Lurasidone is approved for schizophrenia and bipolar depression on the strength of acute trials that mostly reported a single number: the change in PANSS total score. A 2026 post-hoc analysis of the JEWEL Phase 3 trial pulls that number apart, looking separately at five symptom domains and at all 30 individual items of the PANSS — and the picture is more textured than the headline allows.1
Lurasidone 40 mg/day improved every domain in 245 acutely psychotic adults with schizophrenia, but the effect sizes ranged from d = 0.44 for positive symptoms down to d = 0.22 for negative symptoms, and the timing of when each domain separated from placebo varied by two weeks. That spread matters more for clinical decisions than the total score does.
Research Highlights
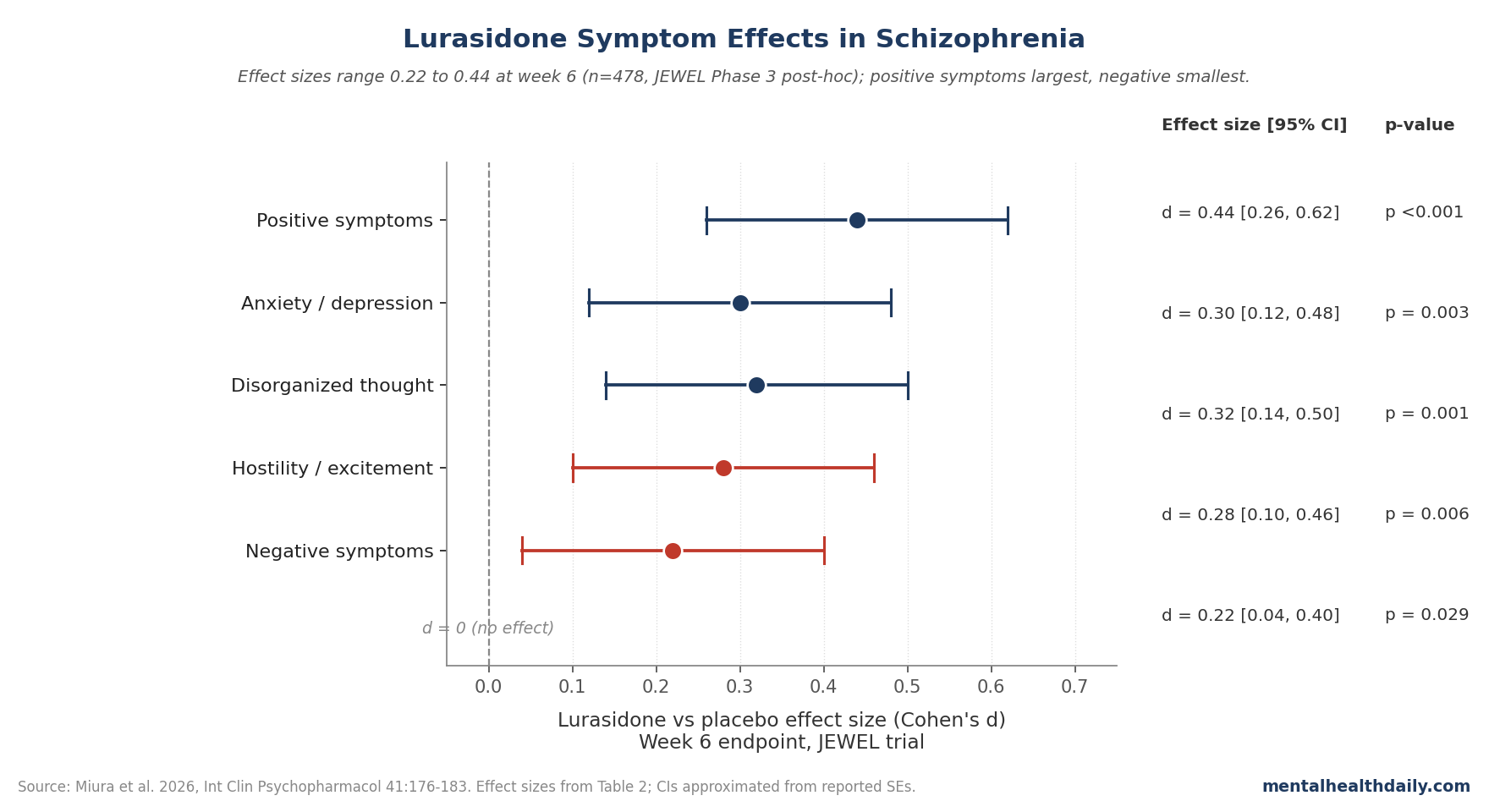
- All five Marder PANSS domains improved significantly with lurasidone 40 mg/day vs placebo at week 6 in the JEWEL trial (n = 478 ITT), with effect sizes of 0.44 (positive), 0.32 (disorganized thought), 0.30 (anxiety/depression), 0.28 (hostility/excitement), and 0.22 (negative).1
- Positive, disorganized, hostility, and anxiety/depression factors separated from placebo by week 2; the negative-symptom factor took until week 4.1
- Five non-positive PANSS items posted endpoint effect sizes > 0.30 with early significance: anxiety, tension, preoccupation, active social avoidance, and conceptual disorganization.1
- The negative-symptom result is hard to interpret because the trial enrolled an acute-positive sample — six of seven negative items had baseline severity ≤3.8, leaving little room for measurable change.1
- The effect-size profile is consistent with the 2015 Loebel pooled analysis (five trials, lurasidone 40–160 mg/day) on three of five domains; disorganized thought and negative symptoms improved less in JEWEL.2
JEWEL Effect Sizes: 0.44 for Positive, 0.22 for Negative, Everything Else in Between
