A 60-patient randomized trial of lithium loading for acute bipolar I mania reported faster serum lithium attainment than standard titration and a larger 14-day Young Mania Rating Scale (YMRS) change score, but the same accepted manuscript says absolute YMRS scores at Days 3, 7, and 14 were not significantly different between the 2 arms when both groups also received quetiapine.1
Research Highlights
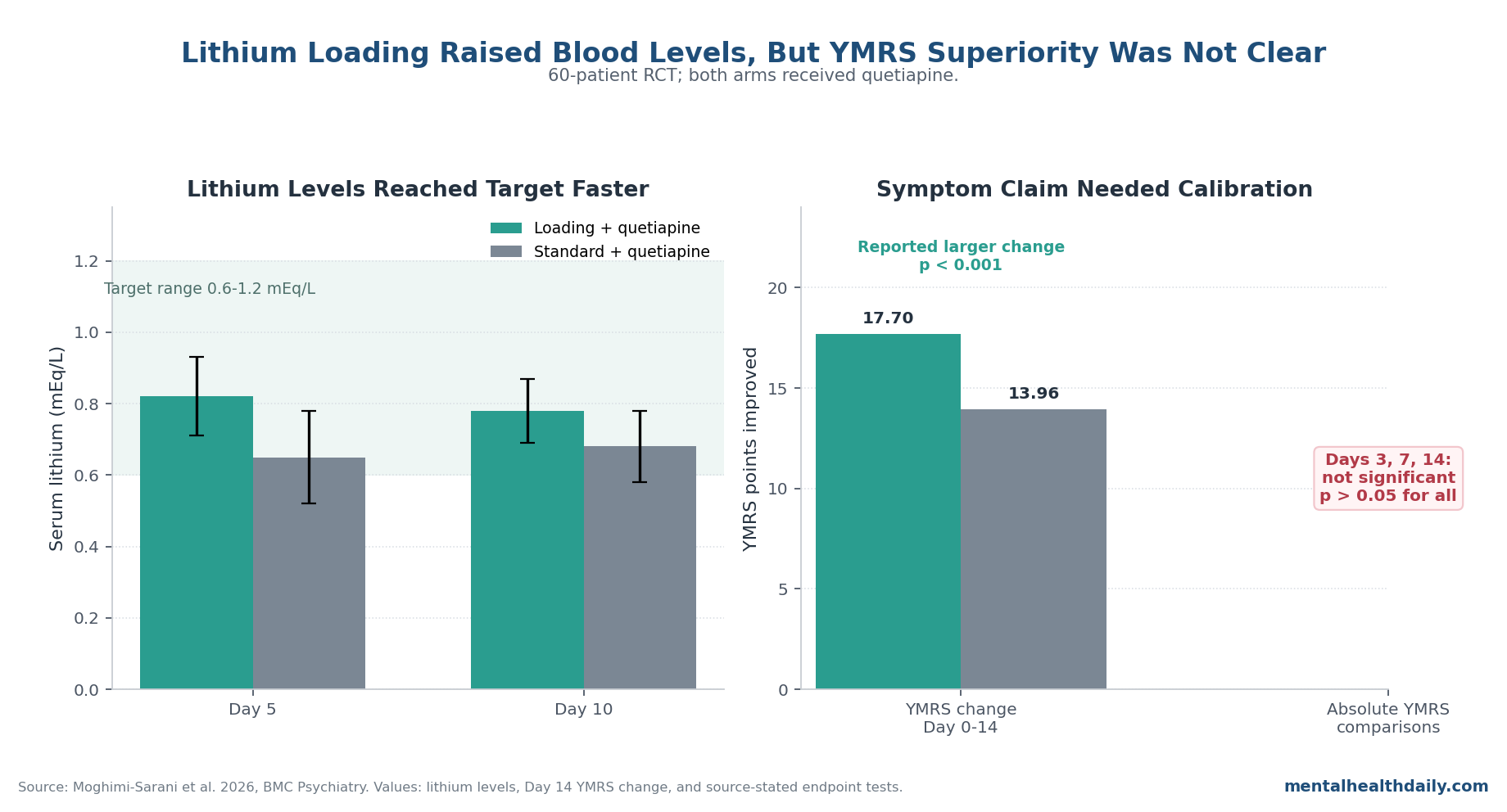
- Loading raised lithium levels faster than standard titration: serum lithium was higher with loading at Day 5 (0.82 ± 0.11 vs. 0.65 ± 0.13 mEq/L) and Day 10 (0.78 ± 0.09 vs. 0.68 ± 0.10 mEq/L).1
- The symptom verdict was mixed: the manuscript reported a larger YMRS decrease from baseline with loading than standard titration (−17.70 ± 3.76 vs. −13.96 ± 2.94; p < 0.001), yet also stated that absolute YMRS scores at Days 3, 7, and 14 were not significantly different (p > 0.05 for all).1
- Baseline imbalance matters: the loading group started numerically more manic (baseline YMRS 32.06 ± 6.97 vs. 28.96 ± 5.30; p = 0.057), so raw change scores are easier to overread than endpoint comparisons.1
- Quetiapine was the active backdrop: both groups received quetiapine titrated toward 400-800 mg/day, and a separate 109-patient youth trial found stronger acute response with quetiapine than lithium (72% vs. 49%; p = 0.012).3
- Safety was reassuring but narrow: no participant exceeded 1.4 mEq/L lithium or dropped out during 14 days, but n = 60 is too small and too short to establish routine loading-dose safety outside monitored inpatient care.1
Lithium loading means giving a higher first-day lithium dose to reach the therapeutic blood range sooner. In this trial, the loading arm received 20 mg/kg on Day 1, capped at 1,800 mg, then moved into ordinary titration.
The standard arm started at 300 mg/day and increased by 300 mg every 2-3 days toward 900-1,200 mg/day. Both arms used serum monitoring to stay in the 0.6-1.2 mEq/L target range.1
The important correction is simple: this is not a clean “loading dose beats standard dosing” paper. It is an early-access accepted manuscript with a real pharmacokinetic signal, a favorable change-score claim, and a final discussion/conclusion that explicitly backs away from short-term superiority.
Loading Raised Serum Lithium by Day 5, But Quetiapine Was in Both Arms
