A 2026 meta-analysis of 56 controlled trials found the clearest gamma-frequency neuromodulation signal in schizophrenia: overall symptoms improved vs. control (g = −0.46), global cognition improved (g = 0.55), and MDD showed a smaller depression effect than the schizophrenia symptom estimate (g = −0.34). Bipolar disorder and autism stayed directional but nonsignificant, and the paper’s modality hypothesis was not confirmed.
Research Highlights
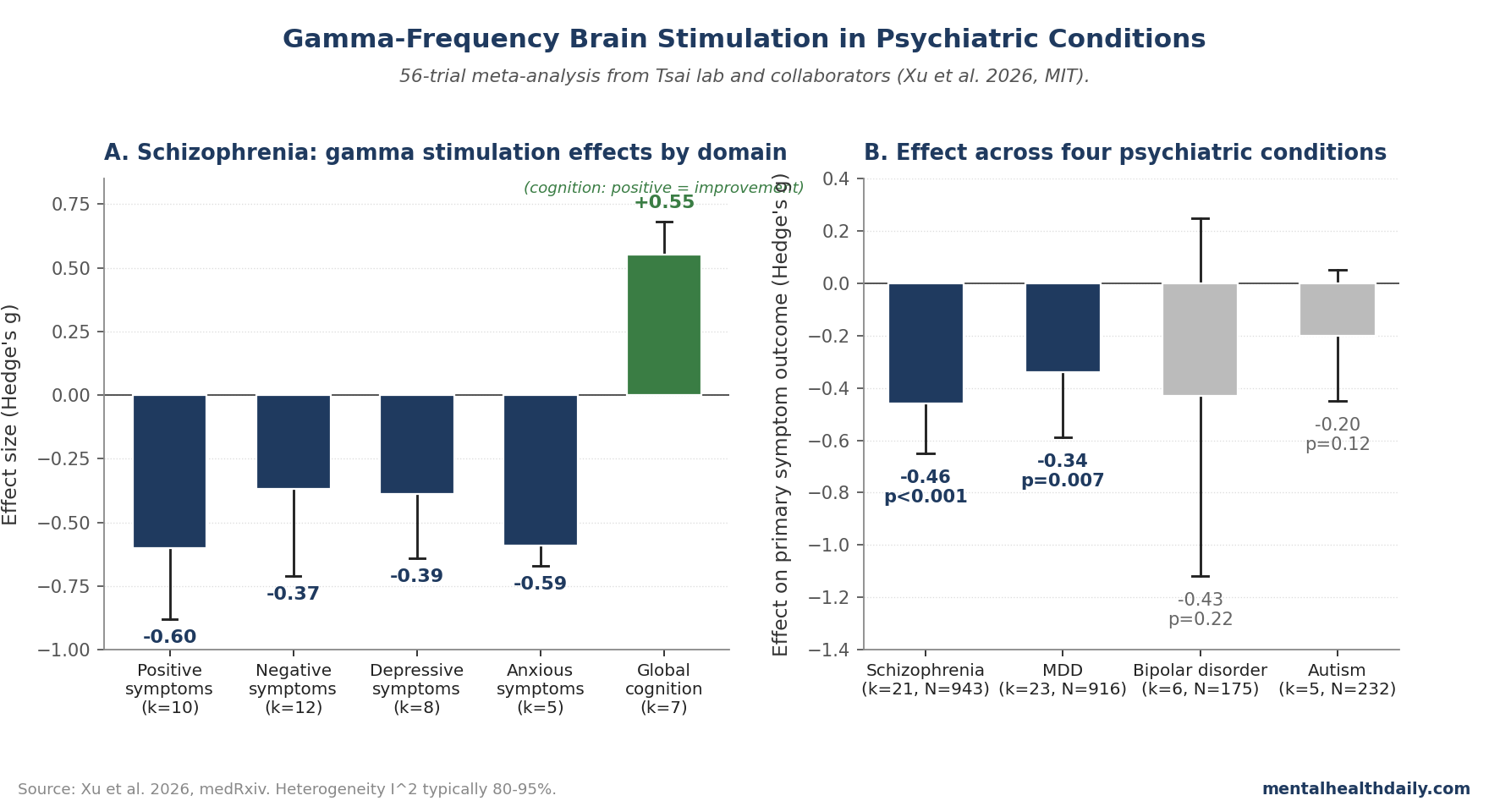
- Schizophrenia had the strongest pooled signal: overall symptoms improved (g = −0.46, 95% CI −0.65 to −0.26) and global cognition improved (g = 0.55, 95% CI 0.42 to 0.68). The effect survived outlier removal, but heterogeneity stayed high.1
- The Xu 2026 meta-analysis pooled 56 controlled trials (n = 943 schizophrenia, 916 MDD, 175 bipolar disorder, 232 autism). Most studies used 50 Hz theta-burst TMS, and contrary to the prediction, modality did not explain clinical efficacy across schizophrenia and MDD.1
- In schizophrenia, gamma stimulation produced moderate effects on positive symptoms (g = −0.60), negative symptoms (g = −0.37), and cognition (g = 0.55). The 50 Hz subgroup outperformed 30–40 Hz; treatment durations of 2–3 weeks or longer outperformed shorter protocols.1
- In MDD, the depression effect was small (g = −0.34, p = 0.007), with publication-bias risk detected on Egger’s test (p = 0.004). After trim-and-fill correction the signal remained but shrank.1
- Bipolar disorder (g = −0.43, p = 0.22) and autism (g = −0.20, p = 0.12) did not reach statistical significance. Directional estimates leaned toward benefit, but confidence intervals crossed no effect.1
Gamma oscillations are generated locally by GABAergic interneurons and tied to perceptual integration, working memory, and large-scale circuit coordination.2 Reduced gamma power and impaired auditory steady-state responses are among the most replicated EEG findings in schizophrenia, with similar gamma-band disruptions in bipolar disorder, autism, and Alzheimer’s disease.
Xu et al. — from Li-Huei Tsai’s lab at MIT, the group that pioneered 40 Hz GENUS sensory stimulation in mice3 — gives the psychiatric side of the story. the calibrated interpretation is not “gamma works” or “gamma fails”; it is that schizophrenia has the most coherent signal, MDD has a smaller and bias-sensitive signal, and the delivery method did not behave the way the researchers expected.1
