Chronic low-grade inflammation rises with age, and a growing body of work asks whether peripheral inflammatory markers in blood track subtle changes in brain white-matter microstructure on MRI. A 2026 preprint from the Healthy Minds for Life cohort by Mishra and colleagues at Johns Hopkins examined 457 dementia-free adults aged 50–79 and found that higher plasma cytokines and CRP were associated with diffusion-MRI signs of altered white-matter organization — even after adjustment for plasma Alzheimer biomarkers.1
The associations are real but consistently small in effect size, partly explained by vascular risk, and they come from cross-sectional data on a preprint that has not yet been peer-reviewed. The honest read: peripheral inflammation appears to leave a measurable fingerprint on midlife white-matter MRI, but the clinical relevance of that fingerprint for any individual is unsettled.
Research Highlights
- Cohort and design. Mishra 2026 analyzed 457 dementia-free adults (mean age 63.8, 69% female) from the multi-site Healthy Minds for Life study, linking plasma inflammatory markers to multi-shell diffusion MRI metrics including mean diffusivity (MD), free water fraction (FWF), and orientation dispersion index (ODI).1
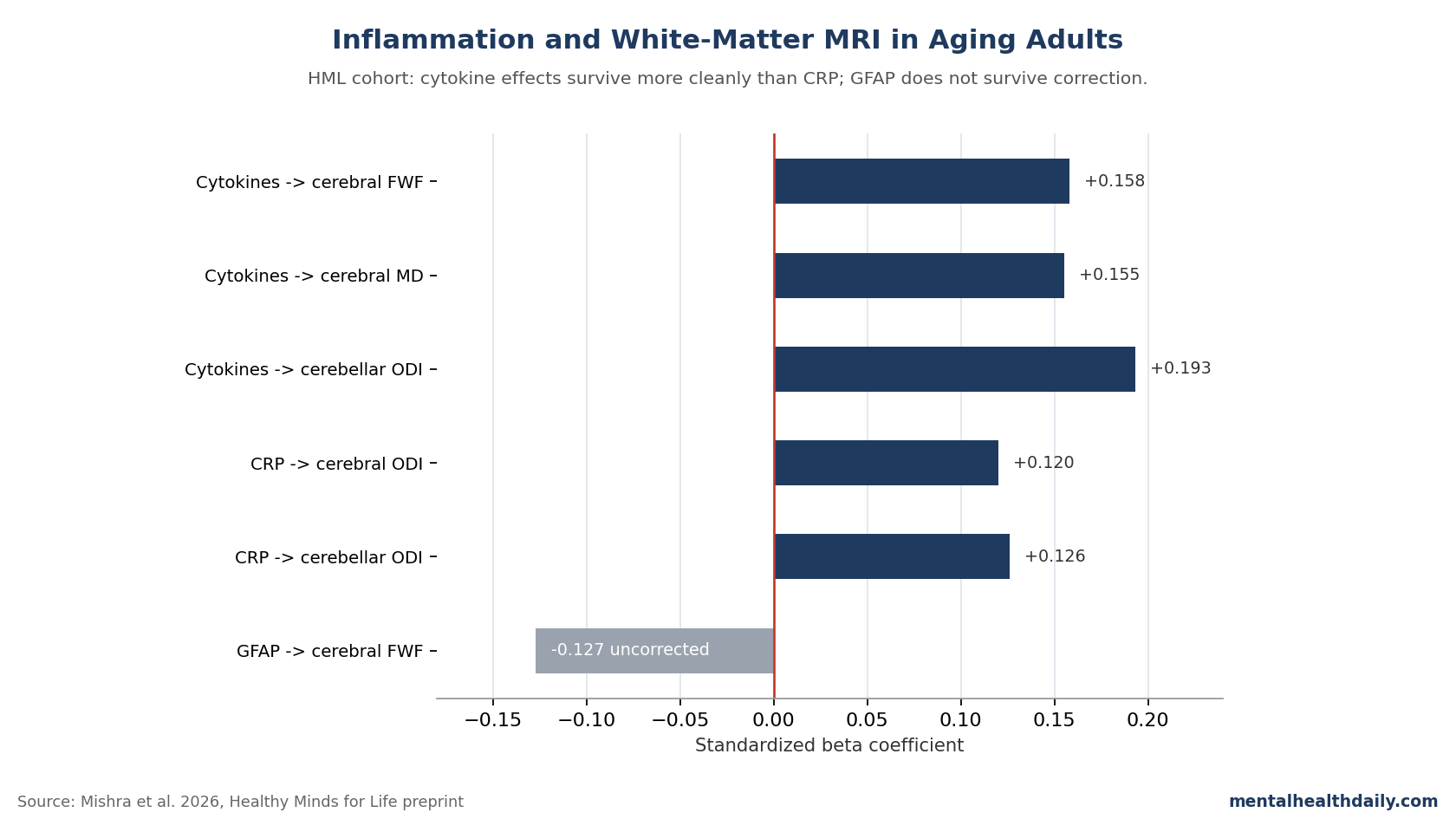
- Cytokine composite was the strongest signal. A z-scored composite of IL-1α, IL-1β, IL-2, IL-6, IL-8, TNF-α, and TNF-β predicted higher cerebral FWF (β = 0.158, p = 0.016), MD (β = 0.155, p = 0.013), and cerebellar peduncle ODI (β = 0.193, p = 0.001) after FDR correction.1
- CRP tracked dispersion, not diffusivity. High-sensitivity CRP was associated with higher cerebral and cerebellar ODI (both FDR-significant) but showed no significant link to MD — a different pattern than older single-shell DTI work.1
- GFAP washed out. Plasma glial fibrillary acidic protein, an astrocytic activation marker, did not survive multiple-comparison correction for any white-matter metric in this dementia-free sample.1
- Vascular risk explained much of CRP. When BMI and a vascular risk summary were added as covariates, CRP associations dropped out entirely; cytokine associations were more robust to vascular adjustment.1
- Effect sizes are small. Standardized betas hovered around 0.12–0.19 — a real signal, but a small fraction of total variance in white-matter metrics, leaving most individual variation unexplained.1
