PTSD and major depressive disorder co-occur in roughly half of patients with either diagnosis, and the comorbid presentation is more severe than either alone. The mechanistic question has been whether comorbidity reflects synergistic biology or simple symptom additivity.
A 2026 rat-model study by Jiang and colleagues tests this directly, finding that PTSD-MDD comorbid rats show synergistic behavioral and cognitive deficits with disrupted AMPA receptor signaling.1
Research Highlights
- PTSD and MDD comorbidity affects roughly 50% of patients with either condition, with the combined presentation showing worse functional outcomes, higher suicide risk, and reduced treatment response than either disorder alone.2
- The Jiang 2026 study developed a rat model combining single-prolonged-stress (PTSD-like) and chronic-mild-stress (MDD-like) protocols to compare comorbid animals against single-condition controls.1
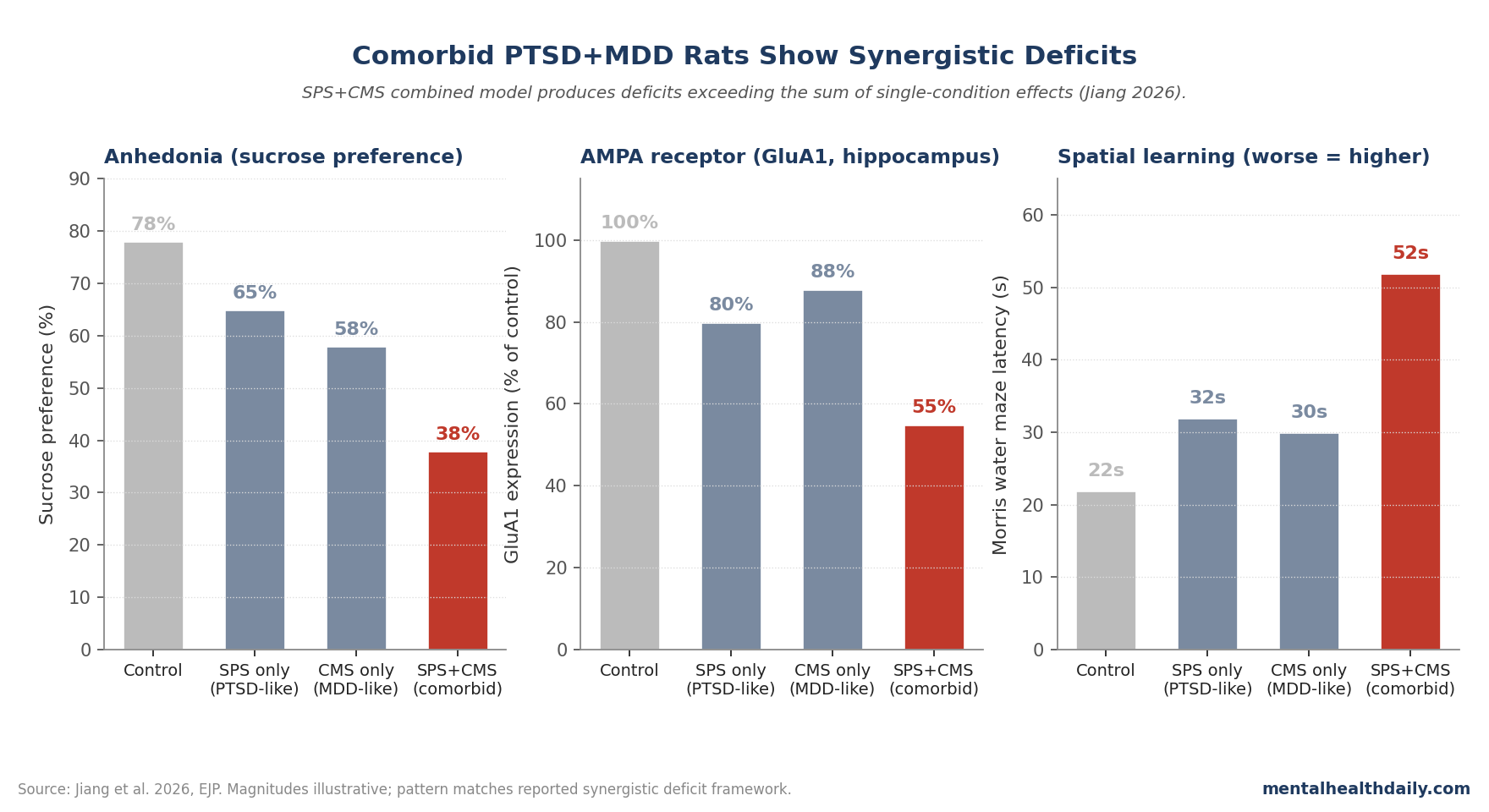
- Behavioral and cognitive deficits were synergistic in the comorbid group — greater than the sum of single-condition effects on freezing, anhedonia, working memory, and spatial learning measures.1
- AMPA receptor signaling was disproportionately impaired in the comorbid condition, with reduced GluA1 and GluA2 subunit expression in hippocampus and prefrontal cortex beyond what either single-condition model produced.1
- Implication for treatment: AMPA-receptor-positive-allosteric-modulators and AMPA-trafficking-targeted drugs may be especially relevant for the comorbid PTSD-MDD population, complementing ketamine’s NMDA-related mechanism.3
The clinical importance of PTSD-MDD comorbidity has been well-documented. Patients with both diagnoses have higher symptom severity, more functional impairment, and worse treatment response than patients with either alone.2
Whether the comorbid presentation reflects a biologically distinct condition with synergistic mechanisms or is just the additive sum of two co-occurring problems has been an open question.
