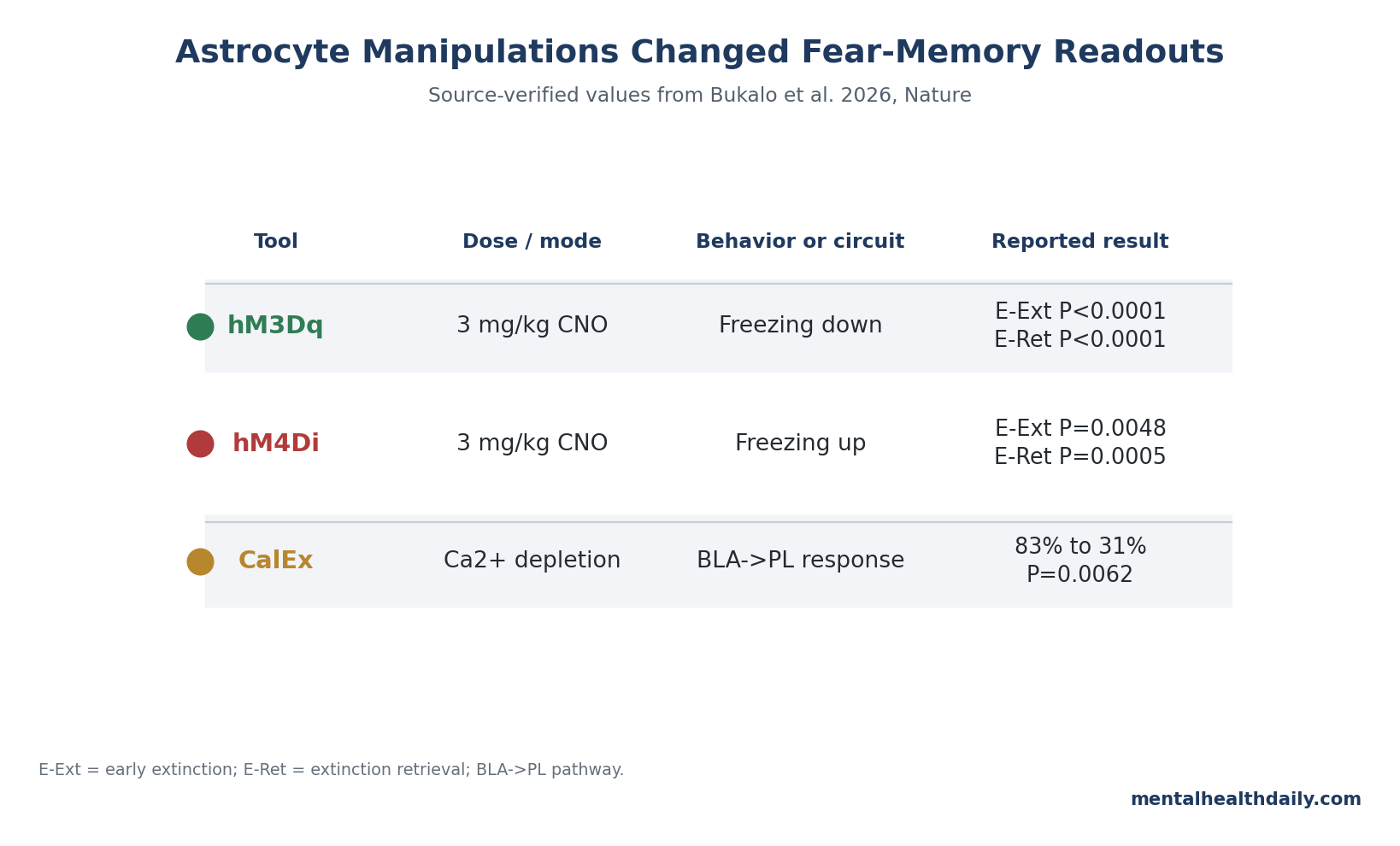
A 2026 Nature mouse study found that basolateral amygdala astrocyte Ca2+ signals tracked fear retrieval and extinction, then showed causality: 3 mg/kg CNO manipulations of astrocyte DREADDs pushed early-extinction freezing in opposite directions while both disrupting later extinction retrieval.1
Research Highlights
- Astrocytes tracked fear state: after 3 tone-shock pairings and 2 days of 25 cue-only extinction trials, CS-related astrocyte Ca2+ activity fell from early to late extinction (P = 0.0231) and recovered during fear renewal (P < 0.0001 vs. late extinction).1
- hM3Dq lowered retrieval freezing: 3 mg/kg CNO before extinction reduced freezing in hM3Dq mice vs. controls at extinction block 1 (P < 0.0001), block 2 (P = 0.0067), and extinction retrieval (P < 0.0001).1
- hM4Di pushed the opposite retrieval direction: the same 3 mg/kg CNO timing increased freezing in hM4Di mice vs. controls at extinction block 1 (P = 0.0048), block 2 (P = 0.0250), and extinction retrieval (P = 0.0005).1
- Neuronal coding weakened: astrocyte hM3Dq actuation lowered estimated CS-related information per BLA neuron from 0.31 in vehicle mice to 0.20 in CNO mice, and decoder coefficients correlated with freezing (r = 0.70, P = 0.039).1
- Output to cortex also depended on astrocytes: chronic CalEx depletion reduced CS-responsive BLA-to-prelimbic neurons from 83% in controls to 31% in CalEx mice (P = 0.0062), narrowing the circuit claim to a measurable amygdala-prefrontal readout.1
Astrocytes are star-shaped glial cells that contact synapses, regulate extracellular chemistry, and signal through slower Ca2+ waves rather than fast action potentials. Calling them “support cells” is still partly true, but the Bukalo et al. paper makes the support active: in the basolateral amygdala (BLA), astrocyte Ca2+ dynamics helped fear-related neuronal representations form, update, and reach a prefrontal output pathway.
Fear extinction does not erase a fear memory. It builds new safety learning that can suppress the old threat response in the right context, which is why extinction biology matters for exposure-based treatments for PTSD, phobias, and anxiety disorders.6
The clinical temptation is to jump from “astrocytes shape extinction in mice” to “astrocyte drugs will improve PTSD therapy.” The stronger read is more precise: this is unusually direct mouse circuit evidence that astrocytes participate in the BLA machinery that exposure therapy tries to recruit, not proof that an astrocyte-targeted intervention works in patients.
Astrocyte Ca2+ Signals Tracked Fear Retrieval Across 25-Trial Extinction Sessions