Xu 2026: 56 Trials, Two Disorders Where Effects Land
The 2026 systematic review followed PRISMA-style methodology, searching five databases through April 2025 and filtering to controlled trials of gamma-frequency stimulation (30–50 Hz) in adult psychiatric populations. After screening, 56 studies entered quantitative synthesis with the following totals: schizophrenia n = 943, MDD n = 916, bipolar disorder n = 175, autism n = 232.1
Stimulation modalities were dominated by theta-burst TMS protocols. iTBS (intermittent theta-burst, excitatory) accounted for 64% of studies, cTBS (continuous, inhibitory) 21%, tACS 7%, bilateral TBS 5%, sensory stimulation 2%, and tVNS 2%.
Stimulation frequencies were 50 Hz (88% of studies), 40 Hz (9%), and 30 Hz (3%). The “gamma” label is broad — iTBS at 50 Hz delivers 50 Hz bursts at theta frequency (5 Hz), which is mechanistically different from sustained 40 Hz tACS, but both fall under gamma-frequency neuromodulation in this synthesis.
The broad label creates a calibration problem because one of Xu et al.’s hypotheses was partly wrong. Contrary to the initial prediction, stimulation modality did not explain clinical efficacy across schizophrenia and MDD; frequency and treatment duration carried the clearer moderator signal.1
Schizophrenia: Moderate Effects on Symptoms and Cognition
The schizophrenia data were the strongest in the meta-analysis. Across 21 studies pooling total PANSS scores, gamma stimulation outperformed sham with a moderate effect size (g = −0.46, 95% CI −0.65 to −0.26, p < 0.001), though heterogeneity was severe (I² = 96.0%).1
Removing eight outlier studies preserved the signal (g = −0.46, 95% CI −0.59 to −0.33, p < 0.001) with reduced but still high heterogeneity (I² = 80.2%).
Symptom-domain breakdowns:1
- Positive symptoms: g = −0.60, p < 0.001 (k = 10 studies). Moderate effect.
- Negative symptoms: g = −0.37, p = 0.03 (k = 12). Small-to-moderate effect.
- Depressive symptoms in SZ: g = −0.39, p < 0.001 (k = 8).
- Anxiety symptoms in SZ: g = −0.59, p < 0.001 (k = 5). Moderate effect with relatively low heterogeneity (I² = 24.8%).
- Global cognitive function: g = 0.55, p < 0.001 (k = 7). Moderate-sized improvement on MATRICS Consensus Cognitive Battery and MoCA scores.
