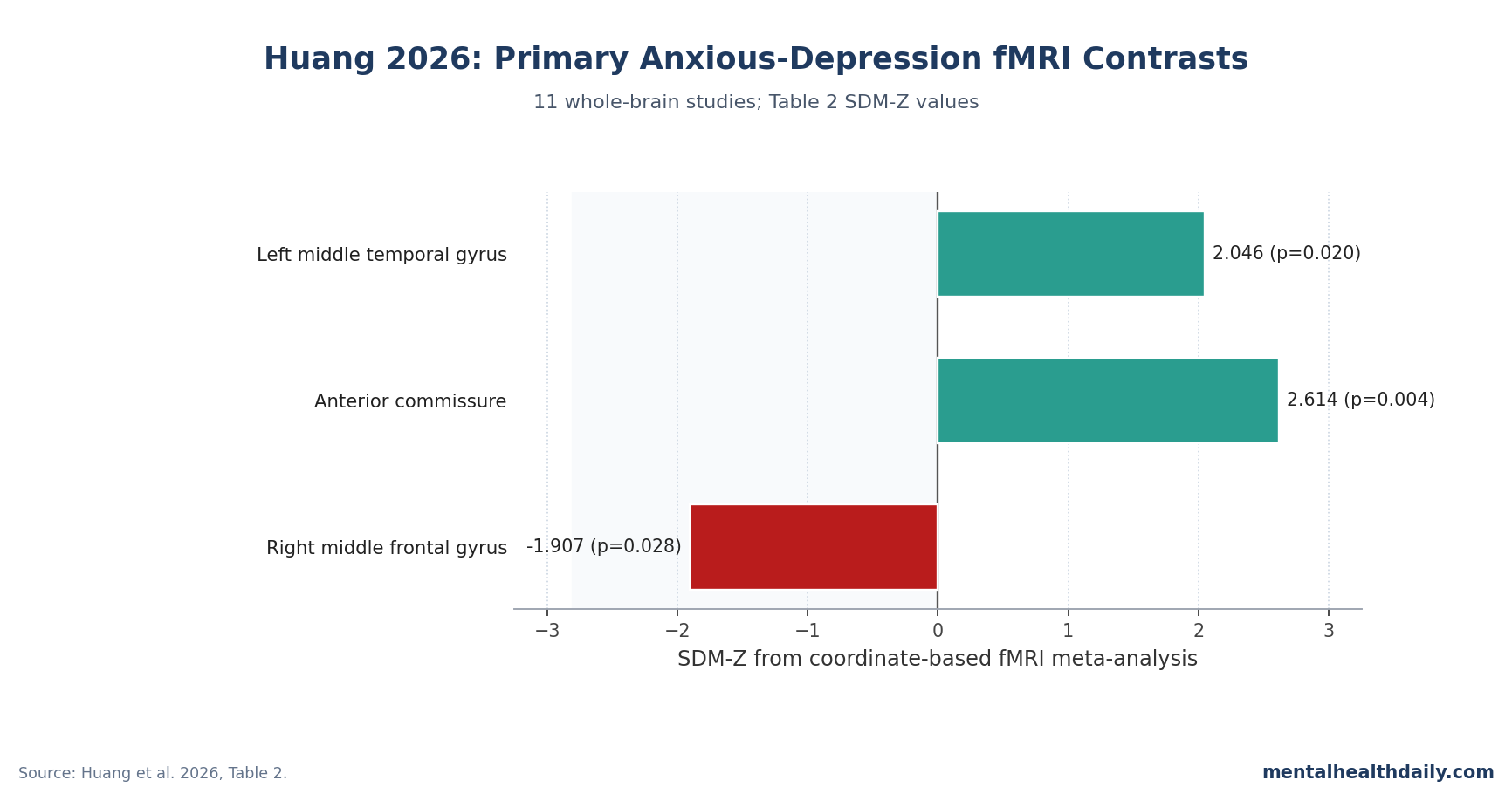
A 2026 fMRI meta-analysis of 11 studies found a narrow anxious-depression signal: 829 anxious-depression patients differed from 681 non-anxious MDD patients in left middle temporal gyrus activation (SDM-Z = 2.046, p = 0.020), while healthy-control contrasts centered on the anterior commissure and right middle frontal gyrus. The calibrated interpretation is not “amygdala and insula hyperactivity proves a fear-circuit subtype”; it is that anxious depression has a reproducible group-level imaging signal, specific and still far from a clinical scan test.1
Research Highlights
- The direct subtype contrast was left temporal: Anxious MDD exceeded non-anxious MDD in the left middle temporal gyrus at MNI −52, −42, 8 (SDM-Z = 2.046, p = 0.020; 359 voxels).1
- Healthy-control contrasts split by direction: Anxious MDD showed higher anterior commissure signal (SDM-Z = 2.614, p = 0.004; 179 voxels) and lower right middle frontal gyrus signal (SDM-Z = −1.907, p = 0.028; 56 voxels).1
- Female-stratified findings were control-relative: Meta-regression linked female aMDD samples with higher left cingulate/anterior cingulate activity (SDM-Z = 2.107, p = 0.018; 539 voxels), higher right anterior thalamic projection signal (p = 0.034), and lower left rolandic operculum signal (p = 0.049).1
- The evidence base was small but not trivial: Huang et al. searched 6523 citations through 19 December 2024 and retained 11 whole-brain fMRI studies covering 2375 participants.1
- Adjacent studies argue against a single-circuit slogan: Prior fMRI work found frontoparietal-network, triple-network, insula/frontal, and subcortical findings across 2015–2024 studies rather than one clean amygdala-only marker.6789
Anxious depression means major depressive disorder with prominent anxiety symptoms, usually measured through an anxious-distress specifier, comorbid anxiety diagnosis, or high anxiety/somatization scores inside a depressive episode. The construct matters clinically because STAR*D found worse antidepressant outcomes in anxious vs. non-anxious depression, and DSM-5 anxious-distress validation work also tied the specifier to greater illness burden.23
The imaging question is more delicate. A patient can have worse clinical outcomes without a clean scan-based subtype, and a group-level brain signal can be statistically real without being useful for individual diagnosis.
11 Whole-Brain fMRI Studies Narrowed to 3 Main Group Signals
Huang et al. searched PubMed, Medline, Embase, and Cochrane for functional MRI studies of anxious depression through 19 December 2024, then filtered 6523 citations down to 11 eligible whole-brain studies.1 The final dataset included 829 anxious MDD patients, 681 non-anxious MDD patients, and 865 healthy controls.