The biological premise was plausible. Lithium is a mood stabilizer used for acute mania and relapse prevention, but its clinical effect can lag because therapeutic serum levels are reached gradually.
Quetiapine is a second-generation antipsychotic that blocks dopamine D2 and serotonin 5-HT2A signaling, among other receptors, and can reduce manic symptoms quickly enough to be useful during the acute inpatient window.1
Moghimi-Sarani et al. tested whether accelerating lithium exposure would add something on top of that fast antipsychotic backdrop. The loading arm did what a loading arm is supposed to do pharmacokinetically: Day 5 lithium levels averaged 0.82 ± 0.11 mEq/L vs. 0.65 ± 0.13 mEq/L in standard titration, and Day 10 levels remained higher at 0.78 ± 0.09 vs. 0.68 ± 0.10 mEq/L.1
Those values matter because faster blood-level attainment is the whole argument for loading. But they do not, by themselves, prove faster clinical recovery.
The clinical question is whether the higher early lithium level changes mania outcomes once every patient is already receiving quetiapine.
The YMRS Results Point in 2 Directions
YMRS is the Young Mania Rating Scale, a clinician-rated symptom scale used to track manic severity. The trial’s primary endpoint was change in YMRS from baseline to Day 14.
The abstract and results text reported a larger 14-day decrease with loading-dose lithium: −17.70 ± 3.76 vs. −13.96 ± 2.94 with standard titration, p < 0.001.1
Read alone, that line sounds like a decisive loading-dose win. The rest of the manuscript does not support such a clean claim.
The same results section says direct comparisons of absolute YMRS scores between groups at Days 3, 7, and 14 “did not reveal statistically significant differences” (p > 0.05 for all). The discussion goes further, stating that adding a lithium loading dose “did not provide a statistically significant benefit in either the speed or magnitude of symptom reduction” compared with standard titration.
The conclusion repeats the same verdict: lithium loading with quetiapine “did not provide short-term advantages” over standard titration with quetiapine.1
That contradiction should not be hidden. The fairest read is that loading achieved faster serum levels and produced some favorable change-score arithmetic, but the trial did not establish clear endpoint superiority.
