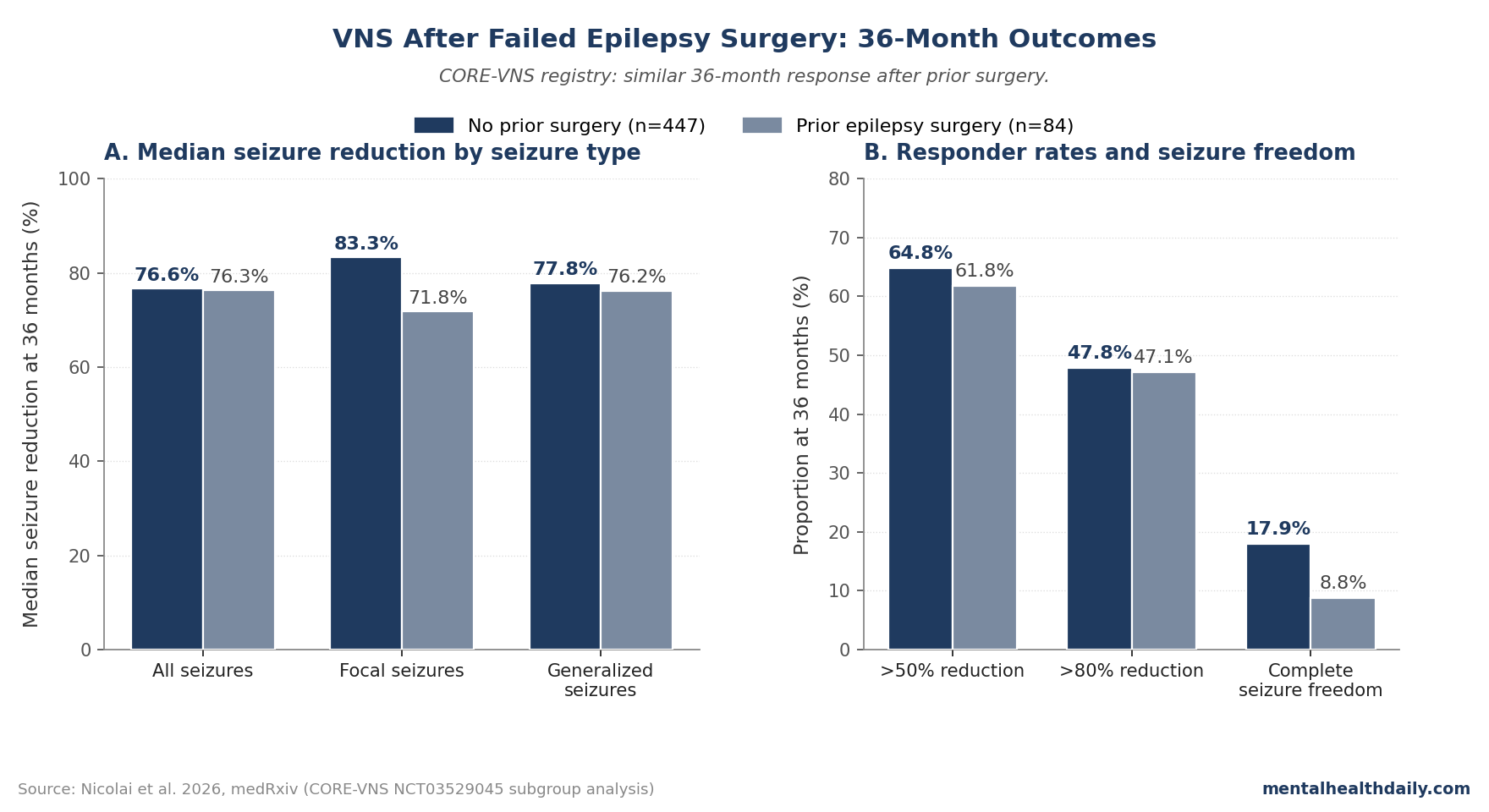
A 2026 CORE-VNS registry analysis found that implanted vagus nerve stimulation still produced large 36-month seizure reductions after failed intracranial epilepsy surgery: median all-seizure reduction was 76.3% with prior surgery vs. 76.6% without prior surgery.1
Research Highlights
- Prior surgery did not blunt the main VNS signal: CORE-VNS reported 76.3% median all-seizure reduction at 36 months in patients with prior intracranial surgery for epilepsy vs. 76.6% in those without prior surgery.1
- The responder rates were close: 61.8% of prior-surgery patients and 64.8% of no-prior-surgery patients reached more than 50% seizure reduction; 47.1% vs. 47.8% reached more than 80% reduction.1
- Complete seizure freedom remained harder after failed surgery: 8.8% of prior-surgery patients vs. 17.9% of no-prior-surgery patients reported freedom from all seizures in the 3-month reporting interval at 36 months.1
- The safety comparison also matched the hypothesis: overall adverse events were 36.9% vs. 41.2% (IRR 0.91, 95% CI 0.70–1.19), and serious adverse events were 3.6% vs. 7.2% (IRR 0.56, 95% CI 0.20–1.56).1
- The evidence is stronger than the old label warning: the current VNS Therapy label language says safety and efficacy are not established after prior brain surgery, but this registry analyzed 84 prior-surgery patients and 447 patients without prior intracranial surgery.1
Vagus nerve stimulation (VNS) is an implanted neuromodulation therapy that sends intermittent electrical pulses through the cervical vagus nerve, then into brainstem circuits that project widely through norepinephrine and serotonin systems. In drug-resistant epilepsy, the goal is usually seizure reduction rather than cure.
The specific question in Nicolai et al. was narrower than “does VNS work?” The researchers tested whether prior intracranial surgery for epilepsy (ICSE) changed VNS effectiveness or safety. That question matters because the VNS label still carries a caution that safety and efficacy are not established in patients with prior brain surgery, mostly because the original approval-era trials did not include enough such patients to answer the subgroup question.1