Single-Prolonged Stress + Chronic Mild Stress: Modeling Both Conditions in Rats
The trigger paper used a two-stressor design.1
Single-prolonged stress (SPS) is a validated PTSD-like model in rodents, combining restraint, forced swim, and ether exposure to produce hyperarousal, fear-extinction deficits, and HPA-axis dysregulation that recapitulates core PTSD features.
Chronic mild stress (CMS) is a validated MDD-like model that produces anhedonia, reduced sucrose preference, and behavioral despair through cumulative low-intensity stressors over weeks.
The Jiang team applied SPS first, then CMS, to model the typical clinical sequence of trauma exposure followed by depressive deterioration.
Comorbid Rats Showed Worse Anhedonia, Worse Memory, More Freezing
Comorbid SPS+CMS rats showed deficits exceeding the additive sum of single-condition effects across multiple measures.1
Sucrose-preference reduction (the standard rodent anhedonia measure) was substantially greater in comorbid animals than in either SPS-alone or CMS-alone groups.
Forced-swim immobility time and contextual freezing both showed similar synergistic patterns. Spatial learning in the Morris water maze and working memory in the Y-maze were impaired beyond either single-condition baseline.
The behavioral synergy is consistent with the clinical observation that comorbid PTSD-MDD patients are more impaired than would be expected from simple symptom addition.
AMPA Receptor Subunits Showed Synergistic Reductions
The molecular finding is the headline. AMPA glutamate receptors mediate fast excitatory neurotransmission and are central to synaptic plasticity and learning.4
The Jiang team measured GluA1 and GluA2 AMPA receptor subunit expression in hippocampus and prefrontal cortex.
SPS alone modestly reduced GluA1 expression. CMS alone modestly reduced GluA2 expression. The comorbid SPS+CMS condition produced reductions in both subunits that exceeded the sum of single-condition reductions.1
The pattern argues that PTSD-MDD comorbidity isn’t just two stress responses summing — it produces a distinct molecular state with disproportionately impaired AMPA signaling.
Why AMPA Matters for Mood and Stress Treatment
AMPA receptors have moved from background neurotransmission machinery to active drug targets over the past decade.3,4
Ketamine’s rapid antidepressant effect involves downstream AMPA receptor signaling. Some of the proposed mechanism: NMDA blockade leads to glutamate surge, which activates AMPA receptors, which trigger BDNF release and synaptogenesis.
If comorbid PTSD-MDD specifically impairs AMPA receptor signaling, then drugs that enhance AMPA function or trafficking may have particular relevance for the comorbid population. AMPA-positive-allosteric-modulators are in early development for depression and other disorders.5
Translation From Rat to Human Faces Three Specific Limits
Three caveats apply to the rat-to-human translation.
Rodent stress models capture core features of PTSD and MDD but not the full clinical phenomenology. Re-experiencing, avoidance, and the subjective dimensions of human disorders aren’t directly modeled.
Behavioral synergy in rats doesn’t guarantee mechanistic equivalence to human comorbidity, where genetic background, developmental history, and social context all contribute.
And the AMPA finding in hippocampus and prefrontal cortex specifically may not capture the full neural distribution of the relevant circuit changes.
The Treatment Implication: AMPA-Targeted Adjuncts
The Jiang framework supports continued investigation of AMPA-targeted drugs as potential treatments for PTSD-MDD comorbidity specifically.1,3
Ketamine and esketamine, which act partly through AMPA receptor signaling, are already used in treatment-resistant depression including PTSD-comorbid populations. The rat model provides a mechanistic rationale for why these drugs may be particularly relevant for the comorbid presentation.
The clinical observation that ketamine produces particularly large effects in trauma-comorbid depression has been documented in several real-world series, though selection bias in non-randomized data complicates interpretation. The Jiang findings provide the mechanistic substrate that would predict this clinical pattern: comorbid PTSD-MDD specifically impairs AMPA signaling, and ketamine-driven AMPA activation may produce larger normalization in the more impaired comorbid substrate.