The Mishra preprint sits within roughly 15 years of work linking blood markers like CRP, IL-6, and TNF-α to white-matter alterations on diffusion MRI. The Atherosclerosis Risk in Communities (ARIC) cohort, the Lothian Birth Cohort, and several smaller midlife and late-life samples have all reported that higher peripheral inflammation predicts lower fractional anisotropy or higher mean diffusivity, particularly in late-life follow-ups of midlife inflammation.2,3,4
What Mishra adds is the use of NODDI — a multi-shell diffusion model that decomposes the signal into three compartments: water inside neurites, water between them, and unrestricted extracellular “free” water.5 NODDI is more biophysically interpretable than the older fractional-anisotropy framework, and animal work suggests its FWF and ODI metrics map onto astrocytic and microglial activation more directly than conventional diffusion tensor imaging.6
Healthy Minds for Life Cohort: 457 Adults Across Four U.S. Sites
Mishra 2026 drew from the Healthy Minds for Life study of the Precision Aging Network, with sites at Johns Hopkins, Arizona, Miami, and Emory. Inclusion required age 50–79, no diagnosis of memory loss or dementia, and no history of psychotic illness.1
The analytic sample of 457 was 69% female, 80% white, with mean MoCA score of 26.3 and mean BMI 28. Critically, only 4.5% had abnormal plasma p-tau181 and 11% had abnormal Aβ42/Aβ40 ratios — a largely cognitively normal, low-AD-pathology cohort.
That demographic shape matters: this is a relatively healthy late-middle-aged sample, not a memory-clinic population. Effects in such a cohort are expected to be smaller than in dementia or mild cognitive impairment groups.
Cytokine Composite Showed the Most Robust White-Matter Signal
The pro-inflammatory cytokine composite combined seven analytes — IL-1α, IL-1β, IL-2, IL-6, IL-8, TNF-α, TNF-β — each z-scored and averaged. Higher composite values were associated with all three diffusion metrics in cerebral white matter (β = 0.140–0.158, p < 0.05) and with cerebellar peduncle ODI (β = 0.193, p = 0.001).1
Three of these survived false-discovery-rate correction: cerebral FWF, cerebral MD, and cerebellar ODI. In supplementary analyses with individual cytokines, IL-1α, IL-6, and IL-8 carried most of the signal.
This finding is notable because two earlier studies in dementia-free older adults — Gu 2017 and Boots 2020 — found no significant link between individual cytokines (IL-6, IL-1β) and conventional diffusion metrics.7,8 The Mishra positive result likely reflects the gain in sensitivity from compositing across multiple cytokines plus the addition of NODDI metrics, which capture aspects of tissue organization that single-shell DTI misses.