The cognitive finding is particularly notable. Antipsychotics consistently fail to address cognitive deficits in schizophrenia, which are among the strongest predictors of functional impairment.4
A moderate cognitive improvement from non-invasive stimulation, if replicable in larger trials, would address a treatment gap that has resisted decades of pharmacological development.
The schizophrenia result also fits a 2024 network meta-analysis of theta-burst stimulation protocols. Kishi et al. reported that left dorsolateral prefrontal iTBS reduced overall symptom severity (SMD = −0.57), with larger effects on negative (SMD = −0.89), depressive (SMD = −0.70), and anxious symptoms (SMD = −0.58) than on positive symptoms.10
Xu et al. found a stronger positive-symptom estimate than that earlier TBS review, but both analyses point to schizophrenia as the psychiatric condition where the gamma-range TBS-heavy evidence is most developed.
Subgroup analyses identified two parameters that moderated treatment response:1
- Stimulation frequency. 50 Hz protocols outperformed 30 Hz and 40 Hz (50 Hz: g = −0.54, k = 18 studies; 30 Hz: g = +0.06, k = 1; 40 Hz: g = +0.01, k = 2). The 30 Hz and 40 Hz subgroups are very small and shouldn’t be over-interpreted, but the 50 Hz finding is robust.
- Treatment duration. 2–3 weeks or longer outperformed shorter protocols (2–3 weeks: g = −0.61, k = 8; >3 weeks: g = −0.50, k = 8; <1 week: g = +0.06, k = 1; 1–2 weeks: g = −0.23, k = 4). Cumulative rather than acute effects.
Stimulation modality (iTBS vs cTBS vs tACS) and pulses-per-session didn’t significantly moderate effects, though most studies used iTBS (62%) so the modality comparisons are dominated by within-iTBS variation.
MDD: A Small Effect Tempered by Publication Bias
23 studies pooled depressive symptoms in MDD with active gamma stimulation vs. sham control. The overall effect was small but significant (g = −0.34, 95% CI −0.59 to −0.09, p = 0.007), with very high heterogeneity (I² = 95.3%).1
Publication bias was detectable. Egger’s test was significant (p = 0.004), and visual inspection of the funnel plot showed asymmetry consistent with selective reporting.
The trim-and-fill method shrank the effect estimate but didn’t eliminate it. After excluding nine outlier studies, the pooled effect remained statistically significant (g = −0.30, 95% CI −0.42 to −0.19, p < 0.001) with persistent high heterogeneity (I² = 76.9%).
Subgroup analyses didn’t identify any single moderator (modality, frequency, duration, pulses-per-session) that reliably explained the heterogeneity in MDD — possibly because the underlying mechanisms of gamma-stimulation effects in depression are more variable, or because trials are smaller and dose-response signals are noisier.
The effect size is in the same range as the wider TBS-for-depression literature. A 2024 network and pairwise meta-analysis of 23 randomized trials (n = 960) framed TBS as an efficient depression neuromodulation family, not as a uniquely gamma-specific mechanism.11
Xu’s MDD estimate therefore looks less like a new standalone gamma finding and more like a small effect inside the broader rTMS/TBS treatment class.
Bipolar and Autism: Trends Without Significance
Bipolar disorder and autism analyses had smaller pooled samples and weaker signals.1
Bipolar (k = 6 studies, n = 175): Gamma stimulation produced a positive directional trend on depressive symptoms (g = −0.43, 95% CI −1.12 to 0.25, p = 0.22), with severe heterogeneity (I² = 93.9%). Confidence intervals span “substantial benefit” to “no effect”; the sample is small and the data don’t support a clinical claim.
Autism (k = 5 studies, n = 232): Gamma stimulation showed a small directional trend on social-impairment outcomes (g = −0.20, 95% CI −0.45 to 0.05, p = 0.12), with very high heterogeneity (I² = 90.6%). Again, no statistically significant effect.
For both conditions, the published evidence base is too small to conclude either way, and any future application to clinical populations would require purpose-designed trials with larger samples and standardized protocols.
Why Gamma? The Neurobiological Rationale
Gamma oscillations emerge from local circuit interactions between fast-spiking parvalbumin-positive (PV) interneurons and excitatory pyramidal neurons.2 The PV interneuron-to-pyramidal cell synchronization produces the 30–100 Hz rhythm that supports working memory, attentional gating, and large-scale neural communication.
