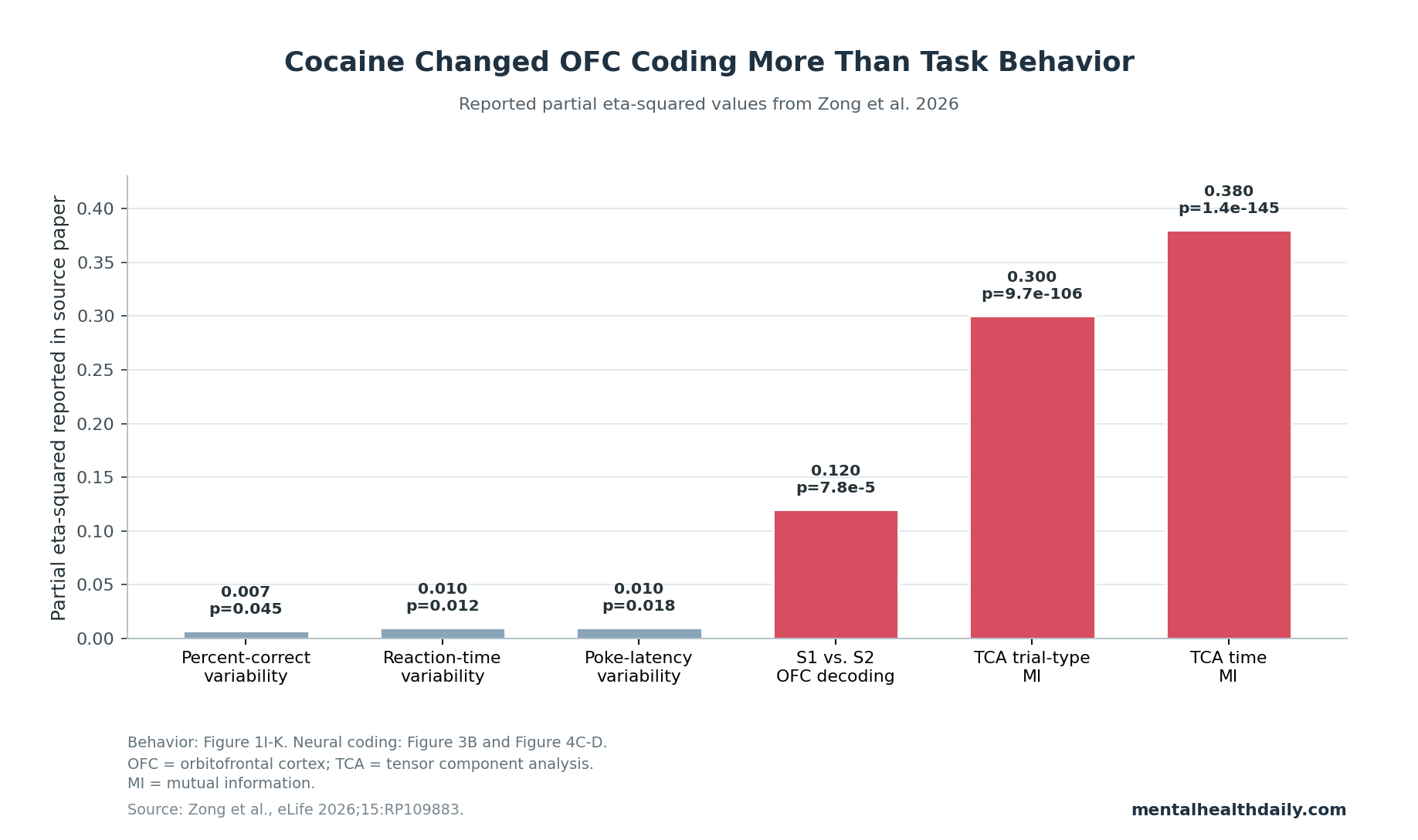
A 2026 eLife study recorded 3,881 lateral orbitofrontal cortex units in rats and found that prior cocaine use made the OFC over-distinguish task positions that controls treated as functionally equivalent. The headline result was not gross task failure: cocaine-experienced rats still performed the odor task, but their OFC ensembles showed higher S1-vs.-S2 decoding than sucrose controls (F(1,118) = 16.8, p = 7.8 × 10−5, ηp2 = 0.12), consistent with impaired hidden-state compression.1
Research Highlights
- OFC over-distinguished irrelevant sequence cues: across 1,000-neuron pseudoensembles, cocaine-experienced rats showed higher S1-vs.-S2 decoding than sucrose controls (F(1,118) = 16.8, p = 7.8 × 10−5, ηp2 = 0.12).1
- The recording scale was large for a rat single-unit study: researchers analyzed 3,881 lateral OFC units, including 1,699 units from 3 cocaine-experienced rats and 2,182 units from 3 sucrose-control rats.1
- Behavior shifted in the same direction: cocaine-experienced rats showed larger S2-minus-S1 differences than sucrose-control rats for percent correct (p = 0.045), reaction time (p = 0.012), and poke latency (p = 0.018).1
- Population decoding showed a generalization problem: within-sequence decoding differed by group (p = 0.027), and across-sequence decoding also differed (p = 0.046), indicating altered position representation rather than a single-position artifact.1
- Tensor analysis supported a broader OFC organization change: a 10-component tensor component analysis found lower position-general structure in cocaine-experienced rats, with large group effects for mutual information with trial type (ηp2 = 0.30) and time (ηp2 = 0.38).1
Hidden states are the underlying situations that make different surface cues mean the same thing. In this paper, 2 odor sequences looked different at some points, but the middle positions had the same behavioral role.
