An ear-clip that modulates mood by stimulating the vagus nerve has obvious appeal — but the evidence base for non-invasive tVNS in depression has been mixed for a decade. A 2026 cross-over RCT by Forbes et al. sharpens what specifically tVNS does well.1
Research Highlights
- Transcutaneous vagus nerve stimulation (tVNS) is a non-invasive ear-electrode version of an FDA-approved invasive therapy for treatment-resistant depression. Modulating mood with a clip on the auricle has obvious appeal; the evidence base is real but smaller than the popular framing suggests.2
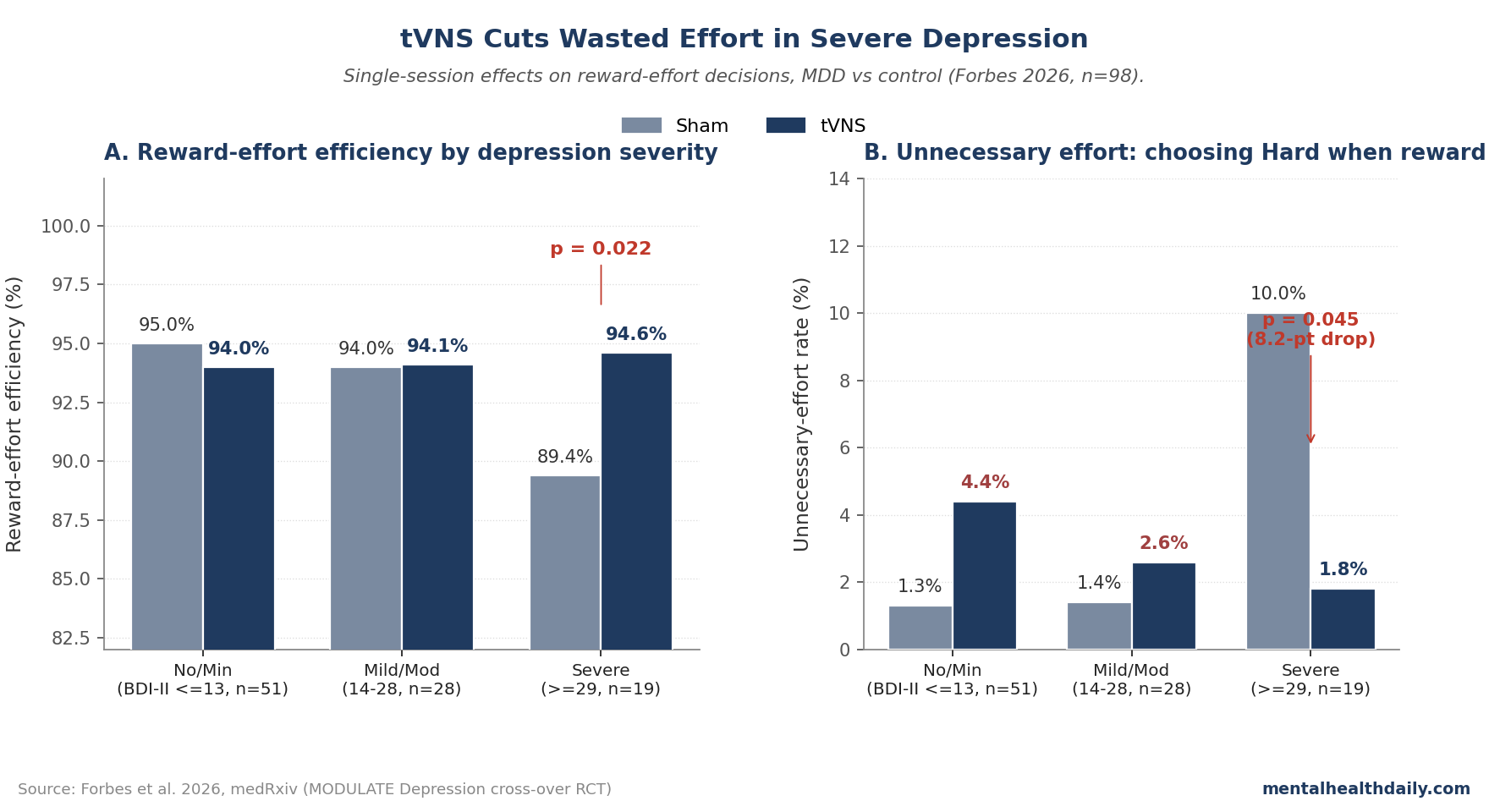
- The Forbes 2026 RCT (n = 98, 53 MDD + 45 controls) tested how tVNS affects effort-reward decisions, not symptom scores. A single session of tVNS improved reward-effort efficiency in patients with severe depressive symptoms (BDI-II ≥ 29), with no effect in those with mild-moderate or no symptoms.1
- The mechanism was reduction of unnecessary effort, not enhancement of necessary effort. Severe-MDD patients exerted high effort on no-reward trials; tVNS dropped that wasted-effort rate from 10.0% to 1.8% (p = 0.045) without changing how often they engaged effort when it actually mattered.1
- The severe-symptom subgroup is small (n = 19). The categorical finding replicated when BDI-II was treated as continuous (p = 0.020), but n = 19 is the sample driving the headline.
- tVNS for symptom reduction has multi-week RCT evidence (Hein 2013, Li 2022, Rong 2016). Effects are modest and heterogeneous, and the meta-analytic picture has been muddier than enthusiasts suggest.3,4 The Forbes finding adds a behavioral mechanism candidate: improved cost-benefit allocation.
Vagus nerve stimulation has an unusual evidence trajectory in psychiatry. The invasive surgically implanted version has been FDA-approved for treatment-resistant major depression since 2005.
The non-invasive tVNS version — the ear clip targeting the auricular branch of the vagus — has been studied in dozens of small trials over the past decade with mixed results.