Bukalo et al. trained male mice with 3 pairings of a tone-conditioned stimulus and footshock, then tested extinction across 2 days of repeated cue-only presentations: 25 conditioned-stimulus trials on the first extinction day and 25 more on the second. Early extinction captured fear-memory retrieval, late extinction captured reduced freezing after repeated safety exposure, and later tests measured extinction retrieval and renewal.
The first result was state tracking. Cytosolic GCaMP6f expression was selective for astrocytes, with around 2% neuronal expression after viral targeting, and astrocyte Ca2+ responses followed the learned threat state rather than merely responding to any tone.
During conditioning, astrocytes responded strongly to the footshock, not the tone. During early extinction, after the tone had become threatening, astrocytes responded to the tone.
Those tone-linked astrocyte responses then moved with behavior. CS-related astrocyte Ca2+ activity decreased from early extinction to late extinction (P = 0.0231) and from early extinction to extinction retrieval (P = 0.0262), then recovered during fear renewal (P < 0.0001 vs. late extinction; P = 0.0002 vs. extinction retrieval).
Across test sessions, astrocyte Ca2+ transients correlated tightly with freezing (r = 0.97, P = 0.0261).1
That pattern matters because it separates the paper from a weak “astrocytes lit up during a task” story. The signal was more than general arousal, immobility, or a fluorescence artifact.
Control conditions omitting the shock or tone showed minimal CS-related activity, and non-extinguished mice did not show the same trial-wise decline. A second imaging approach, 2-photon Ca2+ imaging of astrocyte events, also decoded CS presentation from individual astrocyte-event activity in early extinction (110 events from 6 mice; P = 0.0265).
3 mg/kg CNO Made Astrocyte Tools Push Retrieval in Opposite Directions
Tracking is not causation, so the paper then used astrocyte-targeted manipulations. The key detail is that the DREADD tools were not simple “on” switches.
hM3Dq actuation produced an early Ca2+ surge and then a prolonged low-activity period lasting at least 2 hours; hM4Di increased Ca2+ transients in the relevant BLA experiments. The researchers used 3 mg/kg clozapine-N-oxide (CNO) before extinction training to perturb astrocyte Ca2+ dynamics during retrieval and extinction.
With hM3Dq, pre-extinction CNO reduced CS-related freezing compared with viral controls during early extinction. The group difference was strong at extinction block 1 (P < 0.0001), remained present at block 2 (P = 0.0067), and persisted at extinction retrieval (P < 0.0001).