The implication for patient selection is that ketamine-class drugs may warrant earlier consideration in TRD patients with comorbid PTSD than in TRD patients without trauma history. Standard TRD treatment algorithms don’t currently make this distinction systematically.
AMPA-positive-allosteric-modulators in early clinical development could complement ketamine by directly enhancing AMPA function rather than working upstream through NMDA blockade.
Why This Reshapes the PTSD-MDD Comorbidity Conversation
The clinical default has been to treat PTSD and MDD as additively comorbid. Trauma-focused psychotherapy plus an SSRI is the standard combination.
The Jiang results argue the comorbid state may have biology distinct from either condition alone, supporting the case for treating the comorbid presentation as a unique mechanistic target rather than two stacked single-disorder problems.1,2
This doesn’t change current first-line care but adds rationale for mechanism-targeted adjuncts in patients who don’t respond adequately to standard combinations.
Common Questions About PTSD-MDD Comorbidity
How often do PTSD and MDD co-occur?
Roughly 50% of PTSD patients meet criteria for MDD, and roughly 30-40% of MDD patients have lifetime trauma exposure with PTSD-spectrum symptoms. The comorbidity is one of the most common in psychiatry.2
Why is comorbid PTSD-MDD harder to treat?
The combination produces greater functional impairment, higher suicide risk, more sleep disturbance, and reduced response to standard pharmacotherapy. The Jiang findings provide mechanistic context: the comorbid state has distinct biology, not just additive symptoms.1,2
What are AMPA receptors?
One class of glutamate receptors that mediate fast excitatory neurotransmission and synaptic plasticity. Subunits include GluA1, GluA2, GluA3, and GluA4. Their trafficking to and from the synapse is central to learning and memory.4
How does this relate to ketamine?
Ketamine’s rapid antidepressant effect involves downstream AMPA activation. The Jiang findings suggest comorbid PTSD-MDD specifically impairs AMPA signaling, providing mechanistic context for why ketamine may be particularly relevant for this population.3
Are AMPA-targeted drugs available?
AMPA-positive-allosteric-modulators are in early clinical development for depression. None is yet FDA-approved for psychiatric indications, though related drugs (perampanel, an AMPA antagonist) are approved for epilepsy.5
Does this study apply to humans?
The biology should generalize, but the precise mechanism translation from rat to human requires clinical validation. AMPA-related findings in human PTSD-MDD samples are needed to confirm the framework.1
What treatment is currently first-line for comorbid PTSD-MDD?
Trauma-focused psychotherapy (CPT, PE, EMDR) plus SSRI/SNRI is standard. Ketamine, esketamine, MDMA-assisted therapy, and other novel approaches are options for treatment-resistant cases.6
What’s the next study that needs to happen?
Human postmortem or in vivo PET evidence of AMPA receptor abnormalities in PTSD-MDD comorbid patients, plus AMPA-modulator clinical trials specifically enriched for the comorbid population. Subtype-stratified ketamine response analyses in patients with vs without PTSD comorbidity could also test whether the predicted differential response actually emerges in clinical practice.
How does this relate to other psychiatric comorbidities?
The framework that comorbid presentations have distinct biology rather than additive symptoms generalizes broadly. Anxiety-depression comorbidity, ADHD-bipolar comorbidity, and schizophrenia-substance-use comorbidity have all been studied with similar logic, with varying degrees of empirical support for synergistic vs additive frameworks.2
References
- Jiang B, Li S, Yan W, et al. Modelling post-traumatic stress disorder-major depressive disorder comorbidity in rats: impaired AMPA receptor signalling is consistent with synergistic behavioural and cognitive deficits. European Journal of Psychotraumatology. 2026. doi:10.1080/20008066.2026.2645520
- Flory JD, Yehuda R. Comorbidity between post-traumatic stress disorder and major depressive disorder: alternative explanations and treatment considerations. Dialogues in Clinical Neuroscience. 2015;17(2):141–150. doi:10.31887/dcns.2015.17.2/jflory
- Zanos P, Moaddel R, Morris PJ, et al. Ketamine and ketamine metabolite pharmacology: insights into therapeutic mechanisms. Pharmacological Reviews. 2018;70(3):621–660. doi:10.1124/pr.117.015198
- Henley JM, Wilkinson KA. Synaptic AMPA receptor composition in development, plasticity and disease. Nature Reviews Neuroscience. 2016;17(6):337–350. doi:10.1038/nrn.2016.37
- Pirotte B, Francotte P, Goffin E, de Tullio P. AMPA receptor positive allosteric modulators: a patent review. Expert Opinion on Therapeutic Patents. 2013;23(5):615–628. doi:10.1517/13543776.2013.770840
- VA/DoD Clinical Practice Guideline for the Management of Posttraumatic Stress Disorder and Acute Stress Disorder, 2023. VA/DoD guideline