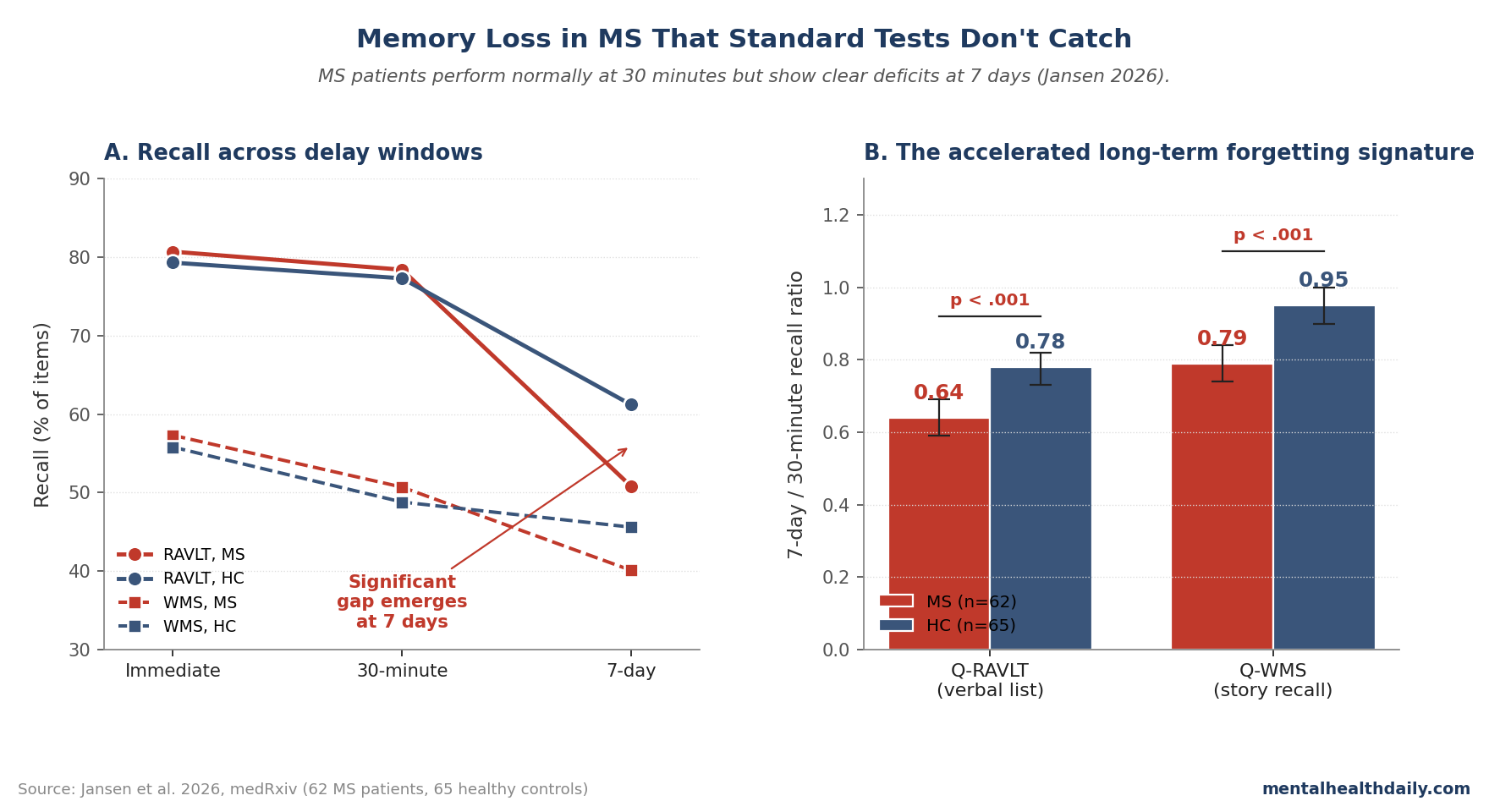
Standard memory testing can tell a multiple-sclerosis (MS) patient that recall is normal after 30 minutes while missing a deficit that appears days later. Jansen et al. tested that exact blind spot in 62 MS patients and 65 matched controls: 7-day/30-minute recall ratios were lower in MS on both verbal-list recall (0.64 vs. 0.78, p < 0.001) and story recall (0.79 vs. 0.95, p < 0.001).1
Research Highlights
- 7-day recall exposed the deficit: MS patients had lower QRAVLT scores than controls (0.64, 95% CI 0.59–0.69 vs. 0.78, 0.73–0.82; p < 0.001) and lower QWMS scores (0.79, 0.74–0.84 vs. 0.95, 0.90–1.00; p < 0.001).1
- Short-delay testing looked normal: RAVLT recall was 80.75% vs. 79.28% immediately (p = 0.924) and 78.39% vs. 77.33% at 30 minutes (p = 0.923), so the group difference emerged only after the long delay.1
- Subjective complaints had an objective correlate: 22 of 62 MS patients reported increased forgetfulness vs. 7 of 65 controls (p = 0.002), and the combined ALF score discriminated subjective memory impairment with AUC 0.735.1
- The result extends earlier MS data: Stalter et al. had reported accelerated long-term forgetting in 28 mildly impaired MS patients; the 2026 study replicated the verbal-memory signal in a larger and more heterogeneous sample than the 28-patient earlier study.10
- The clinical limit is important: the 2026 study supports 7-day verbal recall as a promising marker, not a ready-made diagnostic test; its ROC cut-off was 0.71 in one sample and needs external validation.1
Accelerated long-term forgetting (ALF) means normal learning and short-delay recall followed by unusually steep loss over a longer interval, often 7 days. That pattern matters in MS because subjective memory complaints are common, but routine cognitive batteries usually stop at immediate or 30-minute recall.2
The Jansen study did not show that every MS memory complaint is explained by ALF. It showed something narrower and more useful: in a carefully selected MS sample without major depression or global cognitive impairment, extending verbal recall to 7 days found a memory-retention deficit that standard short-delay testing missed.