A healthy OFC should learn that shared role and compress away irrelevant odor-sequence differences.
The cocaine result is sharper than a vague “addiction damages the frontal lobe” claim. Zong et al. tested a specific computational function: whether the OFC can represent latent task structure after cocaine exposure, rather than staying stuck on surface details.1
Hidden States Are Task Meanings, Not Odor Labels
Orbitofrontal cortex (OFC) is a frontal-brain region involved in using context, expected outcomes, and task structure to guide decisions. The OFC does more than track whether a cue is pleasant or unpleasant.
In cognitive-map models, it helps represent the “state space”: the internal map of which situation the animal is in, which outcomes are likely, and which cues are irrelevant surface variation.2
A hidden state is not directly visible. It is inferred from cues.
A simple example is learning that 2 different rooms have the same rule: if both rooms use the same reward schedule, the room color may change while the underlying state stays the same. OFC-dependent generalization means treating those rooms as equivalent when the difference does not matter.
That abstraction is clinically relevant because addiction often looks context-bound. A person may learn reasons not to use cocaine in 1 setting, then fail to carry that learning into a cue-heavy setting where drug seeking returns.
The rat task does not model human relapse by itself, but it isolates a neural operation that relapse models need: transferring learning across superficially different situations.
The Figure-8 Odor Task Tested Compression Across Shared Positions
Zong et al. trained 6 male Long-Evans rats on a figure-8 odor sequence task before drug exposure. The task used 6 odors arranged into 2 four-odor sequences: S1 was 5+, 0−, 1−, 2+, and S2 was 3+, 0−, 1−, 4+.
Positions P2 and P3 were shared across the 2 sequences, while P1 and P4 used different odors at the sequence edges.1
The design matters because P2 and P3 gave the researchers a clean test of hidden-state compression. If the rat has learned the task structure, the OFC should not keep treating the shared middle positions as strongly different just because they sit inside different sequence paths.
The surface path differs; the behavioral role is the same.
After the initial training, 3 rats self-administered cocaine at 0.75 mg/kg/infusion for 14 consecutive days, while 3 rats self-administered 10% wt/vol sucrose. Researchers then implanted electrodes and recorded lateral OFC activity while the animals returned to the odor task.
Each analysis therefore asks about prior cocaine exposure after task learning, not acute intoxication during task performance.
High task performance stayed intact: both groups showed consistently high discrimination performance across the odor positions. That point prevents an overread.
Cocaine did not make rats unable to do the task. The more interesting defect appeared underneath performance, in how OFC neurons represented equivalence across positions.
Cocaine Experience Kept OFC Units From Compressing Shared Positions
During recording, researchers analyzed 1,699 lateral OFC units from cocaine-experienced rats and 2,182 units from sucrose-control rats. Single-unit selectivity between comparable S1 and S2 positions was low in controls, especially at the shared P2 and P3 positions where compression was expected.
Cocaine-experienced rats maintained more selectivity, especially around late P3 and much of P4, where group comparisons showed significant differences (χ2 values > 4.8, p values < 0.03).1
Ensemble decoding made the same point more cleanly: S1-vs.-S2 decoding across positions and epochs was higher in cocaine-experienced ensembles than in sucrose-control ensembles (F(1,118) = 16.8, p = 7.8 × 10−5, ηp2 = 0.12).
