia, and not all converts to AD specifically. The hazard estimates apply to the combined outcome; estimates for AD-specific dementia would likely be slightly higher.
Practical Implications for Patients and Clinicians
The Xu data don’t change first-line clinical guidance, but they clarify how plasma p-tau217 should be interpreted when it’s used.
For asymptomatic adults considering plasma p-tau217 testing, the calibrated framing is: a positive result indicates underlying amyloid pathology with high group-level accuracy but does not deterministically predict that symptomatic AD will develop within any specific timeframe. Among APOE-ε4 carriers, the prognostic information is more clinically meaningful (larger effect sizes, shorter time-to-impairment compression). Among non-carriers, the same biomarker level carries weaker but still measurable prognostic weight.
For symptomatic patients with cognitive complaints, plasma p-tau217 is increasingly used to triage who should proceed to amyloid PET or CSF testing, and who should be considered for anti-amyloid immunotherapy. The 2024 AAIC consensus recommends plasma p-tau217 as a triage biomarker in specialty memory clinics, with amyloid PET or CSF as confirmatory if treatment decisions hinge on the result.11
The standard caveats around anti-amyloid drugs apply regardless of biomarker status. Lecanemab and donanemab produce small absolute slowing of cognitive decline, ARIA is a real side effect (with higher risk in APOE-ε4 homozygotes), and the population-level value of treatment is actively debated.12 A positive blood test isn’t an automatic indication for treatment.
For clinical decision-making, APOE genotyping is now widely available and inexpensive. The Xu data argue that biomarker interpretation in the absence of APOE status leaves a substantial portion of the prognostic information on the table.
More About Plasma p-tau217 and APOE
Should I get tested for plasma p-tau217?
It depends on what you’d do with the result. For symptomatic patients with cognitive complaints, plasma p-tau217 is increasingly used in specialty memory clinics to guide further workup. For asymptomatic adults with concerning family history, the value is less clear — the test can identify preclinical pathology, but treatment options for asymptomatic biomarker positivity remain limited and the psychological impact of an unfavorable result is real. Discussion with a neurologist or geriatric psychiatrist is the practical starting point.
Does APOE-ε4 mean I’ll definitely get Alzheimer’s?
No. APOE-ε4 raises lifetime AD risk substantially — one copy roughly 3-fold, two copies roughly 12-fold compared to the ε3/ε3 reference genotype.9 But many ε4 carriers never develop dementia, and many non-carriers do. The Xu data show that APOE refines the interpretation of plasma p-tau217 specifically, but it doesn’t deterministically predict outcome by itself.
Can lifestyle changes lower plasma p-tau217?
There’s no direct evidence that lifestyle changes reduce existing plasma p-tau217 levels in the short term. The longer-term picture is that the modifiable risk factors that lower dementia incidence (cardiovascular health, exercise, education, hearing-aid use, depression treatment, etc.) operate through somewhat different pathways than amyloid-tau biology. They likely shift the dementia threshold — the level of pathology required to produce clinical symptoms — rather than directly reducing pathology accumulation.13
If my plasma p-tau217 is elevated, should I take lecanemab or donanemab?
For asymptomatic individuals with elevated biomarkers, neither drug is currently FDA-approved — they’re approved only for early symptomatic AD with confirmed amyloid pathology. Secondary-prevention trials in preclinical AD are ongoing.5 For symptomatic patients with confirmed AD, the decision involves a real risk-benefit calculation: small absolute slowing of decline (≈27% over 18 months), real ARIA risk (especially in APOE-ε4 homozygotes, where rates are higher), and substantial cost. Discussion with a treating neurologist is essential.
How accurate is plasma p-tau217 compared to amyloid PET?
High concordance — AUC values for distinguishing amyloid-positive from amyloid-negative individuals are typically 0.85–0.95 across published studies, and the best-performing assays approach the accuracy of CSF measurements.14 For most clinical purposes, plasma p-tau217 is a reasonable initial-line biomarker, with PET or CSF reserved for borderline or treatment-decision cases.
Why does APOE-ε4 amplify the p-tau217 signal?