62 MS Patients and 65 Controls Were Tested Across 3 Recall Windows
Jansen et al. recruited 76 MS patients and 67 controls in Oldenburg, Germany; after exclusions for incomplete testing, MoCA < 26, BDI-II ≥ 20, or recent CNS-active medication, the analyzed sample included 62 MS patients and 65 healthy controls. The MS group included 52 patients with relapsing-remitting MS, 4 with secondary progressive MS, and 6 with primary progressive MS.
Mean age, sex distribution, and education did not differ significantly between groups.1
The protocol used 2 verbal memory tasks:
- RAVLT: Rey’s Auditory Verbal Learning Test, a 15-word list learned across 5 trials.
- WMS-IV Logical Memory: story recall from the Wechsler Memory Scale, scored by remembered story details.
Both tests were administered immediately, after 30 minutes, and again after 7 days by telephone. The primary outcomes were QRAVLT and QWMS, calculated as 7-day recall divided by 30-minute recall.
A ratio closer to 1.0 means little extra loss after the standard delay; a lower ratio means more forgetting between the 30-minute test and the 7-day test.
30-Minute Recall Was Normal; 7-Day Recall Was Not
Immediate learning did not separate the groups. RAVLT immediate recall was 80.75% in MS vs. 79.28% in controls (p = 0.924), and WMS-IV immediate recall was 57.29% vs. 55.82% (p = 0.457).
Thirty-minute recall also looked normal: RAVLT 78.39% vs. 77.33% (p = 0.923), and WMS-IV 50.74% vs. 48.80% (p = 0.508).1
Seven days later, the pattern changed. MS patients recalled 50.75% of RAVLT items vs. 61.23% in controls (p = 0.004) and 40.06% of WMS-IV story details vs. 45.60% in controls (p = 0.007).
The long-delay ratios made the difference clearer: QRAVLT was 0.64 in MS vs. 0.78 in controls, and QWMS was 0.79 vs. 0.95.
Plain English: the MS group learned the material about as well as controls and still remembered it at 30 minutes, but lost more of it across the following week. That is the ALF signature.