The same manipulation also abolished CS-related astrocyte Ca2+ responses during extinction (P < 0.0001 at minute 35).1
With hM4Di, the direction reversed during retrieval: pre-extinction CNO increased freezing at extinction block 1 (P = 0.0048) and block 2 (P = 0.0250), and the higher-freezing pattern was still detectable at extinction retrieval (P = 0.0005). Astrocyte Ca2+ activity during extinction was also increased by the hM4Di manipulation (P < 0.0001 at minute 35).
The important calibration is that these opposite retrieval effects did not mean one astrocyte state was simply “good” and the other “bad.” Both perturbations impaired extinction memory formation. In plain English: disrupting the timing and level of BLA astrocyte Ca2+ signaling changed immediate fear expression in opposite directions, but extinction learning seemed sensitive to either kind of astrocyte disruption.
Disrupted Astrocytes Made Amygdala Neuronal Codes Less Informative
The paper becomes more than a behavior study because the researchers paired astrocyte manipulation with neuronal imaging. If astrocytes matter to fear retrieval, the neuronal code in the BLA should change when astrocyte Ca2+ signaling is perturbed.
That is what the one-photon neuronal imaging experiments tested.
After hM3Dq actuation during fear-memory retrieval, fewer neurons showed the normal conditioning-related increase in CS response. A population decoder could distinguish CNO-treated mice from vehicle-treated mice using neuronal activity from positively CS-modulated neurons (P = 0.0264).
More importantly, the decoder that tried to identify CS presentation from neuronal ensembles performed worse in the CNO group across ensemble sizes.
The paper quantified this as lower information carried by individual BLA neurons: 0.20 in the CNO group vs. 0.31 in the vehicle group. The mouse-level decoder coefficient also tracked behavior, correlating with CS-related freezing on fear retrieval (r = 0.70, P = 0.039).
That gives the behavioral change a circuit-level read: when astrocyte signaling was disrupted, the BLA neuronal representation of the threat cue became less informative, and the weaker code related to lower freezing.
This is the point where older neuron-only models start to look incomplete. The result does not say astrocytes store fear memories by themselves.
It says BLA astrocyte Ca2+ dynamics helped neurons express and update the representation that behavior reads out as fear retrieval and extinction.
BLA-to-Prelimbic Cortex Readout Dropped From 83% to 31%
Fear expression also depends on outputs from the amygdala to downstream regions. Bukalo et al. therefore tested a BLA-to-prelimbic cortex pathway, using CalEx to chronically deplete astrocyte Ca2+ and optically tagged recordings to identify BLA neurons projecting to the prelimbic prefrontal cortex.
The key circuit number is stark: during early extinction, 83% of control BLA-to-prelimbic phototagged neurons were CS-responsive, compared with 31% in CalEx mice (P = 0.0062). Freezing-related unit activity was also altered (P = 0.0275).
Non-tagged neurons did not show the same pattern, which keeps the claim anatomically sharper than a generic “amygdala activity changed” statement.1
The prelimbic cortex has been tied to fear expression and conflict-sensitive behavior in prior amygdala-circuit work. Here, the astrocyte result connects BLA glial signaling to a defined cortical output channel.
That makes the finding more useful for psychiatric neuroscience: extinction is more than a local BLA computation, and astrocyte disruption can weaken the amygdala-prefrontal communication needed for fear-memory performance.
Other Astrocyte Memory Papers Point in the Same Direction
Bukalo et al. is not isolated. A 2017 Nature Neuroscience study found that central-amygdala astrocytes regulated synaptic transmission in a synapse-specific way and influenced amygdala-related behavior.3
That paper was not the same as a BLA cued-fear extinction study, but it already made amygdala astrocytes behaviorally relevant rather than decorative.
A 2023 Journal of Neuroscience paper moved closer to the central question by showing that BLA astrocytes were engaged during acquisition and expression of contextual fear memory.2 Its value here is not that it proves the Bukalo mechanism in advance.
It narrows the prior probability: BLA astrocytes had already been seen changing during fear learning, so Bukalo et al. could ask whether those changes support neuronal representation and extinction rather than merely accompany behavior.
Newer memory-ensemble studies widen the frame. Williamson et al. reported that learning-associated astrocyte ensembles regulate memory recall, while Dewa et al. found that astrocytic ensembles can act as multiday traces that stabilize memory after emotional experience.4,5
Those papers are not PTSD-treatment evidence either. They make the same conceptual correction from different angles: memory circuits are not neuron-only systems with glia in the background.