In schizophrenia, gamma deficits are among the most consistently replicated electrophysiological findings:
- Reduced gamma power during working-memory and attention tasks.6
- Impaired 40 Hz auditory steady-state response (ASSR), a transdiagnostic marker that also appears in Alzheimer’s disease and bipolar disorder.
- Reduced inhibitory control tied to PV interneuron dysfunction and NMDA-receptor hypofunction, which are upstream contributors to gamma generation.7
The therapeutic logic is straightforward: if gamma deficits contribute to cognitive and symptomatic impairment, externally entraining gamma rhythms via TMS, tACS, or sensory stimulation might partially restore normal circuit function. The Tsai lab’s GENUS work in animal models showed that 40 Hz light and sound entrainment reduces amyloid pathology and improves cognition in mouse Alzheimer’s models, motivating broader interest in gamma neuromodulation across psychiatric conditions.3
Whether the gamma-stimulation effects in the human SZ literature reflect direct rhythm restoration, downstream effects on neuroplasticity, or generic rTMS effects that happen to be delivered at gamma frequencies is mechanistically unresolved.
Evidence Limits for Gamma Neuromodulation
Three calibrations matter when interpreting media coverage.
- “Gamma neuromodulation” includes very different interventions. The Xu meta-analysis pools 50 Hz iTBS (a TMS protocol delivered in patterned bursts), 40 Hz tACS (sustained transcranial alternating current), and sensory stimulation (audiovisual entrainment). These mechanisms differ substantially. Coverage that conflates them — particularly conflating the broader iTBS literature with the Tsai-lab 40 Hz GENUS work for Alzheimer’s — misrepresents the evidence base.
- The Alzheimer’s evidence base is more limited than headlines suggest. The Xu 2026 paper covered psychiatric conditions, not AD. The 40 Hz GENUS literature in Alzheimer’s is mechanistically interesting but has produced mixed clinical results in human trials. A separate meta-analysis Xu cites found no significant cognitive benefit of 40 Hz light-and-sound stimulation in AD patients, despite large effects on brain structural measures.8 Cognito Therapeutics’ phase 3 OVERTURE trial of 40 Hz audiovisual stimulation reported mixed primary outcomes, and the clinical case is not yet settled.
- Heterogeneity is severe and the protocol space is huge. I² values around 90% mean trials disagree wildly on the size of the benefit. The 56-trial pooled estimates Xu provides represent the average across heterogeneous protocols; an individual patient’s likely response depends on their specific stimulation protocol, treatment duration, and underlying clinical phenotype. The treatment space is far from optimized.
Limitations of the Xu Synthesis
Study quality is mixed. 10% of included studies were rated high risk of bias, 62% had some concerns, and only 28% were low risk on the RoB 2 tool. Most concerns related to absence of preregistered protocols and the small-trial publication patterns common in this literature.
Heterogeneity is essentially total in some analyses. I² values above 90% indicate that trials disagree wildly on how big the effect is. This isn’t a fixable analytical problem; it reflects genuine differences in protocols, populations, outcome measures, and sample sizes.
The pooled estimates should be treated as ranges of plausible effects rather than precise predictions.
Publication bias was detected in the MDD analysis. Egger’s test was significant for the MDD pool, and trim-and-fill correction shrank the effect estimate. Small negative trials likely went unpublished, inflating the apparent benefit.
The corrected estimate remains positive but smaller than the headline figure.
Most trials are short and small. The modal study lasted 1–3 weeks with 20–60 participants. Long-term outcomes (3-month, 6-month, 12-month durability) are sparse, and the maintenance treatment paradigm hasn’t been adequately tested.
Dose-response work is also limited; the moderator analyses Xu performed were based on coarse parameter categories.
The disorder boundaries are loose. SZ analyses included some treatment-resistant samples and some early-stage samples; MDD analyses ranged from mild to severe symptoms. Subgroup stratification by severity, duration, and prior treatment response would refine the practical translation, but most individual trials were too small to support these analyses.
Practical Implications for Patients and Clinicians
The clinical translation of gamma-frequency neuromodulation depends substantially on which condition and which protocol is being considered.
For schizophrenia, iTBS targeting the dorsolateral prefrontal cortex or related sites at 50 Hz is the most-studied protocol in this synthesis. The pooled effect sizes (positive symptoms g = −0.60, negative g = −0.37, cognition g = 0.55) sit in the moderate-effect range and are larger than typical rTMS-for-MDD effects.
