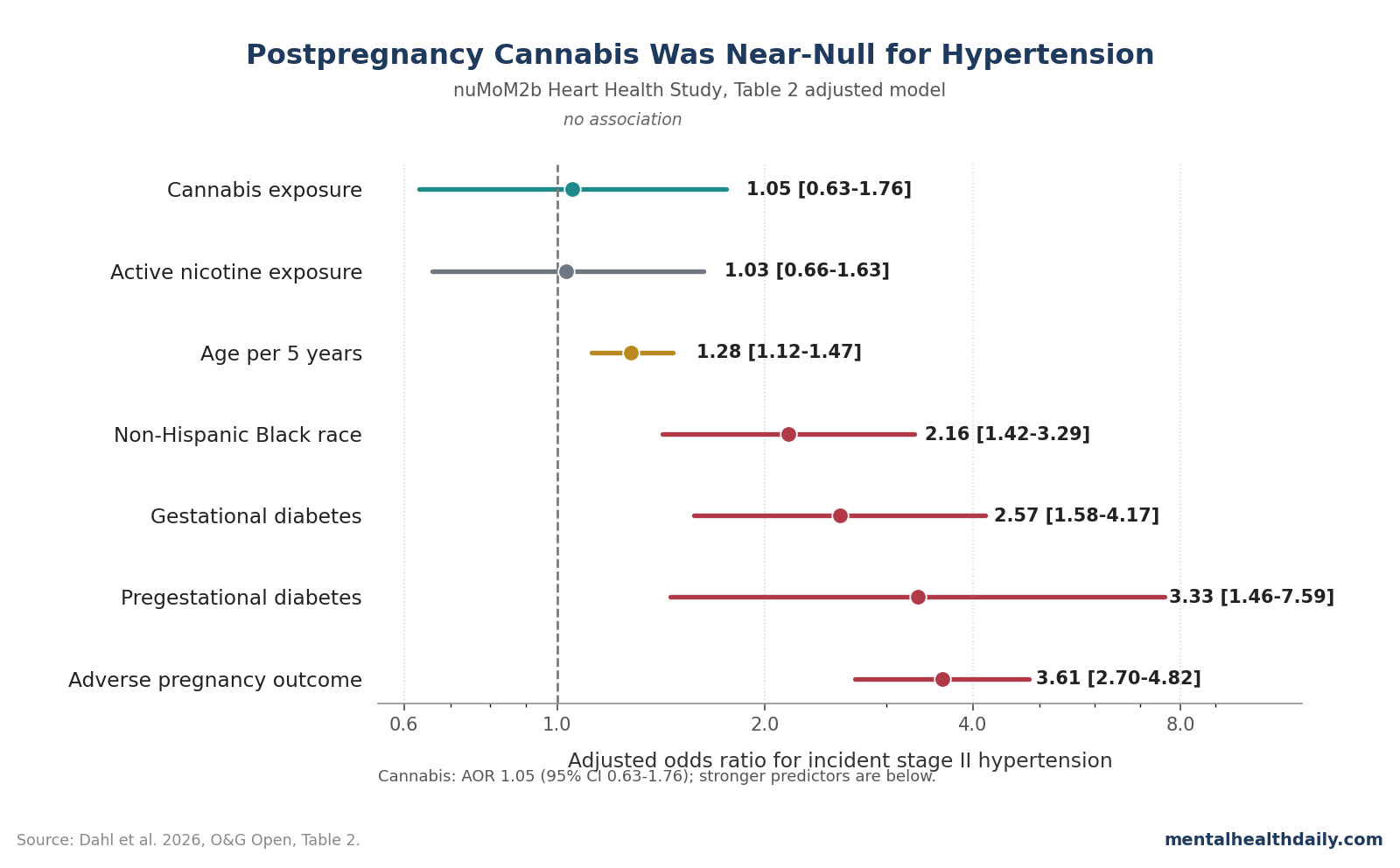
A 2026 nuMoM2b Heart Health Study analysis tested whether urine-confirmed cannabis exposure 2–7 years after a first pregnancy predicted incident stage II hypertension; it did not: adjusted odds ratio (AOR) 1.05, 95% CI 0.63–1.76, with 216 of 4,079 participants developing hypertension.1
Research Highlights
- The headline result was null: cannabis exposure at the 2–7 year postpartum visit was not associated with incident stage II hypertension after adjustment (AOR 1.05, 95% CI 0.63–1.76).1
- Biologic testing found more exposure than self-report: 406 of 4,079 participants (10.0%) were urine-positive for THC-COOH, but only 152 of those 406 (37.4%) self-reported THC exposure.1
- Pregnancy and metabolic risk factors were much larger than cannabis exposure: adverse pregnancy outcomes had AOR 3.61, pregestational diabetes AOR 3.33, gestational diabetes AOR 2.57, and non-Hispanic Black race AOR 2.16 for incident hypertension.1
- The wider hypertension literature stays mixed: Dahl et al. cited a 2025 cardiovascular meta-analysis on event risk, but also 2021–2023 hypertension studies reporting no association or inverse associations.4,5,6,7,8
- The counseling point is calibration, not reassurance: the study measured exposure at 1 visit 2–7 years postpartum and did not measure dose, potency, route, duration, or timing across the postpartum years.1
The 2026 analysis is easy to overread because cannabis has plausible cardiovascular effects and postpartum cannabis use is a real counseling issue. Dahl et al. still reported a null result for the measured hypertension endpoint, not an elevated-risk or dose-response finding.
THC-COOH is the main inactive urine metabolite of THC, the intoxicating cannabinoid in cannabis. In this study, a positive urine result meant THC-COOH of at least 15 ng/mL by confirmatory liquid chromatography tandem mass spectrometry, a laboratory method that separates and measures specific chemicals rather than relying on self-report alone.1
Urine-Positive Cannabis Exposure Was 10.0%, but the Hypertension AOR Was 1.05