Forbes 2026 doesn’t resolve that bigger question, but it adds a sharper finding: in patients with severe depressive symptoms, a single session of tVNS measurably improved how efficiently they allocated effort to gain rewards — specifically by cutting wasted effort, not by boosting effortful pursuit.
The finding is small, single-session, and concentrated in 19 severely symptomatic patients, but it points at a specific motivational mechanism that’s been hard to pin down with symptom-rating-scale outcomes.1
Forbes 2026: A Cross-Over RCT With a Behavioral Endpoint
The trigger paper is the MODULATE Depression study (DRKS00024823), a randomized single-blind cross-over trial conducted at Goethe University Frankfurt. 98 participants completed the analysis: 53 with current major depressive episodes meeting DSM-5 criteria, 45 healthy controls.
Each completed two sessions within 10 days, receiving tVNS at the cymba conchae (left ear) on one day and sham stimulation at the earlobe on the other, with stimulation lasting ~90 minutes per session.1
The behavioral endpoint was the Grip Strength Effort Task (GSET): participants chose between an Easy option (50% of maximum grip strength, 0.10 EUR reward) and a Hard option (90% of max, with rewards of 0.10, 0.20, or 0.40 EUR depending on the trial).
The task structure created two distinct decision categories:
- Necessary-effort trials: Easy was 0.10 EUR, Hard was 0.20 or 0.40 EUR. Choosing Hard rationally trades effort for higher reward.
- Unnecessary-effort trials: Easy was 0.10 EUR, Hard was also 0.10 EUR. Choosing Hard means working harder for no benefit.
The primary outcome was reward-effort efficiency: the share of trials in which participants made the rational allocation (Easy when rewards were equal; Hard when rewards were unequal). Maximizing efficiency requires both engaging effort when it matters and refraining when it doesn’t.
