Lithium prescribing during pregnancy sits in a knot of competing risks. The teratogenicity data have been recalibrated dramatically since the 1970s, while the postpartum-relapse data have moved in the opposite direction.
Most popular framing still treats lithium as a uniquely dangerous fetal exposure to be avoided when possible. The new Chauhan UK cohort shows what that avoidance looks like in practice, and the pattern raises questions the headline numbers don’t answer.
Research Highlights
- UK lithium use during pregnancy is rare, and most women stop. In 752,112 pregnancies (1995–2018), only 1.89 per 10,000 had any lithium prescription during pregnancy. Among 227 pre-conception users, just 15.4% continued through all three trimesters.1
- NICE updates didn’t budge prescribing. The 2006 and 2014 NICE updates produced no detectable shift in rates. Discontinuation patterns held steady across two decades.1
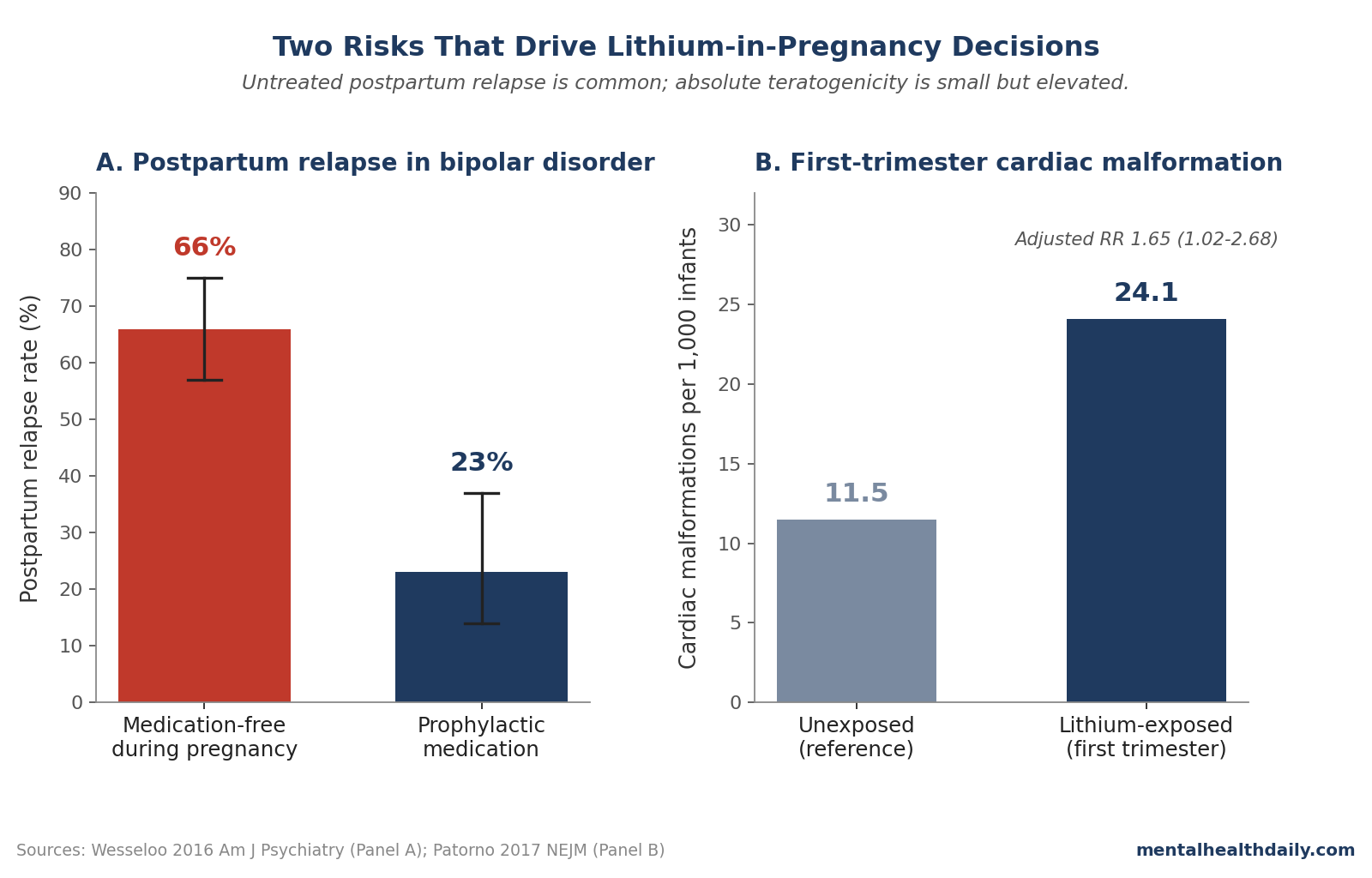
- The headline teratogenicity number has been recalibrated downward. The 1970s claim of a 400-fold rise in Ebstein anomaly came from a heavily selection-biased register. The largest modern analysis (Patorno 2017, >1.3 million Medicaid pregnancies) put the adjusted relative risk for cardiac malformations at 1.65 (95% CI 1.02–2.68).2
- Untreated postpartum relapse is the underweighted risk. Wesseloo 2016 found a 66% postpartum relapse rate in unmedicated bipolar pregnancies vs. 23% on prophylaxis.3 For most women, abrupt discontinuation carries larger expected harm than continued lithium.
- The defensible read isn’t “stop” or “continue” — it’s planning. Late-pregnancy abrupt stops — the modal pattern in Chauhan — combine first-trimester exposure with unmedicated postpartum.
Chauhan 2026: 15.4% Continuation Rate Across 752,112 UK Pregnancies
The study analyzed primary-care records from the Clinical Practice Research Datalink (CPRD) GOLD Pregnancy Register, covering pregnancies that started between 1995 and 2018 and ended in a live birth or stillbirth.1 A few headline numbers worth pulling forward:
- Prevalence of lithium prescribing was low and stable. 3.02 per 10,000 pregnancies pre-conception, 1.89 per 10,000 during pregnancy, and 2.81 per 10,000 postpartum. The most recent year (2018) showed 1.03 per 10,000 during pregnancy.1