JEWEL was a 6-week, multicenter, double-blind, placebo-controlled fixed-dose trial of lurasidone 40 mg/day conducted across 73 sites in Japan, Ukraine, Russia, Romania, and Poland.3 Of 483 randomized adults with an acute exacerbation of DSM-IV-TR schizophrenia, 245 received lurasidone and 233 received placebo in the intention-to-treat population. Mean baseline PANSS total was approximately 102.
The post-hoc analysis fit a mixed-effects model for repeated measures to each Marder 5-factor and each individual PANSS item, with treatment, visit, treatment-by-visit interaction, and baseline score as covariates.1 Effect sizes were calculated as the week-6 least-squares mean difference divided by the model SD — standard Cohen’s d framing.
Week-6 results for the five Marder domains:
- Positive symptoms: lurasidone −6.6 vs placebo −4.3, d = 0.44, p < 0.001
- Disorganized thought: −3.7 vs −2.5, d = 0.32, p = 0.001
- Anxiety/depression: −3.4 vs −2.5, d = 0.30, p = 0.003
- Hostility/excitement: −2.0 vs −1.2, d = 0.28, p = 0.006
- Negative symptoms: −3.8 vs −2.9, d = 0.22, p = 0.029
The positive-symptom effect size of 0.44 is the largest among recently approved atypical oral antipsychotics for schizophrenia. The Huhn 2019 network meta-analysis of 32 antipsychotics reported positive-symptom effect sizes of 0.47 for asenapine, 0.30 for cariprazine, 0.30 for iloperidone, and 0.17 for brexpiprazole.4 Lurasidone sits closer to the asenapine end than to the brexpiprazole end on positive symptoms specifically.
The other four domains landed in the small-to-moderate range. None reached the 0.5 threshold conventionally used to flag a moderate clinical effect, but all five cleared statistical significance.
Four Factors Separated by Week 2; Negative Symptoms Took Until Week 4
The temporal pattern is what makes this analysis more informative than a single endpoint number.