Dahl et al. analyzed 4,079 participants from the nuMoM2b Heart Health Study, a follow-up of nulliparous women after a first pregnancy. Participants had attended an in-person visit 2–7 years after the index pregnancy, had no preexisting hypertension, and had enough data for cannabis exposure, Area Deprivation Index, and hypertension status.1
The primary exposure was biologically measured cannabis exposure at that follow-up visit. The primary outcome was incident stage II hypertension, defined as systolic blood pressure at least 140 mm Hg, diastolic blood pressure at least 90 mm Hg, or antihypertensive-agent use at the 2–7 year visit.
The core model adjusted for nicotine exposure, adverse pregnancy outcome, race and ethnicity, Area Deprivation Index, maternal age, and diabetes. After that adjustment, cannabis exposure was not associated with incident stage II hypertension: AOR 1.05, 95% CI 0.63–1.76.
Active nicotine exposure was similarly flat in the model, with AOR 1.03, 95% CI 0.66–1.63.1
Plain English: an AOR of 1.05 is very close to 1.00, the no-difference line. The confidence interval was wide enough to include lower odds, no meaningful difference, and moderately higher odds, so the study did not provide a statistically confirmed cannabis-hypertension signal.
Adverse Pregnancy Outcomes and Diabetes Dwarfed the Cannabis Estimate
The adjusted model contrast matters because it shows what the analysis could detect. Several established postpartum cardiovascular risk markers were strongly associated with incident stage II hypertension 2–7 years after the first pregnancy.
Adverse pregnancy outcomes had the largest paper-relevant signal: AOR 3.61, 95% CI 2.70–4.82. In Dahl et al., adverse pregnancy outcome included hypertensive disorders of pregnancy, small-for-gestational-age birth weight below the 5th percentile, preterm birth, or stillbirth.1
Diabetes also carried large adjusted associations. Pregestational diabetes had AOR 3.33, 95% CI 1.46–7.59, and gestational diabetes had AOR 2.57, 95% CI 1.58–4.17. Maternal age also mattered, with AOR 1.28 per 5-year increase, 95% CI 1.12–1.47.
