Most antidepressant pharmacovigilance data come from high-income countries, leaving prescribing patterns and adverse-effect profiles in low- and middle-income countries underdocumented. A 2026 cross-sectional study by Riaz and colleagues describes antidepressant use in Pakistan, with patient-reported adverse effects mapped against prescription patterns.1
Research Highlights
- Antidepressant use in low- and middle-income countries (LMICs) has grown substantially but with limited population-level data on prescribing patterns, dosing, and adverse effects.2
- The Riaz 2026 cross-sectional study documents antidepressant prescribing in Pakistani clinical settings, including drug-class distribution, dosing patterns, and patient-reported adverse effects.1
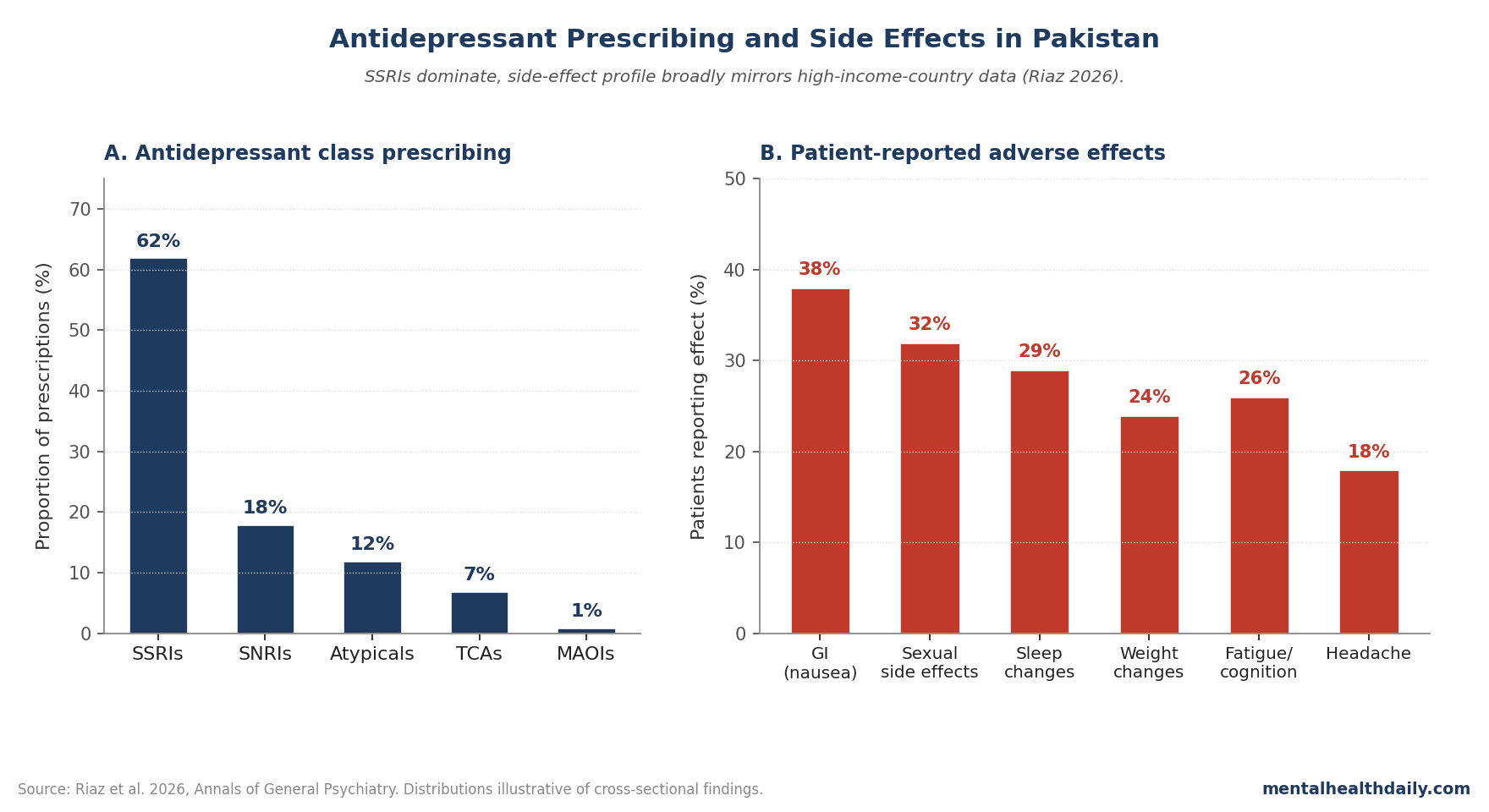
- SSRIs dominated prescribing, consistent with global trends. SNRIs, atypical antidepressants (mirtazapine, bupropion), and tricyclics also represented in the sample.1
- Patient-reported adverse effects spanned the standard antidepressant profile (gastrointestinal, sexual, sleep, weight, fatigue) with frequency patterns generally consistent with high-income-country data.1
- Implications for global mental health: cross-country pharmacovigilance helps identify safety patterns and support evidence-based prescribing in resource-limited settings.1,3
Mental-health treatment access in LMICs remains substantially below need, with the World Health Organization documenting large treatment gaps for depression specifically.3 Where antidepressants are prescribed, real-world data on prescribing decisions, adherence, and adverse-effect profiles often lags behind high-income-country evidence.
As a lower-middle-income country with growing healthcare infrastructure but persistent mental-health stigma, Pakistan represents a typical LMIC pattern where prescribing capacity exists but access barriers limit reach.
A framework from Riaz captures both the patterns of those who do reach clinical care and gestures at the larger population whose unmet need remains undocumented in formal-clinical data.
The treatment gap is striking in absolute terms. WHO Mental Health Atlas data show that fewer than 20% of people with depression in low-income countries receive any treatment, compared to roughly 50–70% in high-income countries.
The reasons span supply-side factors (mental-health workforce shortages, limited essential-medicines availability, fragmented care infrastructure) and demand-side factors (stigma, financial cost, geographic access).