For four of the five Marder domains — positive, disorganized, hostility, and anxiety/depression — the lurasidone group’s change from baseline was significantly greater than placebo by week 2, and that separation was sustained through every subsequent assessment to week 6.1 The negative-symptom factor only reached significance at week 4, then held through week 6.
That two-week lag on negative symptoms isn’t unique to lurasidone — the broader antipsychotic literature consistently shows positive symptoms responding faster than negative ones — but it’s worth flagging because acute-phase clinicians often interpret a flat 2-week curve as treatment failure. For negative symptoms specifically, a 2-week look is too early.
At the individual-item level, the items that hit significance earliest (week 2) were delusions (P1), hallucinatory behavior (P3), active social avoidance (G16), conceptual disorganization (P2), preoccupation (G15), and anxiety (G2). Suspiciousness (P6) and tension (G4) reached significance at week 3. Later-onset significance (weeks 4 or 5) was observed for unusual thought content (G9), difficulty in abstract thinking (N5), poor attention (G11), hostility (P7), uncooperativeness (G8), and poor impulse control (G14).1
Anxiety, Tension, Preoccupation, and Social Avoidance Posted Effect Sizes > 0.30
The most clinically interesting finding sits in the non-positive items. Five PANSS items outside the positive-symptom factor had endpoint effect sizes greater than 0.30:
- Anxiety (G2): d = 0.34
- Active social avoidance (G16): d = 0.36
- Tension (G4): d = 0.33
- Preoccupation (G15): d = 0.30
- Conceptual disorganization (P2): d = 0.28
Anxiety and tension are particularly notable because the trial used concomitant anxiolytics in 26.7% of the lurasidone group and 31.5% of placebo. A sensitivity analysis restricted to patients not taking anxiolytics or sedatives reproduced the effect-size pattern (anxiety/depression d = 0.26 in that subgroup, comparable to 0.30 in the full sample), so the anxiolytic effect isn’t an artifact of co-medication.1
The active-social-avoidance result deserves a separate flag. Of the seven items in the Marder negative-symptom factor, only G16 had a baseline severity score above 3.8 in this acute-positive sample, and it was the only negative-factor item that showed early (week 2) sustained significance.1 The other six negative items started near floor on this trial’s enrollment criteria, leaving them statistically underpowered to detect an effect.
Mechanistically, lurasidone is unusual among atypical antipsychotics in combining D2 antagonism with potent 5-HT7 antagonism and 5-HT1A partial agonism, with very weak H1 and M1 binding.5 The 5-HT7 and 5-HT1A activity is the receptor pharmacology most often cited for plausible direct effects on anxiety, depression, and cognition independent of D2 blockade.6 Whether the JEWEL findings reflect that mechanism or simply reflect downstream improvement secondary to positive-symptom relief isn’t resolvable from a 6-week trial alone, and the authors are explicit about that limit.
Why the Negative-Symptom Result Probably Understates the Drug
The d = 0.22 effect on negative symptoms is the single number most likely to be misread.
JEWEL’s enrollment criteria required a PANSS subscale score ≥4 (moderate) on at least two of delusions, conceptual disorganization, hallucinations, suspiciousness, or unusual thought content — all positive-symptom items. The trial was, by design, enriched for active positive symptomatology and was not enriched for negative symptoms.1 The consequence is visible in the baseline data: six of seven items in the Marder negative-symptom factor had baseline mean scores ≤3.8 (between minimal and moderate), and 16 of 30 PANSS items overall hit baseline severity in the 2.4–3.8 range.
Items that start near floor are mathematically harder to move — there’s less room for clinically meaningful change, and the mixed-model SE ends up large relative to any signal. All 16 of the items that failed to reach significance at week 6 had low baseline severity. That doesn’t mean lurasidone doesn’t work for those items; it means JEWEL wasn’t designed to test that question.