The ALF Score Moderately Detected Subjective Memory Impairment
Subjective memory impairment was more common in the MS group: 22 of 62 MS patients reported increased forgetfulness compared with 7 of 65 controls. MS patients also reported higher forgetfulness on a 0–100 scale (37.26 vs. 29.23, p = 0.003), higher Everyday Memory Questionnaire scores (31.31 vs. 21.63, p = 0.004), and higher FEAG everyday-memory scores (90.65 vs. 79.83, p = 0.001).1
The combined ALF score, defined as the mean of QRAVLT and QWMS, discriminated MS patients with subjective memory impairment from those without it at AUC 0.735 (95% CI 0.585–0.884). The best cut-off in this sample was 0.71, with sensitivity 0.727 and specificity 0.725.
That is moderate accuracy. It is not strong enough to make ALF a stand-alone clinical diagnostic test, but it is strong enough to explain why a patient can experience day-to-day memory failures despite normal 30-minute recall.
The test is measuring a later memory process than the standard battery usually measures.
Why a 7-Day Delay Can Find What Short Tests Miss
Memory does not become stable the moment a person repeats a word list back correctly. Memory consolidation is the slower process by which recently learned information becomes more stable across hours and days, involving interactions between the hippocampus, neocortex, sleep, and distributed brain networks.6
ALF can be read as a consolidation problem: initial encoding works, early retention works, but the memory trace loses stability over the longer interval. In MS, that is biologically plausible because demyelination, white-matter injury, cortical thinning, and disrupted hippocampal connectivity can interfere with the networks that support long-term memory stabilization.12
The pattern is not unique to MS. ALF was first emphasized in temporal lobe epilepsy and has also been reported after traumatic brain injury, transient ischemic attack or minor stroke, limbic encephalitis, and presymptomatic autosomal-dominant Alzheimer’s disease.3,7
The MS-specific contribution of the 2026 paper is the combination of normal short-delay performance, lower 7-day ratios, and a link to subjective memory complaints in a larger sample than the earlier MS pilot.
Standard MS Batteries Usually Stop Before 7 Days
The standard MS cognitive screening tool is the Brief International Cognitive Assessment for Multiple Sclerosis (BICAMS), which includes processing speed, verbal learning, and visuospatial memory tasks.8 Its value is real: processing-speed impairment is one of the most reproducible cognitive findings in MS, and validated batteries are far more useful than casual bedside impressions.
The limitation is narrower. Standard MS batteries usually do not ask whether a patient remembers the same material 7 days later.
Jansen et al. therefore identify a structural blind spot rather than a failure of the whole assessment tradition: short-delay tests can be valid for the domains they measure and still miss long-delay forgetting.
That distinction changes the patient-side interpretation. A normal 30-minute memory score should not automatically be used to dismiss an MS patient’s memory complaint as anxiety, fatigue, or exaggeration.
Fatigue and mood can influence subjective complaints, but the 2026 data show that at least some mismatch between symptoms and testing may come from the test window itself.
Clinical Translation Is Promising but Not Ready-Made
Adding ALF assessment to specialty MS cognitive workups would be technically simple: administer a standard verbal memory test, then call the patient 7 days later for delayed recall. The practical barrier is workflow, not theory.
Clinics would need follow-up scheduling, standardized instructions that avoid rehearsal, local norms, and a decision rule for interpreting the ratio.
The 2026 study’s cut-off should not be imported as a universal threshold. A combined ALF score of 0.71 maximized the Youden index in this sample, but cut-points for cognitive tests are population-dependent.
Age, education, disease stage, depression exclusions, and test language can shift the operating point.
For now, the clinical takeaway is direct: when an MS patient reports memory failures despite normal short-delay testing, a 7-day recall paradigm is a plausible next measurement, especially in specialty neuropsychology or research settings. It should complement, not replace, processing-speed measures, mood/fatigue assessment, sleep evaluation, and ordinary functional history.
Limitations of the 2026 MS ALF Study
Sample and site: the study analyzed 62 MS patients and 65 controls from one German center. That is larger than the 2024 pilot, but still not a multicenter validation cohort.