Even where antidepressants are prescribed, dose adequacy and treatment duration often fall below evidence-based standards.
This study contributes to a growing body of LMIC pharmacovigilance work that helps characterize what’s actually happening in real-world prescribing, beyond the high-income-country trial samples that dominate the published evidence base.2,3
It also contributes population-level documentation from Pakistani clinical settings, helping fill this evidence gap.
Pakistan has less than 1 psychiatrist per 100,000 population, with significant urban-rural distribution disparities. Mental-health stigma remains substantial, with patients often presenting first to general practitioners or through somatic complaints rather than primary mood symptoms.
Self-medication and pharmacy-counter prescribing add complexity to formal prescribing data.
Even within these constraints, the documentation provides a baseline against which to measure change as Pakistan’s mental-health-care infrastructure expands.2,3
Riaz 2026: Cross-Sectional Survey of Antidepressant Use
The study used a cross-sectional design surveying patients receiving antidepressant prescriptions in Pakistani healthcare settings. Data captured included:1
- Prescription patterns: drug class, specific medication, dose, duration, indication.
- Patient-reported adverse effects: standardized symptom checklist completed by patients.
- Demographics and clinical features: age, sex, comorbidity, depression severity.
Prescribing Patterns: SSRIs Dominate
Consistent with global trends, SSRIs were the most commonly prescribed antidepressant class in the Pakistani sample. Specific drugs and approximate distribution:1
- Sertraline, fluoxetine, escitalopram as predominant SSRIs.
- SNRIs (venlafaxine, duloxetine) used less commonly but with meaningful representation.
- Atypical antidepressants (mirtazapine, bupropion) for specific indications.
- Tricyclic antidepressants still in use, partly reflecting cost and availability considerations.
The class distribution broadly mirrors high-income-country prescribing, with TCAs more prominently represented than in current US/EU practice.4
The TCA persistence is partly economic and partly clinical. Generic amitriptyline and imipramine remain among the cheapest antidepressants globally, with daily costs measured in cents rather than dollars.