The 2015 Loebel pooled analysis of five short-term placebo-controlled lurasidone trials (40–160 mg/day) reported a Marder negative-symptom effect size of 0.332 — meaningfully larger than JEWEL’s 0.22. That pooled analysis included higher doses and a broader symptom mix at baseline. The honest read: lurasidone 40 mg/day in a positive-enriched acute trial gives an underpowered look at negative symptoms; the higher-dose, broader-population pooled estimate of d ≈ 0.33 is probably closer to the drug’s actual effect on negative symptoms in mixed-presentation patients.
To genuinely test lurasidone for negative symptoms, the field needs a trial enriched for predominant negative-symptom presentation, with patients who score ≥4 on negative-factor items at entry and minimal active positive symptomatology. No such trial exists for lurasidone as of 2026.
How JEWEL Compares to the 2015 Loebel Pooled Analysis
The Loebel 2015 pooled analysis combined five short-term placebo-controlled trials (n ≈ 1,700) across lurasidone doses of 40, 80, 120, and 160 mg/day, reporting Marder 5-factor effect sizes for the pooled sample.2 JEWEL replicates Loebel almost exactly on three of five domains and underperforms on two:
- Positive symptoms: JEWEL d = 0.44, Loebel d = 0.43 (essentially identical)
- Hostility/excitement: JEWEL d = 0.28, Loebel d = 0.31 (effectively the same)
- Anxiety/depression: JEWEL d = 0.30, Loebel d = 0.31 (essentially identical)
- Negative symptoms: JEWEL d = 0.22, Loebel d = 0.33 (JEWEL lower)
- Disorganized thought: JEWEL d = 0.32, Loebel d = 0.42 (JEWEL lower)
Two methodological differences plausibly explain the gap on the bottom two. Loebel pooled doses up to 160 mg/day; JEWEL used only 40 mg/day. And Loebel pooled multiple trials whose individual enrollment criteria varied, producing a more heterogeneous baseline mix that included patients with higher negative-factor and disorganized-thought severity. The dose argument isn’t trivial — the 40-mg dose is the bottom of lurasidone’s approved range for schizophrenia (40–160 mg/day in the US label), and dose-response curves for atypical antipsychotics on negative and cognitive symptoms tend to shift more between low and moderate doses than positive symptoms do.
Lurasidone also looks reasonable on tolerability. The Pillinger 2020 network meta-analysis of 18 antipsychotics on metabolic outcomes ranked lurasidone among the best on weight gain, glucose, and lipid changes — closer to ziprasidone and aripiprazole than to olanzapine or clozapine.7 That metabolic profile is a meaningful clinical differentiator in long-term schizophrenia care, where antipsychotic-driven cardiometabolic disease is one of the largest contributors to the 15–20 year mortality gap.
Limitations of the JEWEL Post-Hoc
Single dose. Only lurasidone 40 mg/day was tested. Whether higher doses would produce larger effects on negative or disorganized-thought factors is not addressable from this dataset, and the comparison with Loebel’s pooled higher-dose estimates suggests they probably would.
Floor effects on 16 items. The acute-positive enrollment criteria left 16 of 30 PANSS items with baseline severity in the 2.4–3.8 range. Negative symptoms, cognition, and mood items took the brunt. Conclusions about lurasidone’s efficacy on those items from this trial alone would be unsafe.
Six-week duration. Schizophrenia is a chronic condition, and the most clinically important outcomes — relapse, functional recovery, employment, durable cognitive improvement — require months to years to assess. Earlier randomized withdrawal data suggest lurasidone reduces relapse vs placebo over 12 months,8 but the JEWEL post-hoc itself can’t speak to long-term function.
Excluded screen-to-baseline improvers. JEWEL excluded patients with ≥20% PANSS reduction between screening and baseline, which removes a portion of natural-history responders. That sharpens the placebo-vs-drug contrast but slightly shifts the sample toward less-responsive presentations and limits generalizability.