Race and ethnicity reflected the usual structural-risk pattern. Non-Hispanic Black participants had AOR 2.16, 95% CI 1.42–3.29, and Hispanic participants had AOR 1.51, 95% CI 1.00–2.29, compared with non-Hispanic White participants. That does not make race a biological cause; it marks how measured and unmeasured social, medical-access, neighborhood, stress, and cardiometabolic exposures cluster in U.S. cohorts.
The Null Result Pushes Against a Simple Cannabis-Raises-Blood-Pressure Story
The study was not designed as a cannabis safety endorsement. Cannabis has acute cardiovascular effects, including increased heart rate and sometimes blood-pressure changes, and Dahl et al. cited a 2025 systematic review and meta-analysis that connected cannabis and cannabinoids with major adverse cardiovascular events such as cardiovascular death, acute coronary syndrome, myocardial infarction, and nonfatal stroke.4
But the hypertension-specific evidence is less clean. Dahl et al. explicitly described prior research as inconsistent: some reports link cannabis with acute sympathetic activation or intoxication-related hypertension, while other reports show no association or even inverse associations between cannabis exposure and hypertension or blood-pressure levels.5,6,7,8
That is the calibrated read. Cannabis may still affect cardiovascular risk through mechanisms not captured by a single stage II hypertension endpoint.
Dahl et al. noted possible pathways involving lipid-receptor dysregulation and inflammatory signaling, and other cannabis-cardiovascular papers focus on events rather than chronic blood-pressure diagnosis.1,4
For this paper, though, the measured question was narrower: urine-positive cannabis exposure at one postpartum follow-up visit did not predict stage II hypertension once nicotine exposure and known pregnancy/cardiometabolic risk factors were in the model.
Self-Report Missed Most Urine-Positive Cannabis Exposure
The most useful positive finding may be about measurement. Of 406 participants with urine-confirmed THC-COOH, only 152 self-reported THC exposure, which means self-report captured 37.4% of biologically detected exposure.1
That gap fits older pregnancy and postpartum cannabis research. Cannabis disclosure can be shaped by stigma, legal concerns, child-protective-system fears, uncertainty about product contents, and ordinary recall problems.
Brown et al. showed rising cannabis use among pregnant and nonpregnant reproductive-aged women from 2002 to 2014, and Skelton et al. linked recreational legalization with maternal cannabis use in preconception, prenatal, and postpartum periods.2,3
For mental-health readers, the self-report gap matters because postpartum cannabis is often used in the same symptom space as psychiatric care: insomnia, anxiety, pain, low appetite, stress, and the grind of early parenting. Bayrampour et al. found that women discuss cannabis during pregnancy and postpartum through a risk-benefit lens rather than as a purely recreational behavior.12
A blood-pressure null does not erase those behavioral-health questions. It does, however, block the stronger claim that postpregnancy cannabis exposure independently predicted hypertension in this cohort.
nuMoM2b Already Shows Why Postpartum Blood Pressure Needs Follow-Up
Dahl et al. sits inside a larger nuMoM2b Heart Health evidence base. Haas et al. previously used the same broad research platform to show that adverse pregnancy outcomes were associated with hypertension 2–7 years postpartum.9
That context explains why adverse pregnancy outcome was such a strong covariate in the cannabis paper. Hypertensive disorders of pregnancy, preterm birth, fetal-growth restriction, and stillbirth are more than obstetric history; they are early cardiometabolic warning signals.
When Dahl et al. adjusted for adverse pregnancy outcome, diabetes, age, race and ethnicity, and neighborhood disadvantage, cannabis did not add a detectable independent hypertension signal.