CORE-VNS Directly Tested the Old Prior-Surgery Warning
CORE-VNS is an international, multicenter, prospective observational registry of people receiving VNS for drug-resistant epilepsy. Nicolai et al. analyzed 531 implanted participants: 84 had prior intracranial surgery for epilepsy, and 447 had no prior intracranial epilepsy surgery.1
the study stated 2 linked hypotheses: VNS effectiveness would not differ after prior ICSE because VNS activation reaches cortex through brainstem-first pathways, and adverse effects such as cough or voice alteration would not differ because they arise from extracranial vagus-nerve motor fibers rather than the previously operated brain tissue.1
That hypothesis was broadly confirmed. The prior-surgery group did not show worse median seizure reduction, did not show a safety penalty, and did not diverge on the main responder thresholds.
The paper’s more cautious point is that complete seizure freedom was lower after failed surgery, which is exactly what one would expect if that subgroup had more refractory epilepsy at baseline.
The 84 prior-surgery patients were not a single clean surgical subtype. Prior procedures included lesionectomy in 31 patients (36.9%), lobar resection in 19 (22.6%), callosotomy in 16 (19.0%), anterior temporal lobectomy in 6 (7.1%), anterior temporal lobectomy plus amygdalohippocampectomy in 5 (6.0%), laser ablation in 4 (4.8%), lobar disconnection in 4 (4.8%), multilobar resection in 3 (3.6%), multiple subpial transections in 3 (3.6%), and several rare categories with 1 case each.1
36-Month Seizure Reduction Was Nearly Identical
For all seizures, the median monthly seizure count fell from 24.00 to 6.00 in the prior-surgery group and from 15.00 to 3.00 in the no-prior-surgery group by 36 months. Expressed as percent reduction, that was 76.3% vs. 76.6%.1
Focal and generalized seizure categories pointed in the same direction:
- Focal seizures: 71.8% median reduction after prior surgery vs. 83.3% without prior surgery at 36 months.
- Generalized seizures: 76.2% median reduction after prior surgery vs. 77.8% without prior surgery at 36 months.
