A 2026 double-blind randomized controlled trial by Kappes et al. in Psychopharmacology found something the smart-drug users likely wouldn’t have predicted: a single 30 mg dose of methylphenidate reduced dishonest misreporting on a die-rolling task from ~22% of trials in the placebo arm to ~6% (in the methylphenidate arm).1 Atomoxetine, a noradrenergic comparator, had no detectable effect.
Research Highlights
- Kappes 2026: 151 healthy adults were randomized to placebo (n=52), methylphenidate 30 mg (n=49), or atomoxetine 60 mg (n=50) in a double-blind design. Dishonesty was measured 90 minutes post-dose with a private die-rolling task across 25 rounds.1
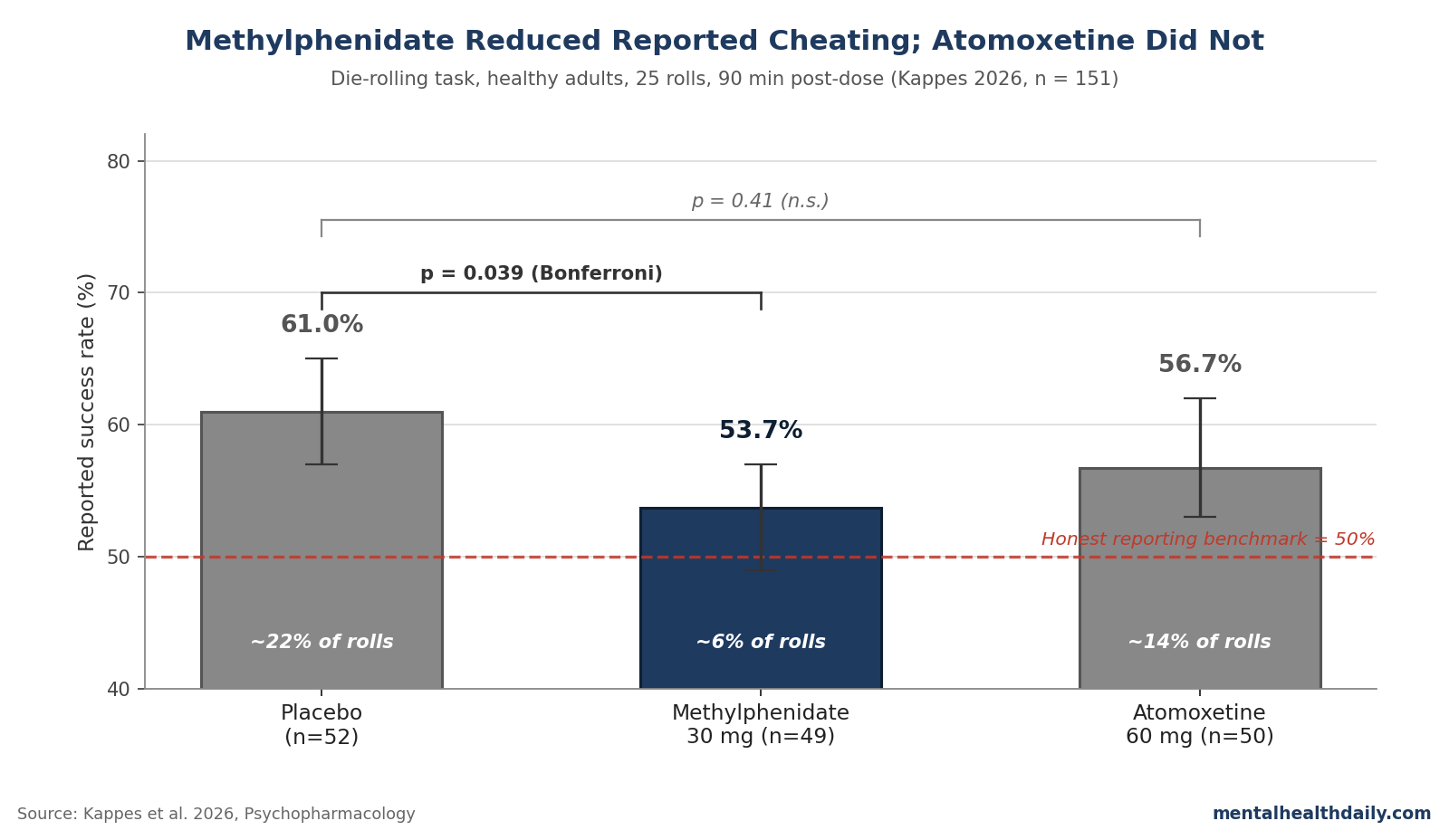
- Reported success rates: placebo 61%, methylphenidate 53.7%, atomoxetine 56.7%. The methylphenidate vs. placebo gap was significant (z statistic = 2.48, p = 0.039 after Bonferroni correction); atomoxetine vs. placebo was not (p = 0.41).1
- Methylphenidate reduced misreporting: translated into cheating rates, the methylphenidate arm fell from roughly 22% of rolls to roughly 6% — a 72% relative reduction.1
- Standard mediators did not explain it: the effect held after controlling for attention, mood, impulsivity, risk preference, and self-interest. It also did not look like a demand effect: 62% of participants guessed their drug condition incorrectly.1
- Public intuition missed the direction: a separate sample of 575 US adults — including self-reported smart-drug users — could not predict the effect. Most expected no impact or a small increase in cheating.1
In this paradigm, methylphenidate shifted a private payoff decision toward honest reporting, and that direction — less cheating, not more — runs against both the cognitive-doping concern and the assumption that performance enhancers should help users get away with more.
Smart-drug discourse typically splits two ways: (1) the worry that prescription stimulants give users an unfair edge, and (2) the counter-meta-analytic finding that single-dose effects on actual learning and complex cognition in healthy adults are modest at best.2,3