Blinding was reasonable: of 71 participants who guessed treatment assignment for both sessions, only 11 (15.5%) were correct — below the 25% expected by chance, suggesting active sham masking.1
Effect Concentrated in Severe Depressive Symptoms
Comparing tVNS vs. sham across the full sample, there was no main effect of stimulation on reward-effort efficiency. The signal lived entirely in the depressive-symptom-severity interaction.1
Splitting participants by Beck Depression Inventory-II score into three groups (no/minimal ≤ 13; mild/moderate 14–28; severe ≥ 29):
- Severe BDI-II group (n = 19): Reward-effort efficiency rose from 89.4% (sham) to 94.6% (tVNS), p = 0.022. The pre-specified condition-by-severity interaction was significant (χ² = 6.34, p = 0.042), driven by this group (β = 0.931, SE = 0.370, p = 0.012).1
- Mild/moderate BDI-II group (n = 28): No significant change (94.0% sham vs. 94.1% tVNS, p = 0.939).
- No/minimal BDI-II group (n = 51, mostly healthy controls): No significant change (95.0% sham vs. 94.0% tVNS, p = 0.360).
Treating BDI-II as a continuous variable rather than three categories preserved the interaction (χ² = 5.42, p = 0.020), so the categorical cut at 29 isn’t doing the statistical work.
The severity-restricted effect is consistent with a broader pattern in the cognitive-stimulation literature: tVNS tends to help when baseline performance is suboptimal and provides limited benefit (sometimes detrimental) when performance is already at or near ceiling.5
Reward-effort efficiency in the no/minimal-symptom group was already 95% under sham; there was little room to improve.
Mechanism: Less Wasted Effort, Not More Engaged Effort
The follow-up analysis splitting reward-effort efficiency into its two components is where the mechanistic claim sharpens. In the severe-MDD group:1
- Unnecessary effort dropped substantially. The probability of choosing Hard when both options paid 0.10 EUR fell from 10.0% under sham to 1.8% under tVNS (p = 0.045). The condition-by-severity interaction for unnecessary effort was significant (χ² = 8.56, p = 0.014).
- Necessary effort was unchanged. The probability of choosing Hard when the higher reward was on offer didn’t differ between sham and tVNS in any severity group (interaction χ² = 0.342, p = 0.843).
The asymmetry is interesting. Most depression-effort research has framed the problem as motivational deficit — depressed patients are less willing to engage effort even when rewards are available.6 Pillny’s 2024 meta-analysis of 23 effects found a small overall effect (g = −0.20) of MDD on willingness to exert effort for reward.7
The Forbes data show something different: severe-MDD patients in this trial weren’t shy about exerting effort — they exerted too much when it didn’t help. Under sham conditions, they chose the harder grip 10% of the time even when both options paid the same amount. Healthy controls and mild-MDD patients did this only ~1% of the time. tVNS normalized severe-MDD patients to control-level performance, specifically by reducing this maladaptive over-engagement.
The interpretation Forbes offers is that severe MDD impairs cost-benefit weighting, leading to either too little or too much effort relative to reward magnitude. tVNS may improve this weighting in patients who are over-allocating, particularly through dopaminergic and noradrenergic effects on prefrontal-striatal circuits that compute effort-cost-benefit.8
How tVNS Fits in Depression Treatment
The Forbes paper is one piece of a broader tVNS-for-MDD literature. The headline numbers from that wider literature:
Multi-week tVNS RCTs report modest but consistent symptom reductions. Hein 2013, Li 2022, and Rong 2016 each ran sham-controlled tVNS protocols of 4–12 weeks in MDD and reported greater HAM-D or BDI reductions in the active group.3,9,10 Effect sizes have ranged from small to moderate, with notable between-trial heterogeneity.
Meta-analyses have been cautious. Cimpianu’s 2017 meta-analysis flagged blinding as the chief methodological concern across the tVNS literature.11 Wu’s 2018 meta-analysis pooled 12 trials and reported tVNS as superior to sham for both MDD and anxiety endpoints, but with a high risk-of-bias tail.12 The most rigorous reviews have called the evidence “promising but not definitive.”