The 2–3 week treatment duration emerged as a meaningful moderator, suggesting cumulative rather than acute effects. iTBS is FDA-cleared for treatment-resistant MDD but not specifically for schizophrenia; clinical use for SZ remains investigational at most US centers.
For MDD, gamma-frequency rTMS sits within the broader rTMS-for-depression literature, which is well-established and FDA-cleared. Patients with treatment-resistant MDD who are candidates for rTMS and who have access to iTBS protocols at 50 Hz are not doing something experimental; iTBS at 50 Hz is one of the standard protocols.
The Xu data argue this protocol class produces small but real benefits in pooled analysis.
For bipolar disorder and autism, the evidence base is too thin to support specific clinical recommendations. Future trials with larger samples, pre-registered protocols, and standardized outcome measures are needed before population-level clinical claims are warranted.
Questions About Gamma Brain Stimulation
Does Xu 2026 show that one gamma-stimulation modality is best?
No. The paper expected stimulation modality to moderate response, but modality-specific differences did not emerge across schizophrenia and MDD.
The stronger parameter signal was frequency and duration: 50 Hz and 2–3 weeks or longer looked better in schizophrenia.1
Does this apply to 40 Hz light-and-sound devices for Alzheimer’s disease?
Only indirectly. Xu et al. studied psychiatric conditions, and the evidence base was mostly theta-burst TMS rather than sensory GENUS.
Separate Alzheimer disease reviews describe 40 Hz auditory-visual stimulation as promising but still in need of larger, more robust clinical validation.8
Does this work for cognition in schizophrenia?
The Xu data show a moderate-sized cognitive benefit (g = 0.55, k = 7 trials) on global cognitive measures such as MATRICS and MoCA. Durability and functional translation remain open, but cognition is one of the clearer schizophrenia signals in this meta-analysis.1
References
- Gamma Neuromodulation Provides Therapeutic Potential in Neuropsychiatry: A Systematic Review and Meta-Analysis. Xu M et al. medRxiv. 2026 (preprint). doi:10.64898/2026.04.10.26350641
- Cortical gamma rhythms: a strategic communication mechanism. Buzsáki G, Wang XJ. Annual Review of Neuroscience. 2012;35:203-225. doi:10.1146/annurev-neuro-062111-150444
- Gamma frequency entrainment attenuates amyloid load and modifies microglia. Iaccarino HF, Singer AC, Martorell AJ et al. (Tsai lab). Nature. 2016;540(7632):230-235. doi:10.1038/nature20587
- Cognitive impairment in schizophrenia. Green MF, Kern RS, Heaton RK. Schizophrenia Research. 2004;72(1):41-51. doi:10.1016/j.schres.2004.09.009
- Repetitive transcranial magnetic stimulation for the acute treatment of major depressive episodes: a systematic review with network meta-analysis. Brunoni AR et al. JAMA Psychiatry. 2017;74(2):143-152. doi:10.1001/jamapsychiatry.2016.3644
- Gamma synchrony in schizophrenia. Uhlhaas PJ, Singer W. Schizophrenia Bulletin. 2010;36(4):683-691. doi:10.1093/schbul/sbq038
- Cortical interneurons and schizophrenia. Lewis DA, Hashimoto T, Volk DW. Nature Reviews Neuroscience. 2005;6:312-324. doi:10.1038/nrn1648
- 40 Hz light and sound stimulation for Alzheimer’s disease: a systematic review and meta-analysis. Tian J et al. Alzheimer’s & Dementia: Translational Research & Clinical Interventions. 2025;11(1):e12530. doi:10.1002/trc2.12530
- Safety, ethical considerations, and application guidelines for the use of transcranial magnetic stimulation in clinical practice and research. Rossi S et al. Clinical Neurophysiology. 2009;120(12):2008-2039. doi:10.1016/j.clinph.2009.08.016
- Theta Burst Stimulation Protocols for Schizophrenia: A Systematic Review and Network Meta-Analysis. Kishi T et al. JAMA Network Open. 2024;7(10):e2441159. doi:10.1001/jamanetworkopen.2024.41159
- Theta burst stimulation for depression: a systematic review and network and pairwise meta-analysis. Kishi T et al. Molecular Psychiatry. 2024;29:3893-3899. doi:10.1038/s41380-024-02630-5