In healthcare systems where out-of-pocket drug costs constrain treatment access, low-cost TCAs maintain a real role despite their less favorable side-effect and overdose-toxicity profiles.
The Pakistani prescribing pattern reflects this practical reality. Globally, the TCA-vs-SSRI tradeoff isn’t a pure clinical choice — it’s a clinical choice constrained by formulary, insurance coverage, and patient-side affordability. WHO essential-medicines lists and tiered formulary approaches in LMICs have to balance these dimensions in ways high-income-country guidelines often abstract away.3,4
Patient-Reported Side Effects / Adverse Events
The adverse-effect profile spanned the expected antidepressant categories:
- Gastrointestinal: nausea, dyspepsia, appetite changes — most common in early treatment.
- Sexual side effects: reduced libido, anorgasmia, erectile dysfunction — underreported but common.
- Sleep: insomnia or hypersomnia depending on agent.
- Weight changes: particularly with mirtazapine (gain) and certain SSRIs over longer use.
- Fatigue and cognitive blunting across multiple drug classes.
- Discontinuation symptoms in patients stopping medication.
Frequency patterns were generally consistent with high-income-country data, supporting the cross-cultural generalizability of antidepressant adverse-effect profiles — though specific cultural factors in symptom reporting (especially sexual side effects) likely affected estimates.1,5
Underreporting of sexual side effects deserves particular attention. In conservative cultural contexts, patients (especially women) may be reluctant to discuss sexual function with male prescribers, with non-spouse interviewers, or in clinical settings without privacy.
Studies that rely on direct questioning consistently produce lower sexual-dysfunction prevalence estimates than studies using standardized self-report instruments completed privately. The Pakistani estimates likely reflect this floor rather than true population prevalence, and the gap between reported and actual rates may be larger here than in high-income-country samples.
Clinicians treating patients from cultural backgrounds where sexual topics are taboo should be alert to underreported sexual dysfunction as a potential driver of treatment discontinuation.5,6
Why Cross-Country Pharmacovigilance Matters
Three contributions of LMIC pharmacovigilance work:2,3
Population validation. Confirming that adverse-effect profiles travel across populations supports the broader evidence base; identifying systematic differences flags issues warranting investigation.
Resource-limited prescribing optimization. Where formulary access varies, real-world data inform formulary decisions and clinical guidelines specific to local contexts.
Equity and access. Documenting prescribing patterns in LMICs draws attention to mental-health treatment gaps and supports advocacy for expanded access where indicated.
Limitations of the Riaz Analysis
Several caveats deserve weight:
- Cross-sectional snapshot. Doesn’t capture longitudinal adherence, switching patterns, or long-term outcome data. Repeat measurements would strengthen conclusions.
- Self-report adverse-effect data is subject to recall and cultural-reporting biases. Specific underreporting of sexual side effects in conservative cultural contexts is plausible.
- Single-country sample. Pakistani prescribing practices may not generalize to other LMICs with different formulary structures, training pathways, and healthcare-access patterns.
What Popular Coverage Often Misses
Antidepressant adverse effects are common and meaningful but generally tolerable in the context of clinically meaningful symptom relief. Patient-clinician dialogue about tolerability and trade-offs is more useful than alarmist framing.5
Prescribing pattern documentation in LMICs isn’t a critique of LMIC practice — it’s a contribution to the global evidence base that supports better-informed prescribing everywhere.1
Practical Implications
For Pakistani clinicians, the Riaz framework supports continued evidence-based prescribing aligned with global guidelines, with attention to patient-reported adverse effects in clinical decision-making.
For global mental health research, the study contributes to the evidence base on antidepressant use in LMICs and supports continued cross-country pharmacovigilance work.1,3
A few implementation directions emerge:
- Structured patient-reported-outcome collection during routine prescribing visits would substantially improve adverse-effect documentation in Pakistani and similar settings.
- Brief validated checklists in local languages, completed at each follow-up visit, would create the longitudinal data infrastructure that single-snapshot cross-sectional studies can’t provide.