Exclusions: participants with MoCA < 26 or BDI-II ≥ 20 were excluded. That helped isolate ALF from global cognitive impairment and major depressive symptoms, but it limits inference for the very patients who may have the most complex clinical presentations.
Measurement domain: the study tested verbal memory with RAVLT and WMS-IV Logical Memory. It did not establish a visual-memory ALF marker for MS, and prior MS work has not shown the same clarity for visuospatial 7-day recall.10
Design: the study was cross-sectional. It cannot show whether ALF predicts future cognitive decline, treatment response, employment loss, or later dementia.
Longitudinal follow-up is the next required test if ALF is going to become more than a descriptive marker.
Practical Questions About 7-Day Memory Testing in MS
Does normal 30-minute recall rule out real MS memory impairment?
No. In Jansen et al., MS patients and controls were nearly identical at 30 minutes on both verbal-memory tasks, but the MS group recalled less at 7 days and had lower 7-day/30-minute ratios.1
Is ALF testing available in routine MS care?
Usually not as a standardized routine test. A specialty neuropsychology clinic could add a 7-day phone recall to an existing memory test, but broader use needs external validation, local norms, and agreed cut-points.
Does ALF mean MS is causing Alzheimer’s-type dementia?
No. ALF can appear in different neurological conditions when long-term consolidation networks are disrupted.
MS-related ALF is more plausibly tied to demyelination, disconnection, and cortical or hippocampal network injury than to amyloid-and-tau Alzheimer’s pathology.12
References
- Accelerated long-term forgetting as an objective marker of subjective memory impairment in multiple sclerosis. Jansen C, Stalter J, Reuter S, Witt K. medRxiv. 2026 (preprint). German Clinical Trials Register: DRKS00035204. doi:10.64898/2026.04.21.26351393
- Cognitive impairment in multiple sclerosis: clinical aspects and current evidence. Benedict RHB, Amato MP, DeLuca J, Geurts JJG. The Lancet Neurology. 2020;19(10):860-871. doi:10.1016/S1474-4422(20)30277-5
- Accelerated long-term forgetting: a perspective from neuroscience. Butler CR, Zeman A. Cortex. 2019;110:181-200. doi:10.1016/j.cortex.2018.10.011
- Subjective and objective memory in patients with multiple sclerosis. Krch D, Chiaravalloti ND, DeLuca J. Multiple Sclerosis Journal. 2011;17(8):1005-1011.
- Validation of the Brief International Cognitive Assessment for Multiple Sclerosis (BICAMS): a multicenter international study. Langdon DW, Amato MP, Boringa J et al. Multiple Sclerosis Journal. 2012;18(6):891-898. doi:10.1177/1352458511431076
- The medial temporal lobe and recognition memory. Squire LR, Wixted JT, Clark RE. Annual Review of Neuroscience. 2007;30:259-288. doi:10.1146/annurev.neuro.30.051606.094328
- Accelerated long-term forgetting in presymptomatic autosomal dominant Alzheimer’s disease. Weston PSJ et al. The Lancet Neurology. 2018;17(2):123-132. doi:10.1016/S1474-4422(17)30434-9
- The Brief International Cognitive Assessment for MS (BICAMS): International standards for validation. Benedict RHB et al. BMC Neurology. 2012;12:55. doi:10.1186/1471-2377-12-55
- Cognitive outcomes in disease-modifying treatments for relapsing-remitting multiple sclerosis: a systematic review. Tsivgoulis G et al. Therapeutic Advances in Neurological Disorders. 2018;11:1756286418754555.
- Accelerated long-term forgetting in mildly impaired patients with multiple sclerosis. Stalter J et al. Journal of Neurology. 2024;271(4):2150-2158.
- Cognitive rehabilitation in multiple sclerosis: a systematic review and meta-analysis. Mitolo M et al. Journal of the Neurological Sciences. 2015;354(1-2):1-9. doi:10.1016/j.jns.2015.05.004
- Cortical pathology in multiple sclerosis: differences and similarities with Alzheimer disease. Calabrese M et al. Multiple Sclerosis Journal. 2013;19(7):887-893. doi:10.1177/1352458512457845