- Between-group testing: the 36-month seizure-reduction comparison across seizure types was not significant (p = 0.3485), and the paper reported p > 0.05 for all seizure-frequency reduction comparisons across time points and seizure types.
These numbers do not prove the 2 groups were biologically identical. The prior-surgery group was smaller, had wider confidence intervals, and started with a higher median seizure frequency.
But the core clinical reading is straightforward: failed epilepsy surgery did not erase the long-term VNS response signal.
Responder Thresholds Support VNS, While Seizure Freedom Stays Calibrated
The responder analysis makes the practical read cleaner. At 36 months, 61.8% of prior-surgery patients and 64.8% of no-prior-surgery patients had more than 50% reduction in all seizures reported at baseline.
For the more demanding more-than-80% threshold, the rates were almost the same: 47.1% vs. 47.8%.1
The divergence appears at the strictest endpoint. A 100% reduction in baseline-reported seizures occurred in 16.2% of prior-surgery patients vs. 29.9% of no-prior-surgery patients.
Complete freedom from all seizures, including seizure types not reported at baseline, was 8.8% vs. 17.9%.1
That is not a reason to write off VNS after surgery. It is a reason to avoid selling VNS as a second surgery-level cure.
Prior-surgery context: patients who undergo intracranial surgery and still need VNS are often the hardest group: they may have multifocal epilepsy, a larger epileptogenic network, incomplete localization, or seizure mechanisms not fully captured by the surgical target.
Nicolai et al. note that baseline seizure frequency was higher in the prior-surgery group, which fits that interpretation.1
Safety Did Not Worsen After Prior Intracranial Surgery
The safety hypothesis also held. Overall adverse events were reported in 31 of 84 prior-surgery patients (36.9%) and 184 of 447 no-prior-surgery patients (41.2%), with an incidence rate ratio of 0.91 (95% CI 0.70–1.19).
Events related to stimulation or the device were almost identical: 32.1% vs. 32.0% (IRR 1.04, 95% CI 0.79–1.38).1
Serious adverse events were uncommon in both groups: 3.6% with prior surgery vs. 7.2% without prior surgery. Serious events related to implant or procedure were 2.4% vs. 2.5%, and no serious stimulation- or device-related events occurred in the prior-surgery group.1
The mechanism makes the safety result plausible. VNS stimulation reaches central networks through the vagus nerve and brainstem relays such as the nucleus tractus solitarius, locus coeruleus, and raphe nuclei.
Voice change, cough, throat sensation, and dyspnea are usually tied to peripheral activation of vagal fibers traveling to laryngeal muscles. Prior cortical or callosal surgery should not be expected to create a new extracranial stimulation side-effect pathway.1
Earlier Post-Surgery VNS Studies Were Smaller but Pointed the Same Way
The CORE-VNS analysis is useful because it turns a scattered prior-surgery literature into a larger prospective comparison. Earlier papers were directionally supportive but too small or too narrow to settle the label-level question.
Hatano et al. studied VNS after corpus callosotomy, a palliative surgery that disconnects the 2 cerebral hemispheres to reduce seizure spread rather than removing a single seizure focus. In that retrospective study, 13 patients received VNS after callosotomy and 24 did not.
At 1 year, 53.85% of the post-VNS group vs. 12.5% of the non-post-VNS group achieved at least 50% seizure reduction (p = 0.017); at 2 years, the rates were 41.67% vs. 27.78%.2
Kawai et al. followed a prospective Japanese registry of VNS for drug-resistant epilepsy. Efficacy analyses included 362 patients, 49.7% of whom had prior epilepsy surgery.
Median seizure reduction increased over time from 25.0% at 3 months to 66.2% at 36 months, and responder rates rose from 38.9% to 58.8% over the same interval.3
The newer CORE-VNS generalized tonic-clonic seizure analysis adds a same-registry companion result: by 12 months, participants with generalized tonic-clonic seizures had a 73.9% median reduction in that seizure type.4 Nicolai et al. then extend the registry read to the failed-surgery subgroup and show that prior ICSE did not materially change the 36-month VNS pattern.