CRP Predicted Higher ODI but Not MD — A Different Pattern Than Earlier DTI Work
High-sensitivity CRP was associated with higher orientation dispersion in both cerebral white matter (β = 0.120, p = 0.013) and cerebellar peduncles (β = 0.126, p = 0.004), both FDR-significant.1
What did not replicate from earlier cohorts: a CRP-MD association. Walker 2017 and 2018 in ARIC, and Wersching 2010 and Conole 2021 in other cohorts, all reported CRP linked to higher MD or lower FA.2,9,10
Two factors plausibly explain the discrepancy. First, those earlier cohorts had higher vascular and medical comorbidity than the Healthy Minds for Life sample. Second, when Mishra added BMI and a vascular risk summary to the model, the CRP associations disappeared entirely — suggesting the CRP-white matter link is largely a vascular-confounding signal, not an independent inflammation effect.
GFAP Did Not Survive Multiple-Comparison Correction
Plasma GFAP, a marker of astrocytic activation, showed a single uncorrected association: lower cerebral FWF at higher GFAP (β = −0.127, p = 0.023). After FDR correction across six comparisons, nothing remained.1
This null is consistent with the much larger UK Biobank analysis by Huang 2025, which found that longitudinal increases in plasma GFAP related to lower NODDI intracellular volume fraction but not to extracellular volume or orientation dispersion.11
Studies that do find robust GFAP-diffusion links tend to enroll participants further along the AD spectrum, where pathology and astrocytic activation are both more pronounced.12 The implication: in cognitively healthy midlife and early late-life adults, plasma GFAP appears to be a noisier proxy for brain inflammation than systemic cytokines or CRP — possibly because peripheral GFAP only crosses the blood-brain barrier reliably when that barrier is already compromised.13
Effect Sizes Stayed Small Across Markers
Across the significant Mishra associations, standardized β coefficients clustered in the 0.12–0.19 range. That is a real but modest effect.
For context, β = 0.16 means that a 1-SD increase in the cytokine composite predicts roughly a 0.16-SD increase in cerebral FWF — a small fraction of total variance in the imaging metric, with most variability driven by age, sex, and unmeasured factors.
The clinical translation of such effect sizes is subtle. At a population level, sustained chronic inflammation likely shifts white-matter metrics enough to matter for cognitive aging trajectories — the ARIC follow-ups show midlife inflammation predicting late-life white-matter pathology over decades.2,3 At an individual level, a single CRP or IL-6 reading provides essentially no actionable information about a person’s white-matter health.
Vascular Risk Confounding Is Doing Substantial Work
One of the more useful findings buried in the supplementary tables is what happened when BMI and vascular risk were added to the models. The cytokine composite associations remained largely intact. The CRP associations dropped out.1
This matters because CRP is the inflammatory marker most often cited in popular health coverage as a brain-aging biomarker, partly because it’s cheap and widely measured. The Mishra data argue that, in a cohort without selected vascular pathology, the CRP-white matter link is mostly a vascular-risk shadow rather than an independent inflammation effect.
Consistent with this, a 2024 systematic review of CRP and brain microstructure by Alilou and colleagues concluded that the CRP-diffusion association is real but heavily confounded by cardiovascular risk factors and obesity, with most studies failing to fully adjust for them.14
How NODDI Changes the Inflammation-White Matter Conversation
Conventional diffusion tensor imaging summarizes water diffusion in each voxel with a single tensor, yielding fractional anisotropy and mean diffusivity. Both metrics are sensitive to many tissue properties simultaneously — demyelination, axonal loss, edema, crossing fibers — making biophysical interpretation hard.15
NODDI separates the diffusion signal into three compartments: intra-neurite (axons and dendrites), extra-neurite, and free water (interstitial fluid and CSF).5 Higher FWF reflects more extracellular water, which animal models link to astrocytic activation and edema. Higher ODI reflects more disorganized neurite orientations, which animal work has tied to microglial density.6
This is why the cerebellar finding in Mishra is interesting: the cerebellar peduncles showed elevated ODI with both cytokines and CRP, but no FWF or MD signal. Animal data suggest cerebellar microglia have distinct morphology and immunoreactivity from cortical microglia, and that activated cerebellar microglia release pro-inflammatory cytokines including IL-1β and IL-6.16
Where the Mishra Preprint Falls Short
Four caveats deserve weight before reading too much into these effects.
- Cross-sectional design. All measures were taken at one time point. Whether higher cytokines cause altered white-matter microstructure, whether the relationship runs the other way, or whether a third factor drives both is unknowable from these data.