This also separates the postpregnancy question from pregnancy-exposure papers. Metz et al. reported cannabis exposure and adverse pregnancy outcomes related to placental function in JAMA, but that is a different endpoint: pregnancy and placental outcomes, not maternal stage II hypertension years after delivery.10
Keeping those endpoints separate prevents two opposite errors. One error is minimizing cannabis because this postpartum hypertension study was null.
The other error is importing pregnancy-outcome concerns into a postpartum blood-pressure model and claiming Dahl et al. found a risk that the paper did not find.
The Main Limits Are Timing, Dose, and Endpoint Choice
Timing: cannabis exposure and hypertension status were assessed at the same 2–7 year visit. That cross-sectional structure cannot establish whether cannabis exposure came before elevated blood pressure, followed it, or tracked some third factor such as sleep disruption, anxiety, pain, neighborhood stress, or other substance exposure.
Dose and product detail: urine THC-COOH is a strong improvement over self-report, but it does not tell readers whether someone used a low-dose edible once, smoked high-THC flower daily, used concentrates, used CBD-dominant products, or changed use patterns across the postpartum years. Dahl et al. specifically noted the lack of data on frequency, duration, and amount of cannabis use.1
Exposure window: THC-COOH can be detected for several days after occasional use and for several weeks after chronic use. Participants with occasional use could still be misclassified as unexposed if the follow-up visit missed their detection window.
Endpoint choice: the primary outcome was stage II hypertension or antihypertensive-agent use. A sensitivity analysis using combined stage I and stage II hypertension also remained nonsignificant, which strengthens the null hypertension read, but it still leaves non-hypertension cardiovascular pathways outside the main result.
Interaction testing: cannabis use did not significantly modify the relationship between adverse pregnancy outcomes and incident hypertension, and it did not significantly interact with Area Deprivation Index. The reported interaction P values were 0.64 and 0.79, so the paper did not show a subgroup in which cannabis amplified those risk pathways.1
Postpartum Counseling Should Ask About Cannabis Without Inventing a Hypertension Signal
Good counseling can hold the null result and the caution at the same time. A postpartum patient using cannabis for sleep or anxiety should not be told that Dahl et al. proved cannabis causes hypertension, because it did not.
The better conversation is that cannabis exposure is common, often underreported, and still uncertain for several cardiovascular endpoints.
For blood pressure specifically, Dahl et al. points attention back to the factors that clearly stood out: prior adverse pregnancy outcomes, diabetes, age, and the structural-risk patterns captured by race, ethnicity, and neighborhood disadvantage. Those are reasons to measure blood pressure, connect postpartum care to primary care, and avoid letting the 6-week obstetric visit become the end of cardiovascular surveillance.
For mental health, the practical issue is substitution. If cannabis is being used because postpartum insomnia, pain, anxiety, or depressive symptoms are undertreated, the alternative is not a lecture.
The alternative is better symptom care, safer sleep support, mood-disorder screening that leads somewhere, and direct discussion of what cannabis can and cannot be inferred to do from the available evidence.
Questions About Cannabis After Pregnancy and Hypertension
Did Dahl et al. find that postpregnancy cannabis caused hypertension?
No. The adjusted estimate was AOR 1.05 with 95% CI 0.63–1.76, which is not a statistically confirmed association and is very close to the no-difference value of 1.00.1
Was the cannabis result close to significant?
No. The 95% CI crossed 1.00 widely.
The estimate is compatible with lower odds, nearly no difference, or higher odds, so the correct read is a null hypertension result with limited precision, not a hidden dose-response signal.
Why discuss cardiovascular risk if the nuMoM2b hypertension result was null?
Because hypertension is only 1 cardiovascular endpoint. Dahl et al. cited broader cannabis-cardiovascular literature on acute effects and event risk, including a 2025 meta-analysis, but their own postpartum hypertension model did not show an independent association.4
Did urine testing change the interpretation?
