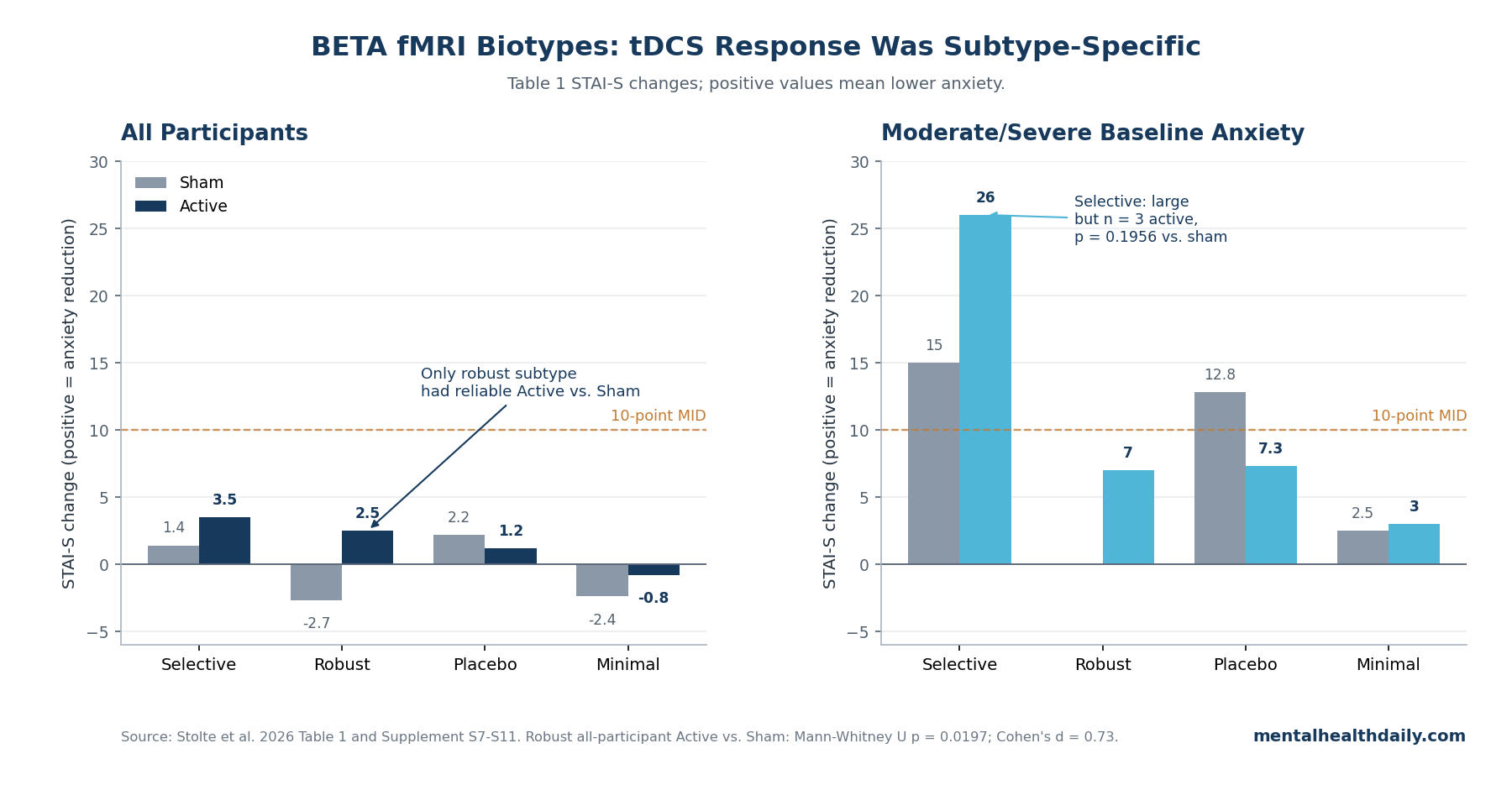
A 2026 BETA analysis of 199 older adults found that resting-state fMRI could separate 4 anxiety-related tDCS response patterns, but the result was narrower than “brain scan predicts treatment”: only the Robust tDCS Responder subtype showed a statistically reliable Active vs. Sham anxiety advantage across all participants.1
Research Highlights
- Only 1 subtype cleared the Active vs. Sham test: the Robust tDCS Responder subtype favored active stimulation across all participants (Mann-Whitney U = 753, p = 0.0197; Cohen’s d = 0.73).1
- The large Selective-subtype number came from 5 people: moderate/severe anxiety improved by 26.0 STAI-S points with active tDCS vs. 15.0 with sham, but the Active vs. Sham permutation test was not statistically reliable (p = 0.1956).1
- Placebo response was not a footnote: the Placebo Responder subtype showed 12.8-point moderate/severe sham improvement vs. 7.3 points with active stimulation, with no reliable between-condition difference (p = 0.3946).1
- The machine-learning signal depended on outcome guidance: adding the clinical-informed loss separated clusters better than the ablated model (Welch’s ANOVA p = 0.02 vs. p = 0.10).1
- Biotype evidence is still research-stage: larger depression/anxiety biotype work than the 199-person BETA sample used 801 treatment-free patients and 250 randomized-treatment participants, while replication critiques show why single-dataset fMRI clusters need external validation.2,3
Transcranial direct current stimulation (tDCS) delivers a weak electrical current through scalp electrodes to change cortical excitability. In the ACT dataset used by Stolte et al., active stimulation delivered 2 mA for 20 minutes during each cognitive-training session, while sham delivered the same setup with only a brief 30-second current period.1
Resting-state fMRI measures how brain regions fluctuate together while a person is not doing a task. BETA used those resting connectivity patterns to build biotypes: data-driven subgroups meant to capture circuit differences hidden inside similar anxiety scores.1
BETA Found 4 Retained fMRI Biotypes After Dropping a 3-Person Cluster
Stolte et al. analyzed the Augmenting Cognitive Training in Older Adults trial, a randomized, double-blind study of tDCS paired with cognitive training in adults aged 65-89. After excluding participants with missing anxiety or fMRI data, the analyzed sample included 199 people: 99 assigned to sham tDCS and 100 assigned to active tDCS.1
The model began with anxiety-related and aging-related brain regions, then turned pairwise resting-state functional connectivity into a 50-dimensional latent embedding. A variational autoencoder is a neural-network compression model: it tries to preserve the important structure in very high-dimensional data while reducing the data to a smaller space that can be clustered.