- NICE guidelines didn’t change practice. Comparing 1995–2006, 2007–2014, and 2015–2018, prevalence remained essentially flat across the two guideline updates that explicitly addressed perinatal lithium use.1
- Bipolar disorder accounted for under half of perinatal lithium use. Of the 337 pregnancies with any perinatal lithium prescription, 48.4% involved a recorded bipolar diagnosis. The remainder were primarily severe depression (91.7% of the non-bipolar group) and anxiety, suggesting lithium is widely used as augmentation for treatment-resistant unipolar illness.1
The strongest finding is the discontinuation pattern. Among 227 pregnancies in women with established pre-pregnancy lithium use, treatment continuation through all three trimesters occurred in only 15.4%. Discontinuation before pregnancy occurred in 20.7%, and discontinuation during the second or third trimester in 30.8%. The remaining 33.0% followed other patterns.1
That late-discontinuation cohort is the worrying subgroup. Stopping lithium in the second or third trimester combines first-trimester teratogenic exposure (already incurred) with the highest-risk postpartum window arriving on an unmedicated brain.
Why the Ebstein Anomaly Number Has Been Recalibrated
The 400-fold relative-risk number that defined lithium-pregnancy decisions for decades came from the 1970s Register of Lithium Babies, a voluntary case-collection where physicians were far more likely to report adverse outcomes than healthy births.4 Cohen’s 1994 re-analysis put the absolute Ebstein risk at roughly 1 in 1,000 to 1 in 2,000 lithium-exposed births, vs. a population baseline near 1 in 20,000 — an elevation, but nowhere near the original headline.5
The largest modern analysis is Patorno and colleagues’ 2017 study in NEJM, which used Medicaid data to compare cardiac malformation rates in 663 first-trimester lithium-exposed pregnancies against more than 1.3 million unexposed pregnancies.2 The headline numbers:
- Lithium-exposed cardiac malformation rate: 24.1 per 1,000 infants (16 of 663).
- Unexposed cardiac malformation rate: 11.5 per 1,000 infants (15,251 of 1,322,955).