In plain English: the cocaine-experienced OFC contained more information about which sequence path the rat was on, even when that distinction was supposed to be irrelevant to the shared task role.
The less-training comparison helps calibrate the finding. Zong et al. compared the current over-trained controls with an earlier OFC dataset from rats that had less training on the same task family.3
Less-trained rats showed decoding similar to the cocaine group (F(1,118) = 0.01, p = 0.92, ηp2 = 9.0 × 10−5) and higher than the current sucrose controls (F(1,118) = 18.1, p = 4.2 × 10−5, ηp2 = 0.13). Cocaine experience made the OFC look less like an over-trained abstraction system and more like an earlier-learning system that had not fully compressed the task.
Behavior Varied More Where OFC Over-Distinguished the Sequences
The behavioral data did not show a dramatic collapse in accuracy. Instead, cocaine-experienced rats became more variable across the 2 sequences.
That is exactly the behavioral signature expected if the OFC is over-weighting sequence-specific differences that should have been treated as irrelevant.
Three group effects captured the pattern. S2-minus-S1 differences were larger in the cocaine group for percent correct (F(1,572) = 4.0, p = 0.045, ηp2 = 0.007), reaction time (F(1,572) = 6.4, p = 0.012, ηp2 = 0.01), and poke latency (F(1,572) = 5.6, p = 0.018, ηp2 = 0.01).
Those effect sizes are small, but they point in the mechanistically coherent direction: behavior became more sequence-specific after cocaine exposure.1
Calibration: small behavioral effects do not make the neural result trivial. The task was already well learned, and both groups performed well.
The behavioral readout is valuable because it shows that neural over-distinguishing was not a detached statistical artifact; it corresponded to measurable differences in how consistently rats treated the 2 sequences as equivalent.
Tensor Analysis Found Fewer Position-General Components After Cocaine
Tensor component analysis (TCA) is a way to decompose high-dimensional neural data into lower-dimensional components that vary across neurons, time, and trials. Here, it asked whether the overall OFC population contained components that generalized across task positions, rather than only pairwise S1-vs.-S2 differences.
The researchers fit a 10-component model because that model produced consistent and reliable results. In sucrose controls, several components generalized across all 8 positions.
In cocaine-experienced rats, those position-general components were absent; all 10 factors distinguished rewarded vs. non-rewarded position pairs instead of preserving the broader position-general structure.1
The mutual-information analysis quantified the same difference. Controls had lower mutual information between trial factors and trial type (F(1,1360) = 571.6, p = 9.7 × 10−106, ηp2 = 0.30), meaning their factors depended less on the particular trial type.
Controls also had higher mutual information between trial factors and time (F(1,1360) = 850.1, p = 1.4 × 10−145, ηp2 = 0.38), consistent with a more temporally organized representation across the task.
The TCA result is why the paper is stronger than a single decoding plot. Pairwise decoding could have reflected an idiosyncratic feature of P3 or P4.
The population-level decomposition found a broader change in how OFC activity organized the task.
Adjacent Cocaine Studies Point to OFC-Dependent Flexibility
Zong et al. fits a longer cocaine-OFC literature, but it adds a narrower mechanism. Calu et al. found that withdrawal from cocaine self-administration produced long-lasting deficits in OFC-dependent reversal learning in rats.4
Schoenbaum and Setlow reported that cocaine made actions insensitive to outcomes without simply preventing extinction, again pointing toward impaired outcome-guided updating rather than a crude inability to learn.5
Lucantonio et al. pushed that mechanism further by showing that orbitofrontal activation could restore “insight” lost after cocaine use in rats.6 In that context, insight means using information about outcomes to guide behavior when old responses are no longer appropriate.
The 2026 hidden-state study gives that insight problem a representational explanation: if OFC ensembles fail to collapse equivalent states, then learning from 1 situation may not transfer well to another.
Newer ensemble work also narrows the anatomy. Mueller et al. reported that prior cocaine use diminished latent-information encoding by orbitofrontal ensembles but not medial prefrontal ensembles.7
Zong et al. sits directly beside that result: the lateral OFC did more than lose signal; it retained the wrong kind of signal, preserving distinctions that the task had made irrelevant.
Human cocaine-addiction data are less mechanistically precise, but they rhyme with the animal work. Ersche et al. found that “carrots and sticks” failed to change behavior in cocaine addiction, a human version of poor updating despite feedback.8
The rat OFC recordings cannot prove the same cellular mechanism in people, but they make a plausible bridge: impaired hidden-state representation could help explain why feedback, consequences, and treatment lessons fail to generalize across contexts.