BETA added a clinical-informed loss term so participants with similar State-Trait Anxiety Inventory-State (STAI-S) change sat closer together in that latent space.1
That detail is essential. BETA was not a purely unsupervised scan-only discovery that later happened to predict response.
The model used STAI-S change during training to shape the space, then assigned held-out participants to clusters during 5-fold cross-validation. The design is still useful, but it is closer to treatment-response-enriched clustering than to an off-the-shelf clinical diagnostic test.1
The researchers initialized 5 clusters, then excluded 1 tiny cluster with n = 3 because its connectivity and clinical trajectories were too idiosyncratic for meaningful inference. The remaining 4 groups were labeled Selective tDCS Responder, Robust tDCS Responder, Placebo Responder, and Minimal Responder.1
Robust tDCS Responder Was the Clearest Active-Stimulation Signal
The headline result lives in the Robust tDCS Responder subtype, not in a generic claim that all fMRI biotypes predicted tDCS response. In the all-participant Robust subgroup, The main response table reported mean STAI-S change of 2.5 points with active tDCS vs. −2.7 points with sham.
Supplementary testing found a statistically reliable Active vs. Sham difference (Welch’s t = 2.95, p = 0.0046; Mann-Whitney U = 753, p = 0.0197; Cohen’s d = 0.73).1
The same subtype also showed the most reliable result among participants with minimal/mild baseline anxiety: active stimulation outperformed sham with Welch’s t = 2.65, p = 0.0105, Mann-Whitney U p = 0.0309, and Cohen’s d = 0.59. Low baseline anxiety limits how much symptom improvement a treatment can show.
The mean baseline STAI-S score was 27.95 on a 20-80 scale, and the paper notes that only around 10-20% of participants in each group had clinically relevant anxiety.1
Clinically, this makes the Robust subgroup interesting but not ready. A 5-point Active vs. Sham separation in a research table can guide trial enrichment; it cannot tell an individual older adult to seek fMRI-based tDCS matching.
Selective tDCS Response Was Large But Statistically Fragile
The Selective tDCS Responder subtype is where a careless draft can overstate the paper. The main response table shows a striking moderate/severe anxiety result: active tDCS improved STAI-S by 26.0 points, while sham improved by 15.0 points.
Both numbers exceed the 10-point minimal important difference the paper cites from Corsaletti et al.1
The problem is the cell size. That comparison used 3 active participants and 2 sham participants.