Yes. Urine testing made exposure measurement stronger than self-report alone: 406 participants were urine-positive, while only 152 of those 406 self-reported THC exposure.
That improves detection but still does not measure dose, frequency, potency, or route.1
Does this null result change advice about cannabis during pregnancy?
No. Dahl et al. studied maternal hypertension 2–7 years after a first pregnancy, not fetal development or placental-function outcomes during pregnancy. Pregnancy-exposure studies answer a different question.10
What should postpartum care do with this finding?
Ask about cannabis use directly, measure blood pressure, and prioritize the larger demonstrated risk markers: adverse pregnancy outcomes, diabetes, age, and structural barriers to cardiovascular follow-up. The cannabis discussion should be honest about uncertainty instead of inflating a null study into a risk claim.
References
- Dahl CM, Chahine R, Wiener LE, et al. Cannabis exposure after pregnancy and development of hypertension in the nuMoM2b Heart Health Study. O&G Open. 2026;3:1–9. doi:10.1097/og9.0000000000000164
- Brown QL, Sarvet AL, Shmulewitz D, Martins SS, Wall MM, Hasin DS. Trends in marijuana use among pregnant and nonpregnant reproductive-aged women, 2002–2014. JAMA. 2017;317:207–209. doi:10.1001/jama.2016.17383
- Skelton KR, Hecht AA, Benjamin-Neelon SE. Association of recreational cannabis legalization with maternal cannabis use in the preconception, prenatal, and postpartum periods. JAMA Network Open. 2021;4:e210138. doi:10.1001/jamanetworkopen.2021.0138
- Storck W, Elbaz M, Vindis C, Déguilhem A, Lapeyre-Mestre M, Jouanjus E. Cardiovascular risk associated with the use of cannabis and cannabinoids: a systematic review and meta-analysis. Heart. 2025;111:1047–1056. doi:10.1136/heartjnl-2024-325429
- Haleem A, Hwang YJ, Elton-Marshall T, Rehm J, Imtiaz S. The longitudinal relationship between cannabis use and hypertension. Drug and Alcohol Review. 2021;40:914–919. doi:10.1111/dar.13266
- Corroon J, Grant I, Bradley R, Allison MA. Trends in cannabis use, blood pressure, and hypertension in middle-aged adults: findings from NHANES, 2009–2018. American Journal of Hypertension. 2023;36:651–659. doi:10.1093/ajh/hpad068
- Shah RM, Patel S, Shah S, Doshi S, Li A, Diamond JA. Sustained cannabis use does not predispose clinical hypertension: findings from a national survey. Journal of Clinical Hypertension. 2023;25:47–52. doi:10.1111/jch.14623
- Vallée A. Association between cannabis use and blood pressure levels according to comorbidities and socioeconomic status. Scientific Reports. 2023;13:2069. doi:10.1038/s41598-022-22841-6
- Haas DM, Parker CB, Marsh DJ, Grobman WA, Ehrenthal DB, Greenland P, et al. Association of adverse pregnancy outcomes with hypertension 2 to 7 years postpartum. Journal of the American Heart Association. 2019;8:e013092. doi:10.1161/jaha.119.013092
- Metz TD, Allshouse AA, McMillin GA, Greene T, Chung JH, Grobman WA, et al. Cannabis exposure and adverse pregnancy outcomes related to placental function. JAMA. 2023;330:2191–2199. doi:10.1001/jama.2023.21146
- Ewing AC, Schauer GL, Grant-Lenzy AM, Njai R, Coy KC, Ko JY. Current marijuana use among women of reproductive age. Drug and Alcohol Dependence. 2020;214:108161. doi:10.1016/j.drugalcdep.2020.108161
- Bayrampour H, Zahradnik M, Lisonkova S, Janssen P. Women’s perspectives about cannabis use during pregnancy and the postpartum period. Preventive Medicine. 2019;119:17–23. doi:10.1016/j.ypmed.2018.12.002