- Adjusted relative risk: 1.65 (95% CI 1.02–2.68).2
Patorno also found a dose-response gradient: the adjusted relative risk was 1.11 for daily doses of 600 mg or less, 1.60 for 601–900 mg, and 3.22 for doses above 900 mg.2 The high-dose subgroup is small, but the gradient is consistent with what would be expected for a true teratogenic effect.
Munk-Olsen’s 2018 meta-analysis pooled six cohorts (727 lithium-exposed pregnancies) and reported odds ratios of 1.81 (95% CI 1.35–2.41) for any congenital malformation and 1.86 (95% CI 1.16–2.96) for any cardiac malformation, comparing against mood-disordered women not exposed to lithium.6 Two independent cohorts with different sampling frames landed in the same range.
The honest read on teratogenicity: first-trimester lithium exposure is associated with about a 65% relative increase in cardiac malformation risk, translating to roughly 1 extra cardiac malformation per 100 first-trimester lithium-exposed live births. That’s elevated and worth informed discussion. It is not the 400-fold catastrophe the older literature implied, and it is not zero.
The Postpartum Relapse Risk That Doesn’t Get the Same Headlines
The other side of the ledger is much larger and gets much less popular coverage. Wesseloo and colleagues’ 2016 systematic review in Am J Psychiatry pooled 37 studies of postpartum relapse in women with histories of bipolar disorder or postpartum psychosis.3 The headline pooled estimates:
- Overall postpartum relapse risk in bipolar disorder: 37% (95% CI 29%–45%).3
- Postpartum relapse if medication-free during pregnancy: 66% (95% CI 57%–75%).3
- Postpartum relapse if on prophylactic medication during pregnancy: 23% (95% CI 14%–37%).3
The gap between 66% and 23% is roughly the expected magnitude of harm from a discontinuation strategy that doesn’t bridge with another mood stabilizer or restart lithium immediately postpartum. For comparison, the cardiac-malformation absolute risk under lithium exposure (~2.4%) is more than an order of magnitude smaller than the unmedicated postpartum-relapse risk (~66%).
Viguera’s 2000 cohort of 42 women with bipolar disorder found that mood-episode recurrence was 2.9 times higher in the 40 weeks after lithium discontinuation than in the year before, and rapid discontinuation (1–14 days) produced more recurrence than gradual (15–30 days).7
For women with histories of postpartum psychosis specifically, the prophylaxis signal is sharper. Bergink’s 2012 prospective study of 70 high-risk pregnancies found that none of the 20 women with postpartum-psychosis-only histories who took postpartum lithium prophylaxis relapsed, vs. 44.4% who declined prophylaxis.8
Why Late Discontinuation Is the Worst of Both Worlds
Chauhan’s most concerning finding is that 30.8% of pre-conception lithium users discontinued during the second or third trimester — not before pregnancy.1 This pattern combines three failure modes:
- First-trimester teratogenic exposure has already occurred. Cardiac formation completes by gestational week 8–10. Stopping in the second trimester does not undo whatever cardiac risk was incurred in the first.
- The mother enters the highest-risk postpartum window unmedicated. Wesseloo’s data put the medication-free relapse rate at 66%; the relapse hazard peaks within the first 1–3 months postpartum.3
- Abrupt rather than tapered discontinuation amplifies relapse risk. Viguera’s data showed roughly twice the recurrence rate when discontinuation occurred over 1–14 days vs. 15–30 days.7 Late-pregnancy stops are often abrupt — driven by sudden risk perception or third-trimester anxieties — rather than planned gradual tapers.
The defensible decision points sit at two extremes. Pre-conception planning either continues lithium throughout pregnancy with rigorous therapeutic drug monitoring, or discontinues before conception with a planned bridging strategy and rapid postpartum restart. Discontinuation in the late second or third trimester, after exposure has already been incurred, doesn’t optimize either ledger.