Supplementary Table S7 reported an observed Active-Sham difference of +11.00 points, but the permutation p-value was 0.1956. In plain language, the point estimate favored active stimulation, but the sample was too small to rule in a reliable between-condition effect.1
For MHD purposes, the calibrated read is simple: the Selective subtype is a lead worth following, not proof that the model has already found a deployable responder class. The article should not flatten it into “no signal,” because the directional number is large.
It also should not market it as confirmed response prediction, because the Active vs. Sham test failed.
Placebo and Minimal Responder Patterns Keep the Claim Honest
The Placebo Responder subtype blocks a simple tDCS-success narrative. In moderate/severe participants, sham improved by 12.8 STAI-S points and active tDCS by 7.3 points.
Supplementary Table S14 reported no reliable Active vs. Sham difference (permutation p = 0.3946; Cohen’s d = −0.56). Across all participants in that subtype, active and sham changes were similar, and Mann-Whitney U testing was far from significant (p = 0.9395).1
The Minimal Responder subtype also did what its name suggests. The main response table reported small all-participant changes: −2.4 with sham and −0.8 with active tDCS.
Supplementary tests were nonsignificant (Welch’s p = 0.5960; Mann-Whitney U p = 0.5822).1
These groups are not failures of the paper. They are the paper’s core argument: same broad clinical context, different response pattern.
The key is to preserve the response-direction detail instead of collapsing everything into “biotypes predict response.”
LOC, Angular Gyrus, and Frontal-Control Connections Drove the Biotype Story
The biotype labels are not arbitrary symptom names. Stolte et al. reported that responsive subtypes tended to show stronger connectivity involving the lateral occipital cortex (LOC), angular gyrus, and frontal-control regions.
The LOC is a visual-processing region, while the angular gyrus helps integrate attention, memory, language, and self-related perspective-taking. Frontal-control regions are relevant because the stimulation montage targeted F3-F4 scalp locations over dorsolateral prefrontal cortex.1
Supplementary Table S3 gives concrete examples. The Selective tDCS Responder subtype showed stronger left superior LOC-medial frontal cortex connectivity (0.399 vs. cohort mean 0.219 ± 0.038) and stronger left superior LOC-left intracalcarine cortex connectivity (0.371 vs. 0.217 ± 0.035).
The Robust subtype showed stronger left superior frontal gyrus-right angular gyrus connectivity (0.450 vs. 0.398 ± 0.034) and stronger angular gyrus-left superior LOC connectivity (0.661 vs. 0.580 ± 0.037).1
That circuit pattern is plausible because anxiety can involve sensory threat processing, salience detection, and top-down regulation. Langhammer et al., in a much larger multicenter resting-state fMRI study, found that anxiety-disorder connectivity patterns differed by diagnosis: panic/agoraphobia, social anxiety disorder, and specific phobia did not collapse into one uniform fMRI signature.4
Biotypes Are Plausible, But Replication Is the Hard Part
Modern biotype work is not fantasy. Tozzi et al. used personalized brain circuit scores in depression and anxiety and identified 6 biotypes across 801 treatment-free patients, then examined treatment-related data in 250 randomized participants.
That study used multiple validation checks, including split-half and stability procedures, and linked biotypes to symptoms, behavior, and treatment response.2
The caution comes from the same literature. Drysdale et al. previously reported 4 depression biotypes from resting-state connectivity, but Dinga et al. later argued that the evidence for those specific biotypes was not robust enough under replication-focused tests.
The lesson for BETA is not “ignore fMRI biotypes.” The lesson is that cluster solutions can look compelling inside one dataset and then weaken when tested in a new sample, scanner environment, preprocessing pipeline, or outcome definition.3,8
BETA is strongest as a trial-design tool. If a future study uses the model prospectively to enroll predicted responders, randomizes within those strata, and still finds the Robust-style active advantage, the clinical case becomes much stronger.
tDCS in Older Adults Still Needs the Usual Clinical Guardrails
tDCS is attractive in older adults because medication options for anxiety can be awkward: sedation, falls, hyponatremia, drug interactions, and anticholinergic burden all carry more risk in geriatric care. But the ACT dataset was not a medication-vs.-tDCS anxiety trial, and participants were relatively healthy older adults rather than a clinic sample selected for anxiety disorder.1,6
Guidelines and reviews generally treat tDCS as tolerable but indication-specific, protocol-dependent, and uneven in efficacy. A 2022 Alzheimer disease pilot combining rTMS and tDCS over bilateral angular gyrus also sits near this question, but it tested a different stimulation target and a different clinical context.5,7
So the practical translation is conservative: fMRI-guided neuromodulation for anxiety in Alzheimer’s-risk older adults is a research direction, not an available personalized-treatment standard.