- Preprint, not peer-reviewed. The medRxiv version posted April 2026 has not gone through journal review. Methods and conclusions may shift before final publication.
- Plasma is not brain. Peripheral inflammation correlates only modestly with central inflammation in CSF or PET imaging. Plasma cytokines reflect systemic processes that may or may not translate to neuroinflammation, particularly with an intact blood-brain barrier.13
- Small effects, possibly inflated. β values in the 0.12–0.19 range in a cross-sectional design with multiple comparisons should be treated as upper bounds. Replication in independent cohorts will likely yield smaller estimates.
Practical Conclusions From the Calibrated Inflammation-Brain Evidence
For someone reading the popular-press version of this literature, the calibrated takeaway has three parts.
- Chronic systemic inflammation does track with brain aging at a group level, across multiple cohorts and imaging modalities. The signal is most consistent for composite cytokine measures and least consistent for single markers like IL-6 alone or plasma GFAP in dementia-free samples.
- Most of the CRP-brain story is probably vascular, not independent inflammation. Public-facing coverage that frames CRP as a stand-alone brain-aging biomarker overstates the cleanest interpretation of the data. Cardiovascular risk-factor management captures the same effect more efficiently.
- Individual inflammatory markers don’t yet earn a place in routine brain-health monitoring. The effect sizes are too small, the vascular confounding too pervasive, and the clinical translation too uncertain. The lifestyle interventions that lower chronic inflammation — physical activity, sleep, weight, smoking cessation — are the same interventions that protect cardiovascular and brain health through other channels.
The Mishra preprint advances the methodology by showing NODDI metrics pick up cytokine signals that single-shell DTI missed. That’s a meaningful technical step. The clinical implication for any individual reading their own CRP or IL-6 result is essentially unchanged.
Common Questions About Inflammation and Brain White Matter
Does my CRP level tell me anything about my brain health?
Not in any actionable way. CRP-brain associations across cohorts are real but small, and disappear in healthier samples once vascular risk is accounted for. CRP remains useful as a general inflammation and cardiovascular-risk screen; framing it as a brain-aging biomarker outruns the data.1,14
What is white-matter microstructure and why does it matter?
White matter is the wiring — axons wrapped in myelin connecting brain regions. Diffusion MRI measures how water moves through this tissue, and the patterns reflect how organized and intact the underlying axons and myelin are. Lower integrity correlates with slower cognitive processing speed and steeper cognitive aging trajectories.15
What’s the difference between MD, FWF, and ODI?
Mean diffusivity averages water movement in all directions. Free water fraction estimates extracellular water, which rises with edema and astrocytic activation. Orientation dispersion index measures how organized neurite directions are. NODDI computes FWF and ODI from a multi-shell diffusion sequence; older single-shell DTI only yields MD and fractional anisotropy.5
Are pro-inflammatory cytokines worse for the brain than CRP?
Mishra 2026 found cytokine composites more robust than CRP, with vascular adjustment knocking out CRP entirely. But the cytokines tested are themselves heterogeneous — IL-1α, IL-6, and IL-8 carried most of the signal in supplementary analyses. There is no single “best” brain-inflammation marker in plasma; composites do better than individual analytes.1
If inflammation correlates with brain aging, do anti-inflammatory drugs slow cognitive decline?
Trials testing NSAIDs, anti-IL-6 agents, and other anti-inflammatories for cognitive aging or dementia prevention have largely been disappointing. Observational correlation with imaging biomarkers does not translate to clinical benefit from broad anti-inflammatory treatment, partly because chronic inflammation is downstream of many lifestyle and metabolic processes.17
What about lifestyle factors that lower inflammation?
Regular aerobic exercise, weight management, adequate sleep, smoking cessation, and Mediterranean-style diets all reduce CRP and cytokine levels and are independently linked to better white-matter outcomes. The lifestyle interventions are well supported even if the inflammation-as-mediator mechanism is only partial.14,17
Why didn’t GFAP show effects in this study?
Plasma GFAP is most informative in samples with active neurodegeneration. In dementia-free midlife adults, the signal-to-noise ratio is low. Plasma GFAP also depends on blood-brain barrier integrity for reliable transit; in healthy adults it tracks central astrocytic activation imperfectly.13
Is this preprint trustworthy?
