Parkinson’s disease has a years-to-decades prodromal stage where motor symptoms haven’t yet emerged but pathology is accumulating. A 2026 review by Schaeffer et al. synthesizes the case for (and limits of) intervening during this window — with implications for high-risk individuals weighing whether to enter preventive trials.1
Research Highlights
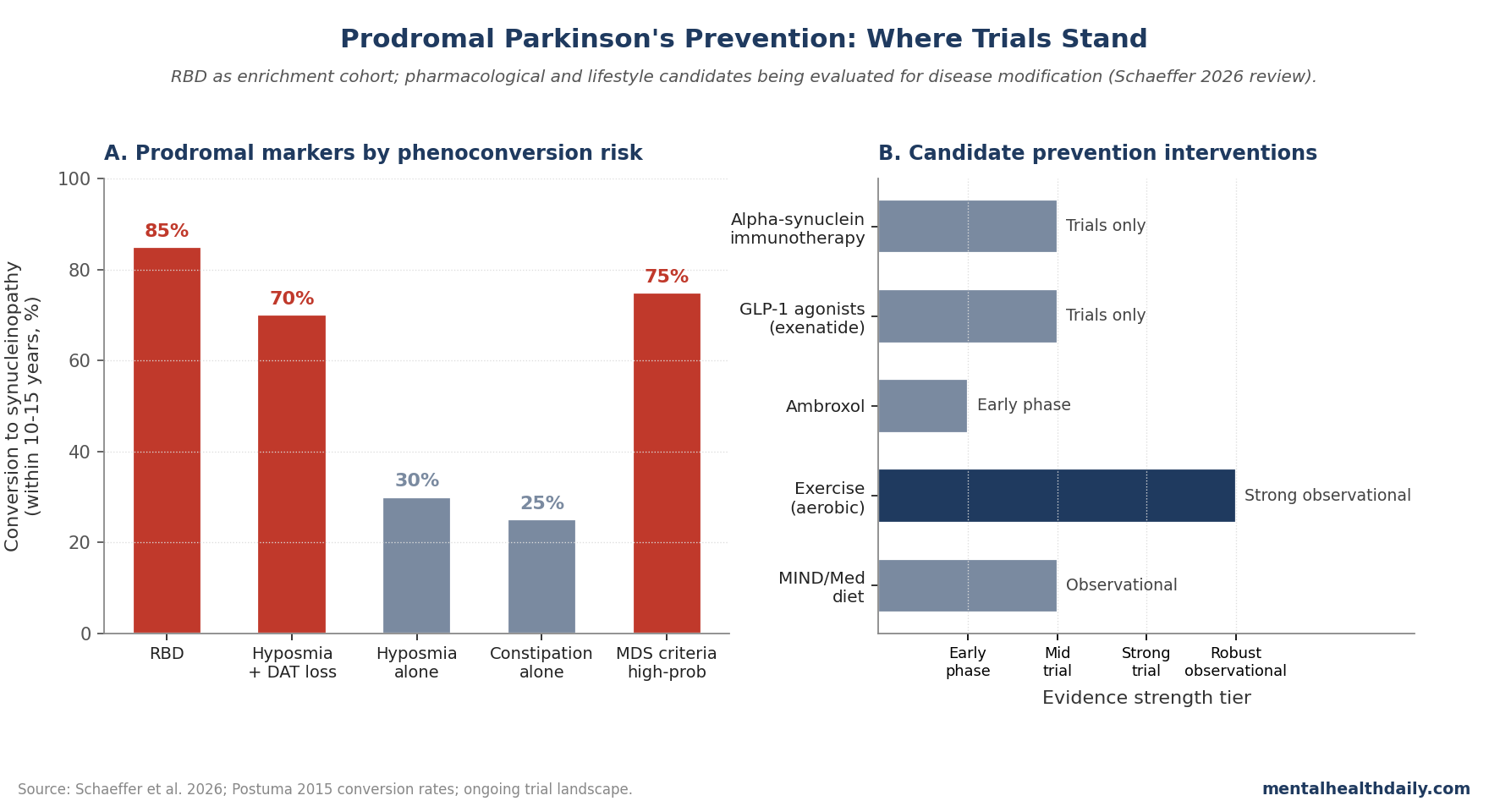
- Prodromal Parkinson’s disease is the period before motor symptoms emerge, characterized by non-motor features including REM sleep behavior disorder (RBD), hyposmia, constipation, depression, and orthostatic hypotension.2
- RBD is the strongest known prodromal marker. Roughly 80–90% of patients with idiopathic RBD develop a synucleinopathy (Parkinson’s, DLB, or MSA) within 10–15 years.3
- The Schaeffer 2026 review integrates prodromal-stage biomarkers, candidate disease-modifying interventions, and the evolving framework for prevention trials in at-risk individuals.1
- Promising candidates include alpha-synuclein-targeted immunotherapies, GLP-1 receptor agonists, ambroxol, and lifestyle interventions (exercise, dietary patterns).1,4
- Current evidence does not support routine preventive medication in prodromal cases. Trials are ongoing; clinical decisions in real-world prodromal patients should be individualized with specialist input.1
Parkinson’s prevalence is rising globally, and the disease’s long prodromal phase has been increasingly characterized through case-finding studies of RBD cohorts and biomarker-positive populations.2,5
The Global Burden of Disease estimates project that PD prevalence will roughly double by 2040 worldwide, driven primarily by population aging. Whether disease-modifying treatments emerge before that demographic wave reshapes elder care depends substantially on how quickly prevention research can translate.
The prodromal-stage approach has theoretical advantages over symptomatic-stage approaches: pathology is less established, brain reserve is greater, and early intervention may produce larger absolute benefits than the same intervention applied later.
Whether the theoretical advantages translate to actual clinical effectiveness is the key empirical question that ongoing trials are designed to answer.1,2
The Movement Disorder Society research criteria for prodromal PD use a probabilistic framework integrating multiple risk markers. High-risk individuals can be identified, but the question of what to do once identified has lagged the case-finding capability.