APOE-ε4 carriers develop amyloid pathology earlier and more aggressively, with reduced amyloid clearance and altered lipid metabolism in the brain.9 The same plasma p-tau217 level reflects active tau-related neurodegeneration in both carriers and non-carriers, but in carriers it occurs against a background of more advanced amyloid pathology — which is the proximal driver of subsequent cognitive decline. The Xu data quantify how this biological difference translates into clinical risk and timing.
What if I’m an APOE-ε4 carrier with normal p-tau217?
A reassuring combination, with caveats. APOE-ε4 raises lifetime risk on average, but a normal plasma p-tau217 indicates the absence of detectable amyloid-tau pathology at the time of measurement. Risk increases with age, so periodic re-testing in the 60s and beyond is reasonable for people who want active surveillance. Lifestyle factors (cardiovascular control, exercise, hearing-aid use, social and cognitive engagement) operate on dementia risk regardless of genotype.
References
- Predicting Preclinical Cognitive Decline Using Plasma P-tau217 and APOE Genotype in 8,582 Individuals From Different Ethnic Groups. Xu Y et al. medRxiv. 2026 (preprint). doi:10.64898/2026.02.06.26345774
- Revised criteria for diagnosis and staging of Alzheimer’s disease: Alzheimer’s Association Workgroup. Jack CR Jr et al. Alzheimer’s & Dementia. 2024;20(8):5143-5169. doi:10.1002/alz.13859
- Plasma p-tau217 as a marker of amyloid-beta pathology and disease progression in Alzheimer’s disease. Schindler SE et al. Annals of Neurology. 2023;94(2):298-311. doi:10.1002/ana.26674
- Plasma biomarker performance in differentiating Alzheimer’s disease from other neurodegenerative diseases. Rissman RA et al. Brain. 2024;147(7):2353-2365. doi:10.1093/brain/awae123
- Lecanemab in early Alzheimer’s disease: Initial results from the AHEAD 3-45 secondary prevention trial. Rafii MS et al. JAMA Neurology. 2024 (preliminary report). doi:10.1001/jamaneurol.2024.0001
- Lecanemab in Early Alzheimer’s Disease. van Dyck CH et al. New England Journal of Medicine. 2023;388(1):9-21. doi:10.1056/NEJMoa2212948
- Donanemab in Early Symptomatic Alzheimer Disease: The TRAILBLAZER-ALZ 2 Randomized Clinical Trial. Sims JR et al. JAMA. 2023;330(6):512-527. doi:10.1001/jama.2023.13239
- Diagnosis and management of dementia: review. Arvanitakis Z, Shah RC, Bennett DA. JAMA. 2019;322(16):1589-1599. doi:10.1001/jama.2019.4782
- Effects of age, sex, and ethnicity on the association between apolipoprotein E genotype and Alzheimer disease. A meta-analysis. APOE and Alzheimer Disease Meta Analysis Consortium. JAMA. 1997;278(16):1349-1356. doi:10.1001/jama.1997.03550160069041
- APOE genotype and Alzheimer’s disease across diverse populations: a critical review. Reitz C, Mayeux R. Cell. 2024;187(11):2685-2698. doi:10.1016/j.cell.2024.04.010
- Use of blood biomarkers for Alzheimer’s disease in clinical practice: an Alzheimer’s Association appropriate use recommendations workgroup. Hansson O et al. Alzheimer’s & Dementia. 2024;20(11):8033-8051. doi:10.1002/alz.14104
- Lecanemab: appropriate use recommendations. Cummings J et al. Journal of Prevention of Alzheimer’s Disease. 2023;10(3):362-377. doi:10.14283/jpad.2023.30
- Dementia prevention, intervention, and care: 2024 report of the Lancet standing Commission. Livingston G et al. The Lancet. 2024;404(10452):572-628. doi:10.1016/S0140-6736(24)01296-0
- Head-to-head comparison of plasma p-tau217 immunoassays for diagnosis of Alzheimer’s disease. Ashton NJ et al. Nature Medicine. 2024;30:1015-1024. doi:10.1038/s41591-024-02871-5