Seed-based d Mapping with Permutation of Subject Images (SDM-PSI) is a coordinate-based neuroimaging meta-analysis method: instead of reanalyzing each participant’s raw fMRI scan, it reconstructs likely regional effects from the peak coordinates and statistics that each paper reported.4 That makes cross-study synthesis possible, but it also means the output is constrained by what the original studies chose to report.
The paper tested 3 main contrasts:
- Anxious MDD vs. non-anxious MDD.
- Anxious MDD vs. healthy controls.
- Non-anxious MDD vs. healthy controls, using the 7 studies that reported the necessary control comparison.
Only the first 2 contrasts produced source-verified headline findings. The non-anxious MDD vs. healthy-control reanalysis did not find significant group differences, which is part of why the anxious-depression result should not be flattened into a generic “all depression has the same fMRI signature” story.
Left Middle Temporal Gyrus Was the Only aMDD vs. MDD Difference
the most direct subtype result was not amygdala, insula, or anterior cingulate. Anxious MDD showed greater functional alteration than non-anxious MDD in the left middle temporal gyrus (MNI −52, −42, 8; SDM-Z = 2.046; p = 0.020; 359 voxels).1
The middle temporal gyrus is involved in language-linked semantic processing, audiovisual integration, attention, and memory-related default-mode activity. In this context, the signal is best read as evidence that anxious depression is more than ordinary MDD plus subjective worry; at the group level, the anxious subtype showed a different temporal-lobe functional pattern.
That result also lines up with older adjacent work. Qi et al. reported gray-matter differences in depressive patients with and without anxiety disorders, and Huang et al. specifically cite Qi’s left middle temporal gyrus finding as consistent with the 2026 meta-analytic result.5
Hou et al. later added a large 600-person subcortical/cingulate/hippocampal ALFF dataset in anxious and depressed individuals, showing that the field has been circling around anxiety-linked neural activity rather than one simple depression-only pathway.9
Anterior Commissure Increased and Right Middle Frontal Gyrus Decreased vs. Controls
Against healthy controls, anxious MDD had 2 significant results with opposite directions. The anterior commissure showed higher functional alteration (MNI 2, 2, 0; SDM-Z = 2.614; p = 0.004; 179 voxels), while the right middle frontal gyrus showed lower functional alteration (MNI 34, 46, 24; SDM-Z = −1.907; p = 0.028; 56 voxels).1
Anterior commissure is a white-matter tract that links temporal and limbic regions across the 2 hemispheres, including circuitry relevant to emotional learning and social behavior. Huang et al. interpreted its higher signal partly through amygdala-linked emotional circuitry, but the measured meta-analytic cluster was anterior commissure, not an amygdala coordinate.