Without proven preventive interventions, identifying prodromal cases creates an awareness gap that can produce psychological distress without offering meaningful action options — a tension the field is actively working to resolve through trial-enrollment infrastructure and lifestyle-intervention guidance.
Schaeffer 2026: Where Prevention Trials Stand
The trigger paper organizes the prevention conversation around three questions:
Who should be in prevention trials? RBD-positive individuals are the strongest signal; biomarker positivity (alpha-synuclein seed amplification assay, DAT scan abnormalities) refines the at-risk pool. The Schaeffer review favors enriched-cohort designs over broader screening.1
What outcomes should be measured? Phenoconversion to motor PD remains the gold-standard endpoint, but it requires long follow-up. Surrogate biomarkers (alpha-synuclein burden, dopaminergic imaging change) are increasingly used for shorter trial designs.
Which interventions show prodromal promise? Three pharmacological agents plus lifestyle:
- Alpha-synuclein-targeted therapies — immunotherapies (prasinezumab, cinpanemab) targeting aggregated alpha-synuclein. Phase 2 data in early symptomatic PD have been mixed; prodromal-stage trials are theoretically better positioned but harder to execute.6
- GLP-1 receptor agonists — exenatide and lixisenatide showed signal in symptomatic PD trials. Mechanism likely involves brain insulin signaling, neuroinflammation, and possibly direct neuronal protection. Prodromal-stage testing is plausible.4
- Ambroxol — a GBA-pathway-modulating drug repurposed from respiratory medicine. Targets glucocerebrosidase function, particularly relevant in GBA-associated PD. Early-phase trials underway.7
Lifestyle interventions — exercise has the most robust evidence as a modifying factor across PD literature. Mediterranean and MIND dietary patterns show observational signals.8
Exercise specifically deserves more emphasis given how often it’s underweighted in clinical conversations.
Multiple longitudinal cohorts show that regular vigorous exercise associates with substantially reduced PD incidence, with hazard ratios of roughly 0.5–0.7 in the most active quartile compared to the least active.
Mechanistically, exercise increases brain-derived neurotrophic factor, supports dopamine-system function, and improves mitochondrial health — all relevant to PD pathophysiology.
Trial-grade evidence for prodromal exercise prevention is limited, but the combination of mechanism plausibility, observational replication, and minimal side-effect profile makes exercise a defensible recommendation even before randomized trial confirmation.