Causal attribution at the item level. Whether improvement on anxiety, tension, preoccupation, and social avoidance reflects direct lurasidone effects via 5-HT7 / 5-HT1A activity or indirect downstream effects of positive-symptom relief is not resolvable from this trial. The receptor pharmacology supports a direct mechanism; the trial design can’t confirm it.
What This Means for Lurasidone Use in Schizophrenia
For acute-phase positive symptoms in schizophrenia, lurasidone 40 mg/day produces a placebo-adjusted effect size around 0.44 by week 6, with separation visible by week 2 — comparable to asenapine and larger than cariprazine, iloperidone, or brexpiprazole on the positive-symptom domain in the Huhn network meta-analysis.4
For anxiety, tension, and social avoidance during acute treatment, the JEWEL data argue lurasidone has measurable benefit (d > 0.30 on each of those individual items) that is not attributable to concomitant anxiolytic use. Whether the mechanism is direct (5-HT1A/5-HT7) or indirect (downstream of positive-symptom improvement) is open, but the clinical effect is present either way.
For negative symptoms specifically, JEWEL is underpowered by design, and the d = 0.22 estimate should be read against the larger d ≈ 0.33 from Loebel’s higher-dose pooled analysis. The honest framing: lurasidone has a real but modest negative-symptom signal at higher doses; the size at 40 mg/day in an acute-positive sample is not the right read.
For long-term tolerability, lurasidone’s metabolic profile is a meaningful advantage over olanzapine, quetiapine, and risperidone, particularly for younger patients facing decades of treatment. That advantage isn’t visible in a 6-week PANSS analysis but is the central reason lurasidone is often the right pick when the metabolic and cardiovascular risk is salient.
Common Questions About Lurasidone in Schizophrenia
How does lurasidone compare to other second-generation antipsychotics for positive symptoms?
In the Huhn 2019 network meta-analysis of 32 antipsychotics, lurasidone’s effect size on positive symptoms in acute schizophrenia was 0.44 — near the top of recently approved oral atypicals, similar to asenapine (0.47) and well above brexpiprazole (0.17), cariprazine (0.30), and iloperidone (0.30).4 Olanzapine and amisulpride outperform on raw efficacy but at substantial metabolic cost.
Does lurasidone work for negative symptoms in schizophrenia?
JEWEL reported a Marder negative-symptom effect size of 0.22 at 40 mg/day, but the trial was enriched for active positive symptoms with 6 of 7 negative items starting near floor severity, so it underpowers the question. The 2015 Loebel pooled analysis across higher doses reported d = 0.33, which is probably closer to the real signal.1,2 No trial enriched for predominant negative symptoms exists for lurasidone.
How fast does lurasidone start working?
Positive symptoms, hostility, anxiety/depression, and disorganized thought all separated from placebo by week 2 in JEWEL and held that separation through week 6. Negative symptoms took until week 4. At the individual-item level, delusions, hallucinatory behavior, social avoidance, conceptual disorganization, preoccupation, and anxiety were all significantly improved by week 2.1
Is lurasidone a good choice for someone with schizophrenia plus anxiety?
The PANSS anxiety item (G2) showed an endpoint effect size of d = 0.34 in JEWEL, and tension (G4) showed d = 0.33, both with early significance. The effect was preserved in patients not taking concomitant anxiolytics, so it isn’t an artifact of co-medication. Receptor-level rationale (5-HT1A partial agonism, 5-HT7 antagonism) supports a plausible direct mechanism.1,6
What dose was tested in JEWEL?
Only 40 mg/day, the bottom of lurasidone’s approved schizophrenia dose range (40–160 mg/day). Higher doses were not tested in this trial. The effect-size gap between JEWEL and Loebel’s higher-dose pooled analysis suggests dose may matter more for disorganized thought and negative symptoms than for positive symptoms.1,2
How does lurasidone compare on weight gain and metabolic side effects?