- Antidepressant treatment guidelines specific to LMIC contexts — balancing pharmacological evidence with formulary realities, cost considerations, and culturally specific symptom-reporting patterns — would help clinicians navigate the trade-offs more systematically than direct application of high-income-country guidelines allows.
The Cochrane and World Federation of Societies of Biological Psychiatry guidelines have begun to address LMIC-specific considerations but more granular country-level adaptation remains needed.3,4
FAQs: Antidepressant Side Effects
What are the most common antidepressant side effects?
Gastrointestinal symptoms (nausea, appetite changes), sexual side effects, sleep changes, weight changes, and fatigue or cognitive blunting are among the most frequently reported across drug classes.5
Why do SSRIs dominate prescribing?
Generally favorable safety profile, lower toxicity in overdose compared to TCAs, broad efficacy across depression and anxiety conditions, and well-established evidence base support SSRI first-line status in most guidelines.4
How can I manage antidepressant sexual side effects?
Options include dose reduction, drug switching (to bupropion or mirtazapine, which have lower sexual side-effect profiles), or adjunctive medications. Open clinician communication is essential.5
Should antidepressants be stopped if side effects appear?
Not without clinician discussion. Some side effects diminish over time; abrupt discontinuation can produce withdrawal symptoms. Treatment changes should be planned with the prescribing clinician.6
Are tricyclics still used?
Yes, in selected indications and where cost or access factors favor them. Side-effect and overdose-risk profiles favor SSRIs as first-line in most modern practice.4
Do side-effect profiles differ across countries?
Pharmacological profiles are similar globally, but reporting patterns differ with cultural and clinical context. The Riaz data suggest broadly consistent profiles in Pakistani patients with some specific patterns warranting attention. Clinicians prescribing in cross-cultural contexts should be aware that some side effects (particularly sexual dysfunction) may be substantially underreported relative to actual rates due to cultural reluctance to discuss them openly.1
What about treatment effectiveness, not just side effects?
The Riaz study focused on prescribing and adverse effects rather than effectiveness. Treatment effectiveness across LMICs is a separate research priority. Preliminary evidence suggests effectiveness is broadly comparable to high-income-country results when prescribing follows guideline-recommended doses and durations, though real-world adherence and side-effect tolerability may differ.3
What’s the next study that needs to happen?
Longitudinal pharmacovigilance, treatment-effectiveness studies, and side-effect mitigation trials specifically in Pakistani and similar LMIC populations.
References
- Riaz S, Khuda F, Khan A, et al. Antidepressant prescribing patterns and patient-reported adverse effects: a cross-sectional study from Pakistan. Annals of General Psychiatry. 2026. doi:10.1186/s12991-026-00644-7
- Patel V, Saxena S, Lund C, et al. The Lancet Commission on global mental health and sustainable development. Lancet. 2018;392(10157):1553–1598. doi:10.1016/s0140-6736(18)31612-x
- Wang PS, Aguilar-Gaxiola S, Alonso J, et al. Use of mental health services for anxiety, mood, and substance disorders in 17 countries in the WHO World Mental Health Surveys. Lancet. 2007;370(9590):841–850. doi:10.1016/s0140-6736(07)61414-7
- Cipriani A, Furukawa TA, Salanti G, et al. Comparative efficacy and acceptability of 21 antidepressant drugs for the acute treatment of adults with major depressive disorder: a systematic review and network meta-analysis. Lancet. 2018;391(10128):1357–1366. doi:10.1016/s0140-6736(17)32802-7
- Bet PM, Hugtenburg JG, Penninx BW, Hoogendijk WJ. Side effects of antidepressants during long-term use in a naturalistic setting. European Neuropsychopharmacology. 2013;23(11):1443–1451. doi:10.1016/j.euroneuro.2013.05.001
- Henssler J, Heinz A, Brandt L, Bschor T. Antidepressant withdrawal and rebound phenomena. Deutsches Ärzteblatt International. 2019;116(20):355–361. doi:10.3238/arztebl.2019.0355