Mouse Circuit Evidence Is Stronger Than the PTSD Treatment Claim
The clinical relevance is real but bounded. Exposure therapy depends on extinction-like learning, and extinction failure is a plausible contributor to persistent fear in PTSD and anxiety disorders.6
If astrocytes shape extinction-related neuronal representations in mice, astrocyte biology belongs in the fear-learning conversation.
The paper does not show that a human astrocyte drug improves exposure therapy. It used male mice, viral tools, calcium reporters, DREADDs, CalEx, implanted optics, and task-specific fear-conditioning procedures.
The Nature reporting summary also states that sample sizes were based on pilot and prior similar experiments rather than a formal a priori power analysis. Those choices are normal for invasive circuit neuroscience, but they limit the clinical inference.
A useful translational reading is therefore narrow and testable. Astrocyte-targeted strategies should not be sold as next-generation PTSD treatments on this paper alone.
They should be treated as candidate mechanisms for improving fear extinction, especially if future work can identify druggable pathways such as noradrenergic, glutamatergic, adenosine, or gliotransmitter systems that alter astrocyte-neuron coordination without bluntly disrupting the timing of Ca2+ signaling.7
Questions About Astrocytes, Fear Memory, and Extinction
Did Bukalo et al. prove astrocytes cause fear memory in humans?
No. The evidence is causal for male mouse BLA circuits, not human PTSD.
The human relevance comes from the shared logic of fear retrieval and extinction, but the paper itself used mouse conditioning, viral targeting, optical recordings, and cell-specific manipulations.
Why did hM3Dq and hM4Di push freezing in opposite directions?
The tools altered astrocyte Ca2+ dynamics differently. hM3Dq actuation produced an early surge followed by a prolonged low-Ca2+ state, while hM4Di increased Ca2+ transients in the BLA experiments.
Opposite retrieval effects are therefore consistent with different astrocyte states, while the shared extinction impairment suggests extinction learning needs properly timed astrocyte signaling rather than simply “more” or “less” activity.
What does 0.20 vs. 0.31 information per neuron mean?
The decoder analysis estimated how much CS-related information individual BLA neurons carried after astrocyte manipulation. Lower information in CNO-treated hM3Dq mice means the neuronal ensemble represented the threat cue less effectively, and that weaker code tracked lower freezing.
Why is the 83% to 31% BLA-to-prelimbic result important?
It moves the finding from local BLA activity to circuit output. CalEx depletion did more than change a bulk amygdala signal; it reduced CS responsiveness in identified BLA neurons projecting to prelimbic cortex, a pathway relevant to fear expression.
Could astrocytes become a drug target for exposure therapy?
Possibly, but the paper supports mechanism discovery before clinical optimism. A realistic next step is identifying molecular levers that tune astrocyte-neuron coordination during extinction without globally disrupting BLA signaling, then testing whether those levers improve extinction retention in animal models before moving toward human trials.
References
- Bukalo O, O’Sullivan R, Tanisumi Y, et al. Astrocytes enable amygdala neural representations supporting memory. Nature. 2026;652:434–441. doi:10.1038/s41586-025-10068-0
- Suthard RL, Senne RA, Buzharsky MD, et al. Basolateral amygdala astrocytes are engaged by the acquisition and expression of a contextual fear memory. Journal of Neuroscience. 2023;43(27):4997–5013. doi:10.1523/JNEUROSCI.1775-22.2023
- Martin-Fernandez M, Jamison S, Robin LM, et al. Synapse-specific astrocyte gating of amygdala-related behavior. Nature Neuroscience. 2017;20(11):1540–1548. doi:10.1038/nn.4649
- Williamson MR, Kwon W, Woo J, et al. Learning-associated astrocyte ensembles regulate memory recall. Nature. 2025;637(8045):478–486. doi:10.1038/s41586-024-08170-w
- Dewa K, Kaseda K, Kuwahara A, et al. The astrocytic ensemble acts as a multiday trace to stabilize memory. Nature. 2025;648(8092):146–156. doi:10.1038/s41586-025-09619-2
- LeDoux JE. Coming to terms with fear. Proceedings of the National Academy of Sciences. 2014;111(8):2871–2878. doi:10.1073/pnas.1400335111
- Singewald N, Schmuckermair C, Whittle N, Holmes A, Ressler KJ. Pharmacology of cognitive enhancers for exposure-based therapy of fear, anxiety and trauma-related disorders. Pharmacology & Therapeutics. 2015;149:150–190. doi:10.1016/j.pharmthera.2014.12.004