The baseline YMRS imbalance helps explain why change scores can look better than absolute score comparisons: the loading group began with numerically higher mania scores (32.06 ± 6.97 vs. 28.96 ± 5.30; p = 0.057), leaving more room to fall.1
Aggressive and Disruptive Behavior Favored Loading at Day 14
The stronger loading signal appeared in a secondary behavioral measure, not the clean absolute YMRS comparison. Aggressive/disruptive behavior scores were similar early, but by Day 14 the loading group had a lower mean score than the standard group: 0.06 ± 0.36 vs. 0.53 ± 0.89, p = 0.012.
Change from baseline also favored loading at Day 3 (−1.66 ± 1.18 vs. −0.60 ± 1.30; p = 0.001), Day 7 (−2.46 ± 1.71 vs. −1.33 ± 1.76; p = 0.008), and Day 14 (−3.00 ± 1.72 vs. −1.86 ± 1.90; p = 0.009).1
This matters clinically because acute mania is more than a mood-score problem. Agitation, impulsivity, and disruptive behavior often determine whether someone remains at high risk on an inpatient unit.
A behavioral-control signal can be meaningful even when the headline YMRS endpoint is ambiguous.
The limitation is placement. The trial was not powered or framed around a definitive behavioral-control endpoint, and the manuscript’s own global conclusion still favored standard titration as sufficient when paired with quetiapine.
The behavioral result should raise interest in a subgroup or secondary-outcome hypothesis, not rewrite the trial as a broad loading-dose victory.
Prior Acute-Mania Evidence Makes the Nullish Endpoint Plausible
The adjacent literature makes this study’s cautious conclusion more believable. A 2019 Cochrane review found lithium more effective than placebo for acute mania, but limited evidence suggested little or no difference between lithium and several other mood stabilizers or antipsychotics, with uncertainty around some comparisons.2
Lithium is real acute-mania treatment; it is not automatically the fastest acute-mania treatment.
Quetiapine’s role is especially relevant because both arms in the 2026 trial received it. In a 109-patient randomized youth trial of early-course bipolar mania or mixed episodes, Patino et al. reported a higher response rate with quetiapine than lithium (72% vs. 49%; p = 0.012), with no significant remission-rate separation.3
That does not directly answer an adult inpatient loading-dose question, but it supports the mechanism of dilution: if quetiapine is already doing much of the early antimanic work, faster lithium levels may have less room to create obvious YMRS separation.
Longer-horizon bipolar data also resist a simple speed story. Bipolar CHOICE, a pragmatic 6-month trial, found no overall efficacy or safety/tolerability difference between lithium and quetiapine when each was used with adjunctive personalized treatment.4
Quetiapine plus lithium or divalproex has maintenance evidence as well; in Trial 126, quetiapine 400-800 mg/day added to lithium/divalproex increased time to recurrence compared with placebo plus lithium/divalproex.5 Those studies point to combination strategy and sustained management, not a guarantee that a Day 1 lithium loading maneuver changes acute endpoint scores.
Safety Looked Fine for 14 Days, Not Settled for Routine Loading
No participant in the Moghimi-Sarani trial exceeded 1.4 mEq/L lithium, no one dropped out, and the manuscript reported no detected tremor, renal complications, urinary-output changes, ECG alterations, hair loss, or dermatologic symptoms in either arm during the 14-day follow-up.1
That is reassuring, but it is not a broad safety license. Lithium’s therapeutic window is narrow: the useful range sits close enough to toxic exposure that dosing has to track kidney function, hydration, interacting medications, and serum levels.
A systematic toxicity review by McKnight et al. found clinically important monitoring issues across renal, thyroid, parathyroid, weight, skin, hair, and pregnancy-related outcomes.6
The trial’s own limitations also warn against overconfidence. Follow-up lasted 14 days, the sample was 60 patients, the study was single-center, and the limitations section says detailed illness-history variables were not systematically collected.
It also notes that the absence of a predefined detailed adverse-event checklist before trial initiation may have led to underreporting of side effects.1
Where Lithium Loading Might Still Make Sense
The 2026 study does not support routine loading-dose lithium for every hospitalized mania patient receiving quetiapine. It also does not make loading irrational. It narrows the use case.