Invasive VNS evidence is older and more established. The implantable VNS device received FDA approval for treatment-resistant MDD in 2005 and has been shown to produce sustained symptom benefit in long-term observational follow-up (Aaronson 2017 reported a 5-year ~67% response rate in TRD vs. 41% on treatment as usual).13 Whether non-invasive tVNS recapitulates these effects, with what magnitude, and over what timescale is the open question Forbes contributes a single mechanism-of-action candidate to.
The Forbes contribution isn’t to resolve whether tVNS treats depression. It’s to offer a behavioral mechanism — improved effort-allocation efficiency in severely symptomatic patients — that fits with dopaminergic-modulation accounts of vagus nerve stimulation and that is mechanistically distinct from the broader “improves mood” framing.
Why Severe Symptoms Specifically?
The severity-restricted effect echoes a pattern across multiple non-invasive brain-stimulation modalities: ECT, rTMS, and ketamine all show larger effect sizes in more severely depressed populations.14 Three explanations are plausible:
- Ceiling effects on reward-effort efficiency. Healthy controls and mild-MDD patients were already at or near 95% efficient under sham; there was no room for tVNS to improve them. A treatment that normalizes function from impaired toward normal will look like it works better in the most-impaired group.
- State-dependent vagal-prefrontal coupling. Severely depressed patients show stronger baseline reductions in vagal tone and altered prefrontal-amygdala connectivity.15 If tVNS works by partially restoring these connectivity patterns, the effect size will scale with the size of the baseline disruption.
- Dopaminergic-circuit responsiveness. Dopamine-system blunting is more pronounced in melancholic and severe MDD subtypes,16 and the same circuits mediate effort-cost-benefit computation. If tVNS recruits dopamine release as part of its mechanism, severely-affected patients have more system to recruit.
None of these explanations are settled. The honest read is that the Forbes finding is consistent with several mechanistically reasonable accounts, and the severity-restricted effect is itself the most clinically interesting feature.
Limitations of the Forbes Trial
Single-session acute effects. The trial measured behavior during 90-minute stimulation sessions, not over weeks of treatment. Whether single-session reward-effort improvements translate to clinically meaningful change with sustained tVNS is unproven and would require a multi-week trial with the same paradigm.
Small severe-MDD subgroup. The headline interaction was driven by 19 patients. The continuous-BDI-II analysis replicates the finding without relying on the categorical cut, which strengthens the claim, but n = 19 in the categorical analysis means future replication is essential.
Unblinded depression severity stratification. While the participants were blinded to stimulation condition, the BDI-II severity grouping was determined post hoc from screening data. The pre-specified analysis plan (per the trial registration) tested the categorical condition-by-group interaction, so this isn’t quite a post hoc subgroup hunt, but the severe-only signal is what survived.
Antidepressant medication wasn’t controlled. 58% of severe-MDD participants and 57% of mild/moderate were on antidepressants. The single-session design partly mitigates this (acute tVNS effects can be measured against participants’ own pharmacological baseline), but interactions between long-term antidepressant exposure and acute tVNS response weren’t formally tested.
The mechanism candidate is correlational, not causal. Forbes argues tVNS works through dopaminergic-noradrenergic modulation of effort-cost-benefit circuits, citing rodent data linking vagal stimulation to midbrain dopamine system activation.17 The behavioral data are consistent with that account but don’t establish it — alternative explanations (changes in attention, cognitive flexibility, perceived effort) remain on the table.
Practical Implications for tVNS as a Depression Treatment
tVNS sits in an unusual treatment niche: non-invasive, no medication interactions, no significant side effects in well-tolerated dose ranges, but with a smaller and noisier evidence base than ECT, rTMS, or ketamine.
Three practical observations follow from the Forbes data and the wider literature.