Preprints have not been peer-reviewed and conclusions can shift. The Mishra analysis uses a well-characterized multi-site cohort, standard pre-processing pipelines, and FDR correction — methodologically conservative. Independent replication in UK Biobank, ARIC, or comparable cohorts would strengthen confidence in the cerebellar ODI finding specifically.1
References
- Mishra S, Pettigrew C, Ugonna C, et al. Plasma inflammatory markers and brain white matter microstructure in late middle-aged and older adults. medRxiv preprint. 2026. doi:10.64898/2026.04.20.26351124
- Walker KA, Power MC, Hoogeveen RC, et al. Midlife systemic inflammation, late-life white matter integrity, and cerebral small vessel disease: the Atherosclerosis Risk in Communities study. Stroke. 2017;48(12):3196–3202. doi:10.1161/strokeaha.117.018675
- Walker KA, Windham BG, Power MC, et al. The association of mid- to late-life systemic inflammation with white matter structure in older adults: the Atherosclerosis Risk in Communities Study. Neurobiology of Aging. 2018;68:26–33. doi:10.1016/j.neurobiolaging.2018.03.031
- Conole ELS, Stevenson AJ, Muñoz Maniega S, et al. DNA methylation and protein markers of chronic inflammation and their associations with brain and cognitive aging. Neurology. 2021;97(23):e2340–e2352. doi:10.1212/wnl.0000000000012997
- Zhang H, Schneider T, Wheeler-Kingshott CA, Alexander DC. NODDI: practical in vivo neurite orientation dispersion and density imaging of the human brain. NeuroImage. 2012;61(4):1000–1016. doi:10.1016/j.neuroimage.2012.03.072
- Yi SY, Barnett BR, Torres-Velázquez M, et al. Detecting microglial density with quantitative multi-compartment diffusion MRI. Frontiers in Neuroscience. 2019;13:81. doi:10.3389/fnins.2019.00081
- Gu Y, Vorburger R, Scarmeas N, et al. Circulating inflammatory biomarkers in relation to brain structural measurements in a non-demented elderly population. Brain, Behavior, and Immunity. 2017;65:150–160. doi:10.1016/j.bbi.2017.04.022
- Boots EA, Castellanos KJ, Zhan L, et al. Inflammation, cognition, and white matter in older adults: an examination by race. Frontiers in Aging Neuroscience. 2020;12:553998. doi:10.3389/fnagi.2020.553998
- Wersching H, Duning T, Lohmann H, et al. Serum C-reactive protein is linked to cerebral microstructural integrity and cognitive function. Neurology. 2010;74(13):1022–1029. doi:10.1212/wnl.0b013e3181d7b45b
- O’Donovan A, Bahorik A, Sidney S, Launer LJ, Yaffe K. Relationships of inflammation trajectories with white matter volume and integrity in midlife. Brain, Behavior, and Immunity. 2021;91:81–88. doi:10.1016/j.bbi.2020.09.006
- Huang X, Han X, Chang H, et al. Associations between trajectories of plasma biomarkers for Alzheimer’s disease, brain structures, and cognitive function: a prospective cohort study in the UK Biobank. Molecular Psychiatry. 2025. doi:10.1038/s41380-025-03166-y
- Bettcher BM, Olson KE, Carlson NE, et al. Astrogliosis and episodic memory in late life: higher GFAP is related to worse memory and white matter microstructure in healthy aging and Alzheimer’s disease. Neurobiology of Aging. 2021;103:68–77. doi:10.1016/j.neurobiolaging.2021.02.012
- Youn W, Yun M, Lee CJ, Schöll M. Cautions on utilizing plasma GFAP level as a biomarker for reactive astrocytes in neurodegenerative diseases. Molecular Neurodegeneration. 2025;20(1):54. doi:10.1186/s13024-025-00846-9
- Alilou S, Valizadeh P, KamaliZonouzi S, et al. The association between C-reactive protein and human brain microstructure: a systematic review of diffusion imaging studies. Brain, Behavior, and Immunity Integrative. 2024;8:100092. doi:10.1016/j.bbii.2024.100092
- Jelescu IO, Fieremans E. Sensitivity and specificity of diffusion MRI to neuroinflammatory processes. In: Advances in Magnetic Resonance Technology and Applications. Vol 9. Elsevier; 2023:31–50. doi:10.1016/b978-0-323-91771-1.00010-1
- Stoessel MB, Majewska AK. Little cells of the little brain: microglia in cerebellar development and function. Trends in Neurosciences. 2021;44(7):564–578. doi:10.1016/j.tins.2021.04.001
- Miller AH, Raison CL. The role of inflammation in depression: from evolutionary imperative to modern treatment target. Nature Reviews Immunology. 2016;16(1):22–34. doi:10.1038/nri.2015.5