Evidence Limits in the BETA tDCS Anxiety Analysis
Low baseline anxiety limits treatment inference: most participants were not highly anxious at baseline. When baseline symptoms are mild, there is less room for an anxiety treatment to show a large clinical effect, and subgroup labels can become sensitive to small score changes.1
Tiny cells drive the most dramatic numbers: the 26.0-point Selective active result came from n = 3, and the corresponding sham cell had n = 2. The Robust moderate/severe group had 5 active participants and 0 sham participants, so that subgroup could not support a condition comparison.1
The paper has internal sign noise: main Table 1 defines positive STAI-S pre-post change as anxiety reduction, but several supplementary tables use negative mean-change signs while the surrounding prose describes those values as reductions. The reliability tests still identify the Robust subtype as the most defensible Active vs. Sham result, but descriptive change values should be read through the main Table 1 definition.1
No external validation exists yet: the model needs an independent cohort and, ideally, a prospective trial that assigns treatment using pre-specified biotype strata before any clinical screening claim is justified.
Questions About fMRI Biotypes and tDCS for Anxiety
Did BETA prove fMRI can choose tDCS treatment for older adults with anxiety?
No. It showed a research-stage pattern in 199 older adults, with the strongest Active vs. Sham evidence concentrated in 1 subtype.
That supports designing the next trial, not routine clinical selection.1
Which BETA subtype had the clearest active-stimulation advantage?
The Robust tDCS Responder subtype. Across all participants, active tDCS beat sham with Mann-Whitney U p = 0.0197 and Cohen’s d = 0.73; the minimal/mild subgroup also favored active stimulation.1
Why is the 26.0-point Selective-subtype result not enough by itself?
Because it came from 3 active participants with moderate/severe baseline anxiety. The active group improved more than the 2-person sham group, but the Active vs. Sham permutation test was not significant (p = 0.1956).1
Should someone get resting-state fMRI before trying tDCS for anxiety?
No. BETA is not validated as a clinical screening tool.
Resting-state fMRI is expensive, analysis-dependent, and not yet tied to a prospective decision rule for anxiety treatment.
What would make the BETA result clinically stronger?
An independent, pre-registered trial would need to assign patients to BETA biotypes before treatment, randomize active vs. sham tDCS inside those strata, and reproduce the Robust-subtype advantage with enough clinically anxious participants in each cell.
References
- Stolte SE, Cheng J, Acharya C, et al. BETA: resting-state fMRI biotypes for tDCS efficacy in anxiety among older adults at risk for Alzheimer’s disease. Proceedings of Machine Learning Research / MIDL 2026. 2026;108:1-29. doi:10.64898/2026.04.24.26351493
- Tozzi L, Zhang X, Pines A, et al. Personalized brain circuit scores identify clinically distinct biotypes in depression and anxiety. Nature Medicine. 2024;30:2076-2087. doi:10.1038/s41591-024-03057-9
- Dinga R, Schmaal L, Penninx BWJH, et al. Evaluating the evidence for biotypes of depression. NeuroImage: Clinical. 2019;22:101796. doi:10.1016/j.nicl.2019.101796
- Langhammer T, Hilbert K, Adolph D, et al. Resting-state functional connectivity in anxiety disorders: a multicenter fMRI study. Molecular Psychiatry. 2025;30:1548-1557. doi:10.1038/s41380-024-02768-2
- Hu Y, Jia Y, Sun Y, et al. Efficacy and safety of simultaneous rTMS-tDCS over bilateral angular gyrus on neuropsychiatric symptoms in patients with moderate Alzheimer’s disease: a prospective, randomized, sham-controlled pilot study. Brain Stimulation. 2022;15:1530-1537. doi:10.1016/j.brs.2022.11.009
- Mendez MF. The relationship between anxiety and Alzheimer’s disease. Journal of Alzheimer’s Disease Reports. 2021;5:171-177. doi:10.3233/ADR-210294
- Lefaucheur JP, Antal A, Ayache SS, et al. Evidence-based guidelines on the therapeutic use of transcranial direct current stimulation (tDCS). Clinical Neurophysiology. 2017;128:56-92. doi:10.1016/j.clinph.2016.10.087
- Drysdale AT, Grosenick L, Downar J, et al. Resting-state connectivity biomarkers define neurophysiological subtypes of depression. Nature Medicine. 2017;23:28-38. doi:10.1038/nm.4246