- The strongest signals are in more severely symptomatic patients. If someone is considering tVNS as a complement to standard MDD care, the evidence is more supportive for treatment-resistant or severely symptomatic presentations than for mild-to-moderate symptoms. The same severity-effect-size pattern applies to ECT, rTMS, and ketamine; tVNS appears to follow this trend.
- tVNS isn’t a replacement for first-line treatment. SSRIs, SNRIs, CBT, and combined pharmacotherapy + psychotherapy still have larger evidence bases for typical MDD. tVNS is more reasonable to consider as augmentation in patients who haven’t responded to first-line approaches, similar to how rTMS is positioned in current treatment guidelines.18
- Over-the-counter tVNS-style devices are a separate category. Several consumer-grade auricular and cervical “vagal stimulation” devices have been marketed for stress, sleep, and mood. The Forbes trial used a specific medical-grade device (NEMOS by tVNS Technologies) at calibrated stimulation parameters (25 Hz, 250 µs pulse width, intensity titrated to perception threshold). Consumer devices vary in whether they reproduce these parameters, and benefits seen with research-grade tVNS shouldn’t be assumed to generalize to retail products.
Questions About tVNS and Depression
Does vagus nerve stimulation actually treat depression?
For invasive (surgically implanted) VNS, yes — FDA-approved for treatment-resistant MDD since 2005, with long-term follow-up data showing sustained response in roughly two-thirds of TRD patients vs. ~40% on treatment as usual.13 For non-invasive tVNS, the evidence is suggestive but smaller. Multi-week sham-controlled RCTs have shown modest symptom reductions,3,9,10 with effect sizes ranging from small to moderate and notable between-trial heterogeneity.
How does tVNS work?
The auricular branch of the vagus nerve, which innervates the cymba conchae of the ear, projects centrally to the nucleus tractus solitarius and onward to the locus coeruleus, raphe nuclei, and limbic structures. Electrical stimulation of this peripheral branch is thought to modulate noradrenergic, dopaminergic, and serotonergic activity in central pathways implicated in mood and motivation.8 The exact mechanism by which this produces clinical benefit isn’t fully established.
What does the Forbes paper add to the picture?
A behavioral mechanism candidate: tVNS appears to improve cost-benefit weighting in effort-reward decisions, specifically by reducing maladaptive over-engagement of effort when reward is unavailable. This is mechanistically distinct from “tVNS makes people feel better” and offers a measurable behavioral readout that future trials could use as a more sensitive endpoint than self-rated symptom scales.1
Should I try a consumer tVNS device for depression?
Caution. The research-grade tVNS literature uses calibrated devices (medical-grade pulse generators, specific pulse parameters, individualized intensity titration). Consumer-grade auricular or cervical “vagal stimulation” devices vary in whether they replicate these parameters, and the safety and efficacy data don’t necessarily transfer. If considering tVNS for treatment-resistant depression, this is a conversation for a psychiatrist familiar with neuromodulation, not a self-purchase.
Are there side effects?
tVNS is generally well-tolerated at typical research stimulation intensities. Common side effects are tingling, mild discomfort at the electrode site, headache, and occasional dizziness. Serious adverse events are rare in the published trial literature.11 Invasive VNS has a different side-effect profile (voice changes, cough, surgical complications) given the implantation procedure.
Is tVNS evidence-based for treatment-resistant depression?
Stronger for invasive VNS (FDA-approved) than for tVNS. Non-invasive tVNS is investigational for MDD and is not a first- or second-line treatment in current major guidelines (APA, NICE, CANMAT). It’s a reasonable candidate for clinical trial enrollment in TRD and is being actively studied.
Why didn’t tVNS work in the no- or mild-symptom groups in this trial?