But the question asked was as follows: When methylphenidate raises tonic dopamine in fronto-striatal circuits that gate cost-benefit weighting around self-interested cheating, what happens to the cheating?4
Methylphenidate vs. Atomoxetine RCT in 151 Healthy Adults
Kappes et al. used a clean stimulant-comparator design:
- Participants: 151 healthy adults after exclusions, gender-balanced, and screened to exclude attention-deficit/hyperactivity disorder (ADHD), prior stimulant use, depression (Beck Depression Inventory > 12), hazardous drinking, and other comorbid conditions.
- Drug arms: placebo (n=52), methylphenidate 30 mg (n=49), and atomoxetine 60 mg (n=50). These doses match standard single-dose levels used in healthy-adult cognition research.1
- Timing: dishonesty was measured 90 minutes post-dose, when both drugs should have active central effects.
The die-rolling task had 3 moving parts:
- Payoff was known before the roll. Participants were told which outcomes paid £1 and which paid nothing.
- The roll was private. Each person rolled the die under a cup alone in an isolated room and self-reported the result.
- Dishonesty was measured at group level. The population-mean success rate should equal 50% under honest reporting; any systematic excess above 50% implies misreporting.
The same task has previously predicted real-world dishonesty in education, prison misconduct, and corruption settings.5
Demand effects were checked directly. Only 38% of participants correctly guessed which drug they had received — no better than chance — and beliefs about drug condition were unrelated to dishonesty.1
Misreporting Fell from 22% to 6%; Atomoxetine Sat in the Middle
All three groups exceeded the 50% honesty benchmark, meaning dishonesty was present everywhere — this isn’t a study of saints versus cheaters. The differences were in degree:
- Placebo: 61% reported successes (95% confidence interval [CI] 57–65%). Implied misreporting on roughly 22% of rolls.
- Methylphenidate: 53.7% reported successes (95% CI 49–57%). Implied misreporting on roughly 6% of rolls.