Clinical Sequencing After Failed Epilepsy Surgery
the most direct clinical takeaway is: prior failed intracranial epilepsy surgery should not be treated as a blanket reason to avoid implanted VNS. The old “not established” label language is weaker than the current evidence base, especially now that CORE-VNS includes 84 prior-surgery patients with similar 36-month seizure-reduction and safety outcomes.1
That does not make VNS a replacement for surgery in a good surgical candidate. Long-term epilepsy surgery meta-analysis and later surgical reviews still support resection or disconnection when the seizure focus is well localized and the risk-benefit profile is favorable.5
VNS fits differently: it is an adjunctive neuromodulation option for patients whose seizures remain drug-resistant, whose epilepsy is not surgically resectable, or whose prior surgery did not produce seizure freedom.
Several limits keep the interpretation calibrated:
- Not randomized: CORE-VNS is prospective, but the prior-surgery subgroup comparison was post-hoc and observational.
- Not procedure-specific: lesionectomy, lobar resection, callosotomy, temporal lobectomy, laser ablation, and disconnection procedures were pooled.
- Not peer reviewed yet: the Nicolai paper was posted as a medRxiv preprint in April 2026.
- Not independent of manufacturer involvement: LivaNova funded the work, several contributors were LivaNova employees or stockholders, and 1 author served on a LivaNova advisory council.
- Not a transcutaneous VNS result: the study concerns surgically implanted cervical VNS devices, not ear-clip or other non-invasive vagus stimulation products.
Questions About VNS After Failed Epilepsy Surgery
Did CORE-VNS find that prior epilepsy surgery made VNS less effective?
No. Median all-seizure reduction at 36 months was 76.3% in the prior-surgery group and 76.6% in the no-prior-surgery group, with no significant between-group difference across seizure types at 36 months (p = 0.3485).1
Why was seizure freedom lower if seizure reduction was similar?
The prior-surgery group likely had more refractory epilepsy. These patients had already failed an invasive intervention and had higher baseline all-seizure frequency (median 24 per month vs. 15 per month).
A large reduction can still leave residual seizures when the starting burden is heavier.1
Should failed surgery move VNS earlier in the treatment sequence?
It supports considering VNS after failed surgery rather than excluding it. It does not establish VNS should replace resective surgery when a patient has a well-localized, surgically favorable seizure focus.
The sequencing decision still belongs in a comprehensive epilepsy center.
Does this apply to responsive neurostimulation or deep brain stimulation?
No. Responsive neurostimulation and deep brain stimulation are separate implanted neuromodulation strategies with different targets, candidacy rules, and evidence bases.
The Nicolai analysis is about cervical implanted VNS after prior intracranial epilepsy surgery.
Does this paper prove non-invasive tVNS works for epilepsy after surgery?
No. CORE-VNS studied implanted VNS devices.
Transcutaneous VNS uses external stimulation, usually around the ear, and should not be assumed to reproduce the 36-month implanted-device outcomes in this registry.
References
- Nicolai EN et al. Vagus Nerve Stimulation in Failed Epilepsy Surgery: 36 Month Outcomes From the CORE-VNS Study. medRxiv. 2026. doi:10.64898/2026.04.17.26351099
- Hatano K et al. Effects of Vagus Nerve Stimulation following Corpus Callosotomy for Patients with Drug-Resistant Epilepsy. Brain Sciences. 2021;11(11):1395. doi:10.3390/brainsci11111395
- Kawai K et al. Outcome of vagus nerve stimulation for drug-resistant epilepsy: the first 3 years of a prospective Japanese registry. Epileptic Disorders. 2017;19(3):327-338. doi:10.1684/epd.2017.0929
- Suller Marti A et al. Reduction of generalized tonic-clonic seizures following vagus nerve stimulation therapy: CORE-VNS Study 24-month follow-up. Epilepsia. 2025;66(7):2307-2314. doi:10.1111/epi.18371
- Tellez-Zenteno JF, Dhar R, Wiebe S. Long-term seizure outcomes following epilepsy surgery: a systematic review and meta-analysis. Brain. 2005;128(Pt 5):1188-1198. doi:10.1093/brain/awh449