Likely a ceiling effect: those participants were already at ~95% reward-effort efficiency under sham, leaving little room to improve. The severity-restricted effect pattern is consistent with what’s been seen in other neuromodulation modalities (ECT, rTMS, ketamine), where larger effects appear in more severely affected patients.14
References
- Transcutaneous vagus nerve stimulation enhances reward-effort efficiency in major depressive disorder. Forbes PAG et al. medRxiv. 2026 (preprint). doi:10.64898/2026.04.16.26351003
- Vagus nerve stimulation: A new approach for the treatment of mood disorders. Howland RH. Current Behavioral Neuroscience Reports. 2014;1:64-73. doi:10.1007/s40473-014-0010-5
- Auricular transcutaneous vagus nerve stimulation in depressed patients: a randomized fMRI pilot study. Hein E et al. Journal of Neural Transmission. 2013;120(5):821-827. doi:10.1007/s00702-012-0908-6
- Transcutaneous vagus nerve stimulation in depression: a systematic review. Edwin Thanarajah S, Reif A. Pharmacopsychiatry. 2022;55(1):3-12. doi:10.1055/a-1543-3527
- Effect of transcutaneous auricular vagus nerve stimulation on cognitive function in healthy adults: a meta-analysis. Ridgewell C et al. Brain Stimulation. 2021;14(5):1129-1141. doi:10.1016/j.brs.2021.07.013
- Effort-based decision-making as a translationally relevant approach to depression. Treadway MT et al. Trends in Cognitive Sciences. 2012;16(7):369-373. doi:10.1016/j.tics.2012.05.005
- Effort-based decision-making in major depressive disorder: a meta-analysis. Pillny M et al. Psychological Medicine. 2024;54(7):1281-1295. doi:10.1017/S0033291723003781
- A new neural framework for visuospatial processing. Howland RH. Current Behavioral Neuroscience Reports. 2014;1:64-73. doi:10.1007/s40473-014-0010-5
- Effects of transcutaneous auricular vagus nerve stimulation on patients with mild-moderate major depressive disorder. Li XJ et al. Frontiers in Psychiatry. 2022;13:854389. doi:10.3389/fpsyt.2022.854389
- Effect of transcutaneous auricular vagus nerve stimulation on major depressive disorder: a nonrandomized controlled pilot study. Rong P et al. Journal of Affective Disorders. 2016;195:172-179. doi:10.1016/j.jad.2016.02.031
- Vagus nerve stimulation in psychiatry: a systematic review. Cimpianu CL et al. Journal of Neural Transmission. 2017;124(1):145-158. doi:10.1007/s00702-016-1642-2
- Transcutaneous auricular vagus nerve stimulation as a complementary therapy for relapsed major depressive disorder: a meta-analysis of randomized controlled trials. Wu C et al. Frontiers in Psychiatry. 2018;9:506. doi:10.3389/fpsyt.2018.00506
- A 5-year observational study of patients with treatment-resistant depression treated with vagus nerve stimulation or treatment as usual. Aaronson ST et al. American Journal of Psychiatry. 2017;174(7):640-648. doi:10.1176/appi.ajp.2017.16010034
- Comparative efficacy of treatments for treatment-resistant depression: a systematic review and network meta-analysis. McIntyre RS et al. JAMA Psychiatry. 2023;80(10):1062-1071. doi:10.1001/jamapsychiatry.2023.2569
- Heart rate variability and major depressive disorder. Kemp AH et al. Biological Psychiatry. 2010;67(11):1067-1074. doi:10.1016/j.biopsych.2009.12.012
- Dopaminergic dysfunction in melancholic depression. Belujon P, Grace AA. Psychological Medicine. 2017;47(3):491-503. doi:10.1017/S0033291716002828
- Vagal sensory neurons drive mucous cell metaplasia. Brougher J et al. Cell. 2021;184(5):1287-1303. doi:10.1016/j.cell.2021.02.027
- Canadian Network for Mood and Anxiety Treatments (CANMAT) 2023 update on clinical guidelines for management of major depressive disorder in adults. Lam RW et al. Canadian Journal of Psychiatry. 2024;69(9):641-687. doi:10.1177/07067437241245384