- Atomoxetine: 56.7% reported successes (95% CI 53–62%). Implied misreporting on roughly 14% of rolls.
The methylphenidate vs. placebo contrast was the only one that survived Bonferroni adjustment (z = 2.48, p = 0.039). Atomoxetine vs. placebo did not reach significance (z = 1.48, p = 0.41), and atomoxetine vs. methylphenidate also fell short (z = 0.99, p = 0.97).1
A Bayesian simulation with 100,000 honest-reporting samples returned a probability of 0.00002 of seeing the methylphenidate-vs.-placebo gap by chance — about 2 in 100,000.
The atomoxetine-vs.-placebo gap, by contrast, would have arisen by chance in roughly 2,170 of those simulations (p = 0.022 unadjusted, but null after correction).1
Methylphenidate vs. Atomoxetine: Striatal Dopamine May Explain Gaps in Dishonesty / Cheating
Both drugs block norepinephrine reuptake, but they diverge on dopamine: methylphenidate raises striatal dopamine, while atomoxetine is essentially selective for norepinephrine.6 That makes the dissociation in the Kappes data informative.
If dishonesty were driven by attention or arousal alone, both drugs should have moved the dial — both sped up sustained-attention reaction times in the trial. Only methylphenidate moved misreporting.
The dopaminergic read also fits a small but consistent prosociality literature.
Levodopa has reduced self-interested behavior in healthy male volunteers; tolcapone, a catechol-O-methyltransferase (COMT) inhibitor that raises prefrontal dopamine, has promoted egalitarian division of resources.7,8
The direction isn’t universal — amisulpride flips with sex9 — but methylphenidate’s placement in this picture is broadly consistent.
Hinshaw 1992 Found the Opposite Effect in 22 Boys with ADHD
The headline has to contend with a 1992 study by Hinshaw et al. that pointed the other way: in 22 medicated boys with diagnosed ADHD, methylphenidate increased cheating on a structured academic-style task — though only on the day of dosing, with an order effect that complicates the read.10
Both findings can be true at once. Hinshaw studied 22 children with an ADHD diagnosis on standing medication; Kappes studied 151 unmedicated healthy adults receiving a single dose.
Single-dose stimulant pharmacology in healthy adults differs from chronic stimulant pharmacology in children with the diagnosis at both receptor-occupancy and behavioral-baseline levels.11 The Kappes finding is the best-controlled evidence so far on healthy adults; it does not extend to children with ADHD on standing therapy.
Mood, Attention, Impulsivity, and Risk Don’t Mediate the Effect
The most defensible methylphenidate-skeptic move is to argue that any behavioral change is downstream of attention or arousal, not the behavior of interest. Kappes ran that gauntlet:
- Attention. Drug-induced changes in sustained-attention reaction time were not associated with dishonesty (β = 0.003, p = 0.97). Controlling for attention reaction time (RT) did not move the methylphenidate effect (F = 4.63, p = 0.030).1
- Mood. Methylphenidate increased self-reported energy compared to placebo and atomoxetine. After adjusting for energy, the dishonesty effect remained (F = 5.57, p = 0.020). Adjusting for contentment also did not change the effect.1
- Impulsivity, risk, self-interest. No significant effect of methylphenidate on delay-discounting impulsivity, lottery-vs.-certain risk preference, or charitable giving. Controlling for these did not eliminate the methylphenidate-on-dishonesty effect.1
The standard candidate mediators — the variables a reviewer would ask about first — don’t carry the result. Whatever methylphenidate is doing to the cost-benefit calculation around honest vs. dishonest reporting, it isn’t obviously routed through any of the obvious cognitive primitives.
Public intuition got the direction wrong. Kappes also ran a parallel survey of 575 US adults, recruited to be representative on age, gender, income, and education. After being walked through the experimental setup and shown the placebo-arm cheating rate, participants were asked — with a $2 incentive for accuracy — to predict how methylphenidate would shift dishonesty.