In the Pillinger 2020 network meta-analysis of 18 antipsychotics on metabolic outcomes, lurasidone was among the best, with weight, glucose, and lipid effects closer to ziprasidone and aripiprazole than to olanzapine or clozapine.7 That advantage is one of the strongest practical reasons to consider lurasidone in younger patients facing long-term treatment.
Does lurasidone help cognition in schizophrenia?
Harvey 2013 reported neurocognitive improvement on lurasidone in a placebo- and active-controlled trial with a 6-month extension.9 JEWEL’s disorganized-thought factor, which contains several cognitively loaded items (poor attention, abstract thinking, conceptual disorganization), improved with d = 0.32. Whether this reflects direct cognitive enhancement or downstream improvement from positive-symptom relief is not settled.
Should lurasidone be taken with food?
Yes — absorption increases roughly two-fold with a meal of at least 350 calories, and the FDA label specifies that lurasidone should be taken with food. Taken on an empty stomach, plasma levels drop substantially and efficacy drops with them. This is a more dose-relevant practical issue than for most other atypical antipsychotics.
References
- The effects of lurasidone on five symptom domains and individual items in schizophrenia: post-hoc analysis from Phase 3 study. Miura I, Takai K, Toya S, Iyo M. Int Clin Psychopharmacol. 2026;41(3):176–183. doi:10.1097/YIC.0000000000000609
- Efficacy of lurasidone across five symptom dimensions of schizophrenia: pooled analysis of short-term, placebo-controlled studies. Loebel A, Cucchiaro J, Silva R, Mao Y, Xu J, Pikalov A, Marder SR. Eur Psychiatry. 2015;30(1):26–31. doi:10.1016/j.eurpsy.2014.08.001
- Efficacy and safety of lurasidone in acutely psychotic patients with schizophrenia: a 6-week, randomized, double-blind, placebo-controlled study. Iyo M, Ishigooka J, Nakamura M, et al. Psychiatry Clin Neurosci. 2021;75(7):227–235. doi:10.1111/pcn.13221
- Comparative efficacy and tolerability of 32 oral antipsychotics for the acute treatment of adults with multi-episode schizophrenia: a systematic review and network meta-analysis. Huhn M, Nikolakopoulou A, Schneider-Thoma J, et al. Lancet. 2019;394(10202):939–951. doi:10.1016/S0140-6736(19)31135-3
- Pharmacological profile of lurasidone, a novel antipsychotic agent with potent 5-hydroxytryptamine 7 (5-HT7) and 5-HT1A receptor activity. Ishibashi T, Horisawa T, Tokuda K, et al. J Pharmacol Exp Ther. 2010;334(1):171–181. doi:10.1124/jpet.110.167346
- Serotonin 5-HT1A receptors as targets for agents to treat psychiatric disorders: rationale and current status of research. Celada P, Bortolozzi A, Artigas F. CNS Drugs. 2013;27(9):703–716. doi:10.1007/s40263-013-0071-0
- Comparative effects of 18 antipsychotics on metabolic function in patients with schizophrenia: a systematic review and network meta-analysis. Pillinger T, McCutcheon RA, Vano L, et al. Lancet Psychiatry. 2020;7(1):64–77. doi:10.1016/S2215-0366(19)30416-X
- A double-blind, placebo-controlled, randomized withdrawal study of lurasidone for the maintenance of efficacy in patients with schizophrenia. Tandon R, Cucchiaro J, Phillips D, et al. J Psychopharmacol. 2016;30(1):69–77. doi:10.1177/0269881115620460
- Effect of lurasidone on neurocognitive performance in patients with schizophrenia: a short-term placebo- and active-controlled study followed by a 6-month double-blind extension. Harvey PD, Siu CO, Hsu J, Cucchiaro J, Maruff P, Loebel A. Eur Neuropsychopharmacol. 2013;23(11):1373–1382. doi:10.1016/j.euroneuro.2013.08.003