Right middle frontal gyrus overlaps with dorsolateral prefrontal cortex territory, a region involved in working memory, attention control, and top-down regulation of emotion. A lower signal there fits a plausible model in which anxious depression combines heightened salience or threat-linked processing with weaker regulatory control, but that is an interpretation layered on top of a coordinate result, not a direct treatment rule.
Female-Stratified Results Were Visible vs. Controls, Not vs. Non-Anxious MDD
The sex analysis needs careful wording because it is easy to overstate. Huang et al. found female-stratified signals when anxious MDD was compared with healthy controls, but the aMDD vs. non-anxious MDD regression did not yield statistically significant sex-stratified results.1
The female aMDD vs. control meta-regression found higher activity in the left cingulate/anterior cingulate region (SDM-Z = 2.107, p = 0.018; 539 voxels), right anterior thalamic projections (SDM-Z = 1.826, p = 0.034; 12 voxels), and right superior longitudinal fasciculus (SDM-Z = 2.010, p = 0.022; 10 voxels). It also found lower signal in the left rolandic operculum (SDM-Z = −1.658, p = 0.049; 3 voxels).1
Those signals are biologically plausible because cingulate, thalamic, and frontal-language/sensorimotor regions all sit near emotion-regulation or cognitive-control systems. They are not proof that women with anxious depression need a different first-line treatment based on fMRI.
The safer implication is that future imaging and treatment trials should avoid burying sex-stratified effects inside mixed samples.
Adjacent fMRI Studies Point to Networks, Not a Single Fear-Circuit Biomarker
The older draft turned anxious depression into a neat fear-circuit story. The neighboring literature is messier and more useful than that.
Pannekoek et al. found resting-state frontoparietal-network connectivity differences in comorbid depression/anxiety, including clusters involving anterior cingulate, insula, angular gyrus, right middle frontal gyrus, and inferior parietal regions.6 Beckmann et al. later focused on triple-network connectivity in MDD with comorbid anxiety, again pointing toward distributed network dysfunction rather than a single region.7
Sindermann et al. used an emotion-processing task and reported bilateral insula plus medial/inferior frontal activation differences in depression with comorbid anxiety disorder.8 that finding supports the interpretation because it supports the broader anxiety-processing picture, but it also explains why the Huang meta-analysis should be read carefully: task-based insula findings in individual studies are not the same thing as Huang’s direct aMDD vs. MDD coordinate-based headline result.
The synthesis is therefore calibrated. Anxious depression probably has brain-level features that differ from non-anxious depression.
The most source-verified Huang result is left middle temporal gyrus activation vs. non-anxious MDD, with anterior commissure and right middle frontal gyrus differences vs. controls. A single amygdala/insula headline is too clean for the evidence.
Treatment Implications Start With Stratified Trials, Not Scan-Based Matching
The clinical literature already treats anxious depression as harder to manage. Fava et al. found different STAR*D outcomes in anxious vs. non-anxious depression, and Zimmerman et al. supported the validity of the DSM-5 anxious-distress specifier as a clinically meaningful marker rather than a decorative label.23
The imaging literature adds a mechanistic reason to stratify trials. If anxious depression has a different temporal/frontal/cingulate profile, then antidepressant, psychotherapy, and neuromodulation studies that pool anxious and non-anxious depression may dilute subtype-specific effects.
One example is transcranial magnetic stimulation. Hutton et al. reported anxiolytic and antidepressant effects of TMS in anxious depression, and Huang et al. discuss DLPFC-targeted repetitive TMS as one clinical direction because the right middle frontal gyrus/DLPFC region is part of the regulatory-control story.10
That does not establish the Huang scan result selects TMS candidates today. It means future TMS studies should report anxious-depression status, anxiety severity, sex-stratified results, and imaging-linked response patterns instead of treating all MDD as one bucket.
Whole-Brain Coordinate Meta-Analysis Still Misses ROI-Only Evidence
Huang et al. excluded region-of-interest studies because ROI methods preselect areas such as amygdala or insula and can violate the equal-likelihood assumption used in whole-brain coordinate meta-analysis.1 That decision makes the meta-analysis cleaner for whole-brain convergence, but it also means clinically interesting ROI-only findings are outside the formal pooled result.