The distribution: 27.9% predicted no effect, 24.0% a small decrease, 21.3% a small increase, 15.2% a substantial increase, 11.7% a substantial decrease. Roughly the same number of people guessed direction-down as guessed direction-up. Self-reported prior users of methylphenidate or other smart drugs (17.4% of the sample) were not measurably better at predicting the direction (χ² = 3.16, p = 0.55).1
The result matters because the direction of methylphenidate’s honesty effect is not predictable from the assumptions people, including users, actually hold. This is one of those rare findings where lay intuition isn’t even biased in a particular direction; it’s diffuse.
Limitations of the Kappes Trial
Four caveats deserve mentioning.
- Single dose, single task, single lab. The 30 mg methylphenidate dose is one acute administration. The die-rolling task is one operationalization of dishonesty, validated against real-world correlates but still a lab paradigm. Replication in other dishonesty paradigms (cheating on cognitive tests, tax-style misreporting) and other doses would tighten the inference.5
- Healthy-adult sample only. Participants were screened to exclude ADHD diagnoses, current or prior stimulant use, depression, and a long list of conditions. The result speaks to a clean baseline. It says nothing about effects in patients with ADHD on chronic methylphenidate, where Hinshaw 1992 found the opposite direction in children.10
- The mechanism remains speculative. The atomoxetine null is consistent with a striatal-dopamine story, but the trial was not designed to localize the effect. Westbrook 2020 work on dopamine’s effect on cost-benefit weighting of cognitive effort offers a plausible mechanism — methylphenidate may shift the weighting toward acting in line with effortful values rather than easy financial gain — but this is interpretation, not mechanism in the rigorous sense.4
- Group-level inference only. The 22% placebo-arm misreporting rate is estimated from the gap between observed and honest-baseline success rates, not directly observed per-participant. Group-level honesty-benchmarking is the standard approach for this paradigm, but it limits any per-individual inference about who cheated and who didn’t.5
For the smart-drug ethics debate: most cognitive-enhancement discussions assume that performance-enhancing drugs either: (A) help users cheat more effectively (the unfair-advantage frame) or (B) are functionally neutral on moral behavior (the “just like coffee” frame). Kappes 2026 doesn’t cleanly fit either.12
A drug that modestly improves attention and reduces dishonesty in the same lab session is a strange object for the ethics debate. It strengthens the case that single-dose effects of methylphenidate on actual high-stakes performance in healthy adults are smaller than users hope — the cognition meta-analyses already point that way2,3 — while complicating the cheating-amplifier worry.
Practically, a single 30 mg dose of methylphenidate in a healthy adult, in this specific lab paradigm, did not facilitate cheating and may have suppressed it.
The result doesn’t support off-label use — as medical risks of unsupervised stimulant use remain separate from this finding. But it does suggest the “Ritalin makes cheating easier” intuition isn’t accurate.
FAQs: Methylphenidate, Atomoxetine, and Smart-Drugs
Does this mean methylphenidate makes people more honest in general?
In this lab paradigm, a single 30 mg dose reduced misreporting in healthy adults.
The effect was not mediated by mood, attention, impulsivity, or risk preference, which strengthens the inference. It has not been replicated outside this study, and it doesn’t test whether the effect generalizes to other forms of moral behavior.1
Does methylphenidate actually improve cognition in healthy adults?
Modestly, on some tasks. Meta-analyses of single-dose stimulants in healthy adults find small improvements in sustained attention and inhibitory control but inconsistent effects on complex cognition, learning, and creativity.
Kappes saw a 41 ms sustained-attention reaction-time improvement vs. placebo — statistically meaningful, behaviorally modest.2,3
What’s the difference between methylphenidate and atomoxetine?
Methylphenidate (Ritalin) blocks reuptake of both dopamine and norepinephrine and is a Schedule II controlled substance. Atomoxetine (Strattera) is a selective norepinephrine reuptake inhibitor with no controlled-substance scheduling.