What Rat OFC Data Can and Cannot Say About Relapse
The strongest inference is computational. Prior cocaine use altered OFC representations in a way that made task-equivalent positions look less equivalent.
That supports a model in which cocaine exposure weakens the brain’s ability to generalize across latent states, especially when surface cues differ.
The weaker inference is clinical. A 6-rat male animal study cannot establish how permanent OFC dysfunction is in humans, whether the same effect appears in females, or whether any specific therapy repairs hidden-state coding.
The study itself flags the small animal count, the active sucrose control, and the possibility that motivational state or attention contributed to the pattern.1
Treatment implication: relapse prevention probably needs more than teaching rules in 1 safe setting. If cocaine exposure makes generalization harder, then treatment should repeatedly train coping and outcome-updating across varied settings, cues, and emotional states.
That implication is not a proven intervention from the Zong study; it is the practical direction suggested by the mechanism and by adjacent OFC-flexibility work.
The most direct boundary is this: cocaine-experienced rats could still perform the task, but their OFC represented the task less abstractly. Addiction, on this model, is more than stronger reward drive.
It is also weaker cognitive mapping across contexts where the surface cues change but the relevant consequences stay the same.
Questions About Cocaine, Hidden States, and OFC Generalization
Did cocaine make the rats unable to do the odor task?
No. Both groups performed at high discrimination levels after extensive training.
The cocaine effect appeared in behavioral variability and OFC representation: the cocaine-experienced group treated S1 and S2 positions as more distinct than controls did.
Is this just another way of saying cocaine increased impulsivity?
No. Impulsivity is too broad for the result.
The paper tested hidden-state identification, meaning the ability to compress different sensory situations into the same underlying task state when their behavioral meaning is equivalent.
Does this prove cocaine permanently damages the human OFC?
No. The study involved 6 male rats after a defined self-administration protocol, not human patients followed over years.
It supports a persistent post-exposure OFC computation change in this animal model; human permanence and recovery require different evidence.
Why use sucrose controls instead of no-exposure controls?
Sucrose controls matched the basic self-administration experience without cocaine. That makes the comparison more relevant than a passive no-exposure group, but it also means sucrose is not a neutral manipulation; extra instrumental experience with a reward could itself shape OFC abstraction.
References
- Zong W, Mueller L, Zhang Z, Zhou J, Schoenbaum G. Prior cocaine use disrupts identification of hidden states by single units and neural ensembles in orbitofrontal cortex. eLife. 2026;15:RP109883. doi:10.7554/eLife.109883
- Schuck NW, Cai MB, Wilson RC, Niv Y. Human orbitofrontal cortex represents a cognitive map of state space. Neuron. 2016;91(6):1402-1412. doi:10.1016/j.neuron.2016.08.019
- Zhou J, Zong W, Jia C, Gardner MPH, Schoenbaum G. Prospective representations in rat orbitofrontal ensembles. Behavioral Neuroscience. 2021;135(4):518-527. doi:10.1037/bne0000451
- Calu DJ, Stalnaker TA, Franz TM, Singh T, Shaham Y, Schoenbaum G. Withdrawal from cocaine self-administration produces long-lasting deficits in orbitofrontal-dependent reversal learning in rats. Learning & Memory. 2007;14(5):325-328. doi:10.1101/lm.534807
- Schoenbaum G, Setlow B. Cocaine makes actions insensitive to outcomes but not extinction: implications for altered orbitofrontal-amygdalar function. Cerebral Cortex. 2005;15(8):1162-1169. doi:10.1093/cercor/bhh216
- Lucantonio F, Takahashi YK, Hoffman AF, Chang CY, Bali-Chaudhary S, Shaham Y, Lupica CR, Schoenbaum G. Orbitofrontal activation restores insight lost after cocaine use. Nature Neuroscience. 2014;17(8):1092-1099. doi:10.1038/nn.3763
- Mueller LE, Konya C, Sharpe MJ, Wikenheiser AM, Schoenbaum G. Prior cocaine use diminishes encoding of latent information by orbitofrontal, but not medial, prefrontal ensembles. Current Biology. 2024;34(22):5223-5238. doi:10.1016/j.cub.2024.09.064
- Ersche KD, Gillan CM, Jones PS, Williams GB, Ward LHE, Luijten M, de Wit S, Sahakian BJ, Bullmore ET, Robbins TW. Carrots and sticks fail to change behavior in cocaine addiction. Science. 2016;352(6292):1468-1471. doi:10.1126/science.aaf3700