The Schaeffer review explicitly endorses lifestyle interventions as part of prodromal-stage care alongside ongoing pharmacological trial development.1,8
Why RBD Is the Anchor Cohort
RBD’s 80–90% conversion rate to a synucleinopathy makes it the most efficient enrichment cohort for prevention trials.
Other prodromal markers (hyposmia alone, constipation alone) have lower conversion rates and require larger samples for adequate statistical power.3
This creates an ethical and practical tension. RBD patients are at high risk and could potentially benefit from intervention; they’re also the most informative trial population. Some RBD cohorts now offer trial enrollment as part of routine care, with explicit informed consent about phenoconversion risk.1
The disclosure question itself is ethically complex. Should newly-diagnosed RBD patients be told their phenoconversion risk approaches 80–90%?
Some specialists argue full disclosure is essential for informed consent and trial enrollment; others worry about psychological harm from learning about a high-probability future disease without proven preventive options.
Patient-preference research suggests most RBD patients want full information when offered the choice, even with the psychological burden it carries.
The emerging consensus favors structured disclosure with mental-health support and clear pathways to trial enrollment, rather than withholding information.
As prevention-trial infrastructure expands, the value of early identification and disclosure rises further.3
Limitations of the Prevention Framework
Three caveats deserve weight:
- Phenoconversion is a slow endpoint. Even in RBD cohorts, the average time to conversion is 8–12 years. Trials need long follow-up or validated surrogate endpoints.
- Trial-positive results in symptomatic PD don’t guarantee prodromal benefit. Mechanisms operating in early symptomatic disease may differ from those in pure prodromal stages.
- Identification of at-risk individuals is uneven. RBD diagnosis requires polysomnography; biomarker testing access is limited. Equity in case-finding is an ongoing concern.1
What Mainstream Coverage Often Misses
Two framings deserve calibration. First, “preventing Parkinson’s” framings sometimes imply established preventive medications exist.
Currently, no medication is approved or evidence-based for prodromal PD prevention; trials are ongoing and patients should consult movement disorder specialists.1
Second, the relationship between PD and broader brain health is real but doesn’t mean every cognitive or motor concern is prodromal PD.
Specific case-finding requires multiple converging features and specialist evaluation, not self-screening.2
Practical Implications
For individuals with RBD or other prodromal markers, the practical advice is to engage with a movement disorder specialist who can characterize risk and discuss trial enrollment options.
- Lifestyle interventions (regular aerobic exercise, dietary patterns, sleep hygiene) have evidence supporting brain health broadly and are reasonable to pursue.1,8
- The exercise-prevention framing deserves expansion. Schenkman’s phase 2 trial in de novo PD patients showed that high-intensity treadmill exercise produced measurable benefit on motor symptom progression.
- Earlier observational work by Chen and colleagues showed that midlife physical-activity levels predicted PD incidence decades later.
The combination supports a continuum where exercise is potentially preventive in prodromal populations and disease-modifying in early symptomatic populations.
The mechanism likely combines several plausible pathways:
- BDNF and other neurotrophic factor upregulation
- Mitochondrial biogenesis in dopaminergic neurons
- Anti-inflammatory effects
- Improved cerebrovascular function
None of these mechanisms is uniquely tied to PD, which is why exercise generalizes as a brain-health intervention beyond Parkinson’s specifically.8
For clinicians, growing recognition of prodromal PD supports more systematic asking about RBD-suggestive symptoms in patients with possible PD risk factors and earlier referral when warranted.2
Brief screening with the RBD Single-Question Screen or the RBD Screening Questionnaire takes only minutes and identifies patients who warrant polysomnography for definitive diagnosis.
Combined with hyposmia testing (Sniffin’ Sticks or UPSIT) and orthostatic-vital-signs assessment, primary-care prodromal screening becomes feasible within standard appointment times.
Questions About Prodromal Parkinson’s Disease
What is prodromal Parkinson’s disease?
The years-to-decades period before motor symptoms (tremor, rigidity, bradykinesia) emerge, characterized by non-motor features and accumulating brain pathology. The Movement Disorder Society has published research criteria for identifying prodromal cases probabilistically.2
What is REM sleep behavior disorder?