Loading is most defensible when the patient is already in a monitored inpatient setting, renal function is acceptable, hydration status is stable, drug interactions are controlled, and prior lithium response makes lithium a high-value long-term choice. In that setting, faster serum attainment may be worth pursuing even if the expected acute YMRS advantage is modest or uncertain.
The argument weakens in outpatient care, medically unstable patients, dehydration risk, kidney impairment, uncertain adherence, pregnancy, or situations where rapid antipsychotic response is already strong and lithium is being added mainly for maintenance. In those cases, standard titration preserves the long-term lithium option without pretending that faster blood levels necessarily mean faster clinical recovery.
Questions About This Lithium Loading Trial
Did lithium loading beat standard lithium titration in this 60-patient trial?
Not cleanly. Loading produced faster lithium levels and the paper reported a larger 14-day YMRS change score, but absolute YMRS scores at Days 3, 7, and 14 were not significantly different.
The manuscript’s own conclusion says loading did not provide a short-term advantage over standard titration when both groups received quetiapine.1
Why does the abstract sound more favorable to loading than the conclusion?
The abstract emphasizes change from baseline (−17.70 vs. −13.96 YMRS points; p < 0.001) and faster serum lithium levels. The conclusion emphasizes the lack of significant absolute-score separation and the likely effect of quetiapine’s rapid antimanic action in both arms.
Baseline YMRS was also numerically higher in the loading group, which makes change-score interpretation less straightforward.1
Does faster lithium level attainment matter if quetiapine is already given?
It may matter for selected patients, especially if lithium is the planned maintenance anchor or if disruptive behavior is a major inpatient problem. But this trial suggests faster serum lithium attainment did not reliably translate into better absolute YMRS scores over 14 days when quetiapine was titrated in both arms.1
Was loading-dose lithium safe in this study?
Short-term monitored safety looked acceptable: 0 participants exceeded 1.4 mEq/L, and there were no dropouts over 14 days. That finding applies to a small inpatient trial with serum monitoring, not to unsupervised outpatient loading or rare toxicities that require much larger samples to detect.1
What should clinicians or families take from the trial?
The practical read is calibration. Lithium remains important in bipolar I disorder, and loading can raise levels quickly under monitoring.
But in acute mania already treated with quetiapine, standard lithium titration appears hard to dismiss, and loading should be reserved for cases where the expected benefit justifies closer monitoring.
References
- Moghimi-Sarani E, Etemad SZ, Dehbozorgi R. Comparative efficacy of lithium carbonate loading dose versus standard dosing, each combined with quetiapine, in acute mania of bipolar I disorder: a randomized single-blind controlled trial. BMC Psychiatry. 2026. doi:10.1186/s12888-026-08084-z
- McKnight RF, de La Motte de Broons de Vauvert SJGN, Chesney E, Amit BH, Geddes J, Cipriani A. Lithium for acute mania. Cochrane Database of Systematic Reviews. 2019;(6):CD004048. doi:10.1002/14651858.CD004048.pub4
- Patino LR, Klein CC, Strawn JR, et al. A randomized, double-blind, controlled trial of lithium versus quetiapine for the treatment of acute mania in youth with early course bipolar disorder. Journal of Child and Adolescent Psychopharmacology. 2021;31(7):485-493. doi:10.1089/cap.2021.0039
- Nierenberg AA, McElroy SL, Friedman ES, et al. Bipolar CHOICE (Clinical Health Outcomes Initiative in Comparative Effectiveness): a pragmatic 6-month trial of lithium versus quetiapine for bipolar disorder. Journal of Clinical Psychiatry. 2016;77(1):90-99. doi:10.4088/JCP.14m09349
- Vieta E, Suppes T, Eggens I, Persson I, Paulsson B, Brecher M. Efficacy and safety of quetiapine in combination with lithium or divalproex for maintenance of patients with bipolar I disorder (international trial 126). Journal of Affective Disorders. 2008;109(3):251-263. doi:10.1016/j.jad.2008.06.001
- McKnight RF, Adida M, Budge K, Stockton S, Goodwin GM, Geddes JR. Lithium toxicity profile: a systematic review and meta-analysis. Lancet. 2012;379(9817):721-728. doi:10.1016/S0140-6736(11)61516-X