Both are Food and Drug Administration (FDA)-approved for ADHD; atomoxetine is the standard first-line non-stimulant.6
Did the participants know which drug they got?
Mostly not. Only 38% of participants correctly guessed their drug condition — no better than chance. Beliefs about drug condition did not correlate with dishonesty, and controlling for those beliefs did not change the methylphenidate effect.1
Should this change anyone’s decision to take Ritalin off-label?
No. The trial is a mechanism finding, not a recommendation.
Off-label methylphenidate use carries cardiovascular, dependence, and legal risks that are entirely separate from this result. Cognition meta-analyses also suggest performance gains in healthy adults are modest with substantial individual variability.2,3
What’s the next study that needs to happen?
- Replication: an independent-lab test using a different dishonesty paradigm.
- Dose-response: lower- and higher-dose methylphenidate arms.
- Mechanism: comparison against a more selective dopaminergic agent, such as a D1 or D2 agonist, to localize the subcircuit.
- Clinical extension: ADHD-diagnosed adults, to see whether the direction reverses with diagnosis as Hinshaw suggested it might in children.
References
- Beyond attention: methylphenidate reduces dishonesty in healthy adults. Kappes A, Cohn A, Maréchal MA, et al. Psychopharmacology. 2026. doi:10.1007/s00213-026-07059-y
- Prescription stimulants in individuals with and without attention deficit hyperactivity disorder: misuse, cognitive impact, and adverse effects. Lakhan SE, Kirchgessner A. Brain and Behavior. 2012;2(5):661–677. doi:10.1002/brb3.78
- Prescription stimulants’ effects on healthy inhibitory control, working memory, and episodic memory: a meta-analysis. Ilieva IP, Hook CJ, Farah MJ. Journal of Cognitive Neuroscience. 2015;27(6):1069–1089. doi:10.1162/jocn_a_00776
- Dopamine promotes cognitive effort by biasing the benefits versus costs of cognitive work. Westbrook A, van den Bosch R, Maier JX, et al. Science. 2020;367(6484):1362–1366. doi:10.1126/science.aaz5891
- Civic honesty around the globe. Cohn A, Maréchal MA, Tannenbaum D, Zünd CL. Science. 2019;365(6448):70–73. doi:10.1126/science.aau8712
- Dissociable and common effects of methylphenidate, atomoxetine and citalopram on response inhibition neural networks. Nandam LS, Hester R, Bellgrove MA. Neuropsychologia. 2014;56:263–270. doi:10.1016/j.neuropsychologia.2014.01.011
- Dopaminergic stimulation increases selfish behavior in the absence of punishment threat. Pedroni A, Eisenegger C, Hartmann MN, et al. Psychopharmacology. 2014;231(1):135–141. doi:10.1007/s00213-013-3210-x
- Dopamine modulates egalitarian behavior in humans. Sáez I, Zhu L, Set E, Kayser A, Hsu M. Current Biology. 2015;25(7):912–919. doi:10.1016/j.cub.2015.01.071
- The dopaminergic reward system underpins gender differences in social preferences. Soutschek A, Burke CJ, Beharelle AR, et al. Nature Human Behaviour. 2017;1(11):819–827. doi:10.1038/s41562-017-0226-y
- Stimulant medication and the treatment of aggression in children with attentional deficits. Hinshaw SP, Heller T, McHale JP. Journal of Consulting and Clinical Psychology. 1992;60(2):274–281. doi:10.1037/0022-006X.60.2.274
- Cognitive effects of methylphenidate in healthy volunteers: a review of single-dose studies. Linssen AMW, Sambeth A, Vuurman EFPM, Riedel WJ. International Journal of Neuropsychopharmacology. 2014;17(6):961–977. doi:10.1017/S1461145713001594
- The future of psychopharmacological enhancements: expectations and policies. Schermer M, Bolt I, de Jongh R, Olivier B. Neuroethics. 2009;2(2):75–87. doi:10.1007/s12152-009-9032-1