A parasomnia where the normal muscle paralysis during REM sleep is absent, allowing patients to act out dreams — sometimes vigorously. Idiopathic RBD is one of the strongest known prodromal markers for synucleinopathy.3
Can Parkinson’s be prevented today?
No medication is currently approved for prevention in at-risk individuals. Lifestyle factors (exercise, dietary patterns, sleep) have observational evidence supporting general brain health. Trials of pharmacological prevention are ongoing.1,8
What about GLP-1 drugs like Ozempic?
GLP-1 receptor agonists have shown signal in symptomatic PD trials and are theoretical candidates for prodromal prevention. Trials in early and prodromal PD are ongoing. There’s no current evidence base for routine clinical use as prevention.4
Should I get tested for prodromal markers?
If you have suggestive symptoms (acted-out dreams, loss of smell, constipation, orthostatic dizziness), discussion with a primary care physician and possibly a movement disorder specialist is reasonable. Routine screening of asymptomatic individuals isn’t supported. The Michael J. Fox Foundation and Parkinson’s Foundation maintain trial-enrollment infrastructure for prodromal populations and can connect interested individuals to participating sites.2
What is the alpha-synuclein seed amplification assay?
A laboratory test detecting misfolded alpha-synuclein in cerebrospinal fluid or other tissues. Highly sensitive and specific for synucleinopathies; emerging as a research and clinical biomarker.5
Should I exercise to prevent Parkinson’s?
Regular aerobic exercise has the strongest observational evidence as a protective factor. While not proven preventive at the individual level, it’s broadly supported as part of brain-healthy lifestyle. Specific recommendations supported by current evidence include moderate-intensity aerobic activity at least 150 minutes per week, with vigorous-intensity activity producing additional benefit. Exercise types matter less than consistency and intensity.8
What’s the next study that needs to happen?
Adequately powered prevention trials in RBD and biomarker-positive cohorts with phenoconversion or validated surrogate endpoints, plus better tools for prodromal case-finding in primary care settings.
References
- Schaeffer E, Becktepe JS, Brockmann K, et al. Preventing Parkinson’s disease in the context of movement disorders: a narrative review of current evidence and future directions. Neurological Research and Practice. 2026;8:27. doi:10.1186/s42466-026-00488-2
- Berg D, Postuma RB, Adler CH, et al. MDS research criteria for prodromal Parkinson’s disease. Movement Disorders. 2015;30(12):1600–1611. doi:10.1002/mds.26431
- Postuma RB, Iranzo A, Högl B, et al. Risk factors for neurodegeneration in idiopathic rapid eye movement sleep behavior disorder: a multicenter study. Annals of Neurology. 2015;77(5):830–839. doi:10.1002/ana.24385
- Athauda D, Maclagan K, Skene SS, et al. Exenatide once weekly versus placebo in Parkinson’s disease: a randomised, double-blind, placebo-controlled trial. Lancet. 2017;390(10103):1664–1675. doi:10.1016/s0140-6736(17)31585-4
- Coughlin DG, Shifflett B, Farris CM, et al. α-Synuclein seed amplification assay amplification parameters and the risk of progression in prodromal Parkinson disease. Neurology. 2025;104(5):e210279. doi:10.1212/WNL.0000000000210279
- Pagano G, Taylor KI, Anzures-Cabrera J, et al. Trial of prasinezumab in early-stage Parkinson’s disease. New England Journal of Medicine. 2022;387:421–432. doi:10.1056/nejmoa2202867
- Mullin S, Smith L, Lee K, et al. Ambroxol for the treatment of patients with Parkinson disease with and without glucocerebrosidase gene mutations: a nonrandomized, noncontrolled trial. JAMA Neurology. 2020;77(4):427–434. doi:10.1001/jamaneurol.2019.4611
- Schenkman M, Moore CG, Kohrt WM, et al. Effect of high-intensity treadmill exercise on motor symptoms in patients with de novo Parkinson disease: a phase 2 randomized clinical trial. JAMA Neurology. 2018;75(2):219–226. doi:10.1001/jamaneurol.2017.3517