That isn’t a hypothetical risk profile. The Chauhan cohort confirms it’s the modal pattern for nearly a third of UK pre-conception lithium users.1
Limitations of the Chauhan Cohort
Outcomes weren’t measured. Chauhan reports prescribing patterns, not whether late discontinuation produced more maternal relapse or worse infant outcomes. Linking to secondary care and inpatient data is flagged as the next research priority.1
Prescribing isn’t adherence. A prescription record doesn’t confirm the medication was taken, and the 15.4% continuous-prescribing rate is a ceiling on continuous use, not a floor.
Specialist prescriptions may be undercounted. CPRD captures primary care; some women managed through specialist mental health services may have lithium issued via in-patient channels. The authors estimate this effect is small but note it could slightly overstate discontinuation.1
Severity is unobserved. CPRD doesn’t record indication or illness severity. The univariate differences between continuers and discontinuers (younger age, higher BMI, more bipolar diagnoses, more smoking) are unadjusted and meant as descriptive only.1
Generalizability is partial. The 1.89-per-10,000 UK rate aligns with broader European data,9 but US Medicaid data show roughly 5 per 10,000.2 NICE-shaped UK practice may discontinue more aggressively than other systems.
Practical Use of Lithium Around Pregnancy
For women with bipolar disorder of childbearing age — a population the original NICE guideline explicitly tries to support with preconception planning — the calibrated framework looks like this:
- Plan before conception, not during pregnancy. The decision about whether to continue or discontinue lithium has dramatically different risk profiles depending on when it’s made. Pre-conception, both options remain on the table; mid-second-trimester, only the worst version of “discontinue” is left. The 2014 NICE update specifically formalized preconception multidisciplinary planning for exactly this reason.10
- Continue if the relapse risk is high. For women with severe, recurrent bipolar disorder — particularly with histories of psychotic mania, postpartum psychosis, or rapid relapse on prior discontinuation — the absolute risk of continuing lithium (one extra cardiac malformation per ~100 first-trimester exposures2) is meaningfully smaller than the absolute risk of an unmedicated postpartum relapse. Therapeutic drug monitoring is essential because pregnancy increases lithium clearance, and dose adjustments are typically required.11
- If discontinuing, do it before conception, gradually, with a bridging plan. Gradual taper (over 4 weeks or longer) reduces relapse risk relative to abrupt stops.7 A documented plan for rapid postpartum re-initiation — ideally in the first 24–48 hours after delivery — is what differentiates a defensible discontinuation strategy from an abandoned one.
- Postpartum prophylaxis is the highest-leverage intervention. For women with histories of postpartum psychosis specifically, lithium initiated immediately postpartum has produced near-complete relapse prevention in prospective cohorts.8 This benefit doesn’t require fetal exposure and is one of the strongest preventive signals in perinatal psychiatry.
- The non-bipolar group needs separate consideration. Roughly half of perinatal lithium use in Chauhan’s cohort was for treatment-resistant unipolar depression or anxiety augmentation.1 For these indications, alternatives with cleaner pregnancy-safety profiles (SSRIs for depression, structured psychotherapy) are typically first-line, and the case for continuing lithium specifically is weaker.
Common Questions About Lithium and Pregnancy
Is lithium safe during pregnancy?
“Safe” is the wrong frame — the question is which risk you’re trading. First-trimester lithium exposure is associated with roughly a 65% relative increase in cardiac malformation risk (adjusted RR 1.65, 95% CI 1.02–2.68 in Patorno 2017), which translates to about one extra malformation per 100 exposed births.2 Untreated bipolar disorder during pregnancy and postpartum carries a 66% relapse risk if no medication is used,3 compared with 23% if prophylactic medication is continued. For most women with established bipolar disorder, the relapse risk of stopping is larger than the teratogenic risk of continuing.
What’s the actual risk of Ebstein anomaly with lithium?
Far smaller than the often-cited 1970s figures suggested. Modern data place absolute Ebstein anomaly risk in lithium-exposed pregnancies at roughly 1 in 1,000 to 1 in 2,000, vs. a population baseline of about 1 in 20,000 — an elevation, but on the order of 5- to 10-fold rather than the 400-fold figure that came from the original heavily selection-biased Register of Lithium Babies.5 Cardiac malformations more broadly run at about 24 per 1,000 lithium-exposed births vs. 12 per 1,000 unexposed.2
Should I stop lithium if I find out I’m pregnant?