Other limits are just as important. The paper used fMRI only, not a combined structural-plus-functional model.
It included adult participants and could not run age or education subgroup analyses because the underlying studies did not report enough detail. Definitions of anxious depression also varied across studies, ranging from anxious-distress ratings to comorbid anxiety diagnoses.
Those limits do not erase the result. They keep the result in the right lane: a group-level coordinate-based synthesis that supports anxious depression as a biologically non-identical MDD presentation, not a diagnostic biomarker ready for individual patients.
Questions About the Huang 2026 fMRI Meta-Analysis
Did Huang et al. find an amygdala or insula headline effect?
No. The source-verified headline aMDD vs. non-anxious MDD contrast was left middle temporal gyrus activation (SDM-Z = 2.046, p = 0.020), while the aMDD vs. healthy-control findings were anterior commissure hyperactivation and right middle frontal gyrus suppression.1
Does the left middle temporal gyrus result diagnose anxious depression?
No. The result comes from a group-level coordinate meta-analysis, so it can support subtype biology without giving clinicians a scan threshold for diagnosing 1 person.
Why did the researchers exclude ROI-only fMRI studies?
ROI studies start with preselected regions, while Huang et al. were trying to test whole-brain convergence across reported significant peaks. That strengthens whole-brain comparability but leaves some amygdala- and insula-focused evidence outside the pooled analysis.1
How should the female meta-regression be read?
Read it as a control-relative signal, not as proof of a sex-specific anxious-depression diagnostic test. Female aMDD samples differed from healthy controls in cingulate, thalamic, superior longitudinal fasciculus, and rolandic-operculum regions, but the aMDD vs. MDD sex-stratified regression was not significant.1
Does this change treatment choices for anxious depression now?
Not directly. The better immediate use is trial design: anxious-depression status should be reported and stratified in antidepressant, psychotherapy, and TMS studies, especially when outcomes might depend on temporal-frontal-cingulate circuitry.210
References
- Huang H, Guo Z, Yang X, Qin L. Functional brain alterations in anxious depression: insights from whole-brain fMRI and meta-analysis. Dialogues in Clinical Neuroscience. 2026;28(1):32–45. doi:10.1080/19585969.2026.2612918
- Fava M, Rush AJ, Alpert JE, et al. Difference in treatment outcome in outpatients with anxious versus nonanxious depression: a STAR*D report. American Journal of Psychiatry. 2008;165(3):342–351. doi:10.1176/appi.ajp.2007.06111868
- Zimmerman M, Clark HL, McGonigal P, Harris L, Holst CG, Martin J. Validity of the DSM-5 anxious distress specifier for major depressive disorder. Depression and Anxiety. 2019;36(1):31–38. doi:10.1002/da.22837
- Radua J, Mataix-Cols D, Phillips ML, et al. A new meta-analytic method for neuroimaging studies that combines reported peak coordinates and statistical parametric maps. European Psychiatry. 2012;27(8):605–611. doi:10.1016/j.eurpsy.2011.04.001
- Qi H, Ning Y, Li J, et al. Gray matter volume abnormalities in depressive patients with and without anxiety disorders. Medicine. 2014;93(29):e345. doi:10.1097/MD.0000000000000345
- Pannekoek JN, van der Werff SJA, van Tol MJ, et al. Investigating distinct and common abnormalities of resting-state functional connectivity in depression, anxiety, and their comorbid states. European Neuropsychopharmacology. 2015;25(11):1933–1942. doi:10.1016/j.euroneuro.2015.08.002
- Beckmann FE, et al. Specific alterations of resting-state functional connectivity in the triple network related to comorbid anxiety in major depressive disorder. European Journal of Neuroscience. 2024;59(7):1819–1832. doi:10.1111/ejn.16249
- Sindermann L, et al. Emotion processing in depression with and without comorbid anxiety disorder. Journal of Affective Disorders. 2022;314:133–142. doi:10.1016/j.jad.2022.06.089
- Hou J, Liu S, van Wingen G. Increased subcortical brain activity in anxious but not depressed individuals. Journal of Psychiatric Research. 2023;160:38–46. doi:10.1016/j.jpsychires.2023.02.013
- Hutton TM, et al. The anxiolytic and antidepressant effects of transcranial magnetic stimulation in patients with anxious depression. Journal of Clinical Psychiatry. 2023;84(1):45017. doi:10.4088/JCP.22m14571