Not without consulting a perinatal psychiatrist, and not abruptly. By the time most women know they’re pregnant, first-trimester cardiac development is already underway, so stopping at that point doesn’t avoid teratogenic exposure but does add postpartum relapse risk. Abrupt stops produce roughly twice the relapse rate of gradual tapers.7 The decision involves weighing your specific relapse history against the modest absolute teratogenic risk.
Can I breastfeed while taking lithium?
Lithium is excreted in breast milk and infant serum levels can reach 10–50% of maternal levels. Most current guidelines (Royal College of Psychiatrists, UK Teratology Information Service) consider breastfeeding compatible with lithium when the infant is healthy, full-term, and can be monitored; the calculus differs for premature or medically fragile infants.12 The decision is individualized rather than categorical.
Why didn’t NICE guidelines change prescribing?
Chauhan found prevalence essentially flat across the 2006 and 2014 NICE updates.1 Pre-existing clinician caution already aligned with the guideline direction, and the underlying rarity of lithium use makes trend changes statistically hard to detect. The guideline’s contribution may sit in the quality of preconception planning, not in raw rates.
What about valproate or other alternatives?
Valproate is contraindicated in women of childbearing potential because its teratogenic and neurodevelopmental risk is substantially higher than lithium’s.10 Lamotrigine has a cleaner pregnancy-safety profile but weaker efficacy for mania prevention. Antipsychotics (especially quetiapine and olanzapine) have larger pregnancy evidence bases than a decade ago and are reasonable in selected cases.
References
- Lithium prescribing in the perinatal period: UK primary care cohort study. Chauhan D et al. The British Journal of Psychiatry. 2026. doi:10.1192/bjp.2026.10632
- Lithium Use in Pregnancy and the Risk of Cardiac Malformations. Patorno E et al. New England Journal of Medicine. 2017;376(23):2245-2254. doi:10.1056/NEJMoa1612222
- Risk of Postpartum Relapse in Bipolar Disorder and Postpartum Psychosis: A Systematic Review and Meta-Analysis. Wesseloo R et al. American Journal of Psychiatry. 2016;173(2):117-127. doi:10.1176/appi.ajp.2015.15010124
- Lithium and pregnancy. I. Report from the Register of Lithium Babies. Schou M et al. British Medical Journal. 1973;2(5859):135-136. doi:10.1136/bmj.2.5859.135
- A reevaluation of risk of in utero exposure to lithium. Cohen LS et al. JAMA. 1994;271(2):146-150. doi:10.1001/jama.1994.03510260078033
- Maternal and infant outcomes associated with lithium use in pregnancy: an international collaborative meta-analysis of six cohort studies. Munk-Olsen T et al. The Lancet Psychiatry. 2018;5(8):644-652. doi:10.1016/S2215-0366(18)30180-9
- Risk of recurrence of bipolar disorder in pregnant and nonpregnant women after discontinuing lithium maintenance. Viguera AC et al. American Journal of Psychiatry. 2000;157(2):179-184. doi:10.1176/appi.ajp.157.2.179
- Prevention of Postpartum Psychosis and Mania in Women at High Risk. Bergink V et al. American Journal of Psychiatry. 2012;169(6):609-615. doi:10.1176/appi.ajp.2012.11071047
- Lithium Use During Pregnancy in 14 Countries. Wittstrom F et al. JAMA Network Open. 2024;7(12):e2451117. doi:10.1001/jamanetworkopen.2024.51117
- Antenatal and Postnatal Mental Health: Clinical Management and Service Guidance (CG192). National Institute for Health and Care Excellence. 2014. nice.org.uk/guidance/cg192
- Lithium dosing strategies during pregnancy and the postpartum period. Wesseloo R et al. The British Journal of Psychiatry. 2017;211(1):31-36. doi:10.1192/bjp.bp.116.192799
- Maternal lithium use and the risk of adverse pregnancy and neonatal outcomes: a Swedish population-based cohort study. Hastie R et al. BMC Medicine. 2021;19(1):291. doi:10.1186/s12916-021-02170-7
