Cognitive fluctuations are a defining feature of dementia with Lewy bodies (DLB), helping distinguish it from Alzheimer’s disease — but they remain notoriously difficult to characterize and measure. A 2026 review by Mahajan and colleagues maps the neurobiology, measurement, and clinical implications of fluctuations across the Lewy body spectrum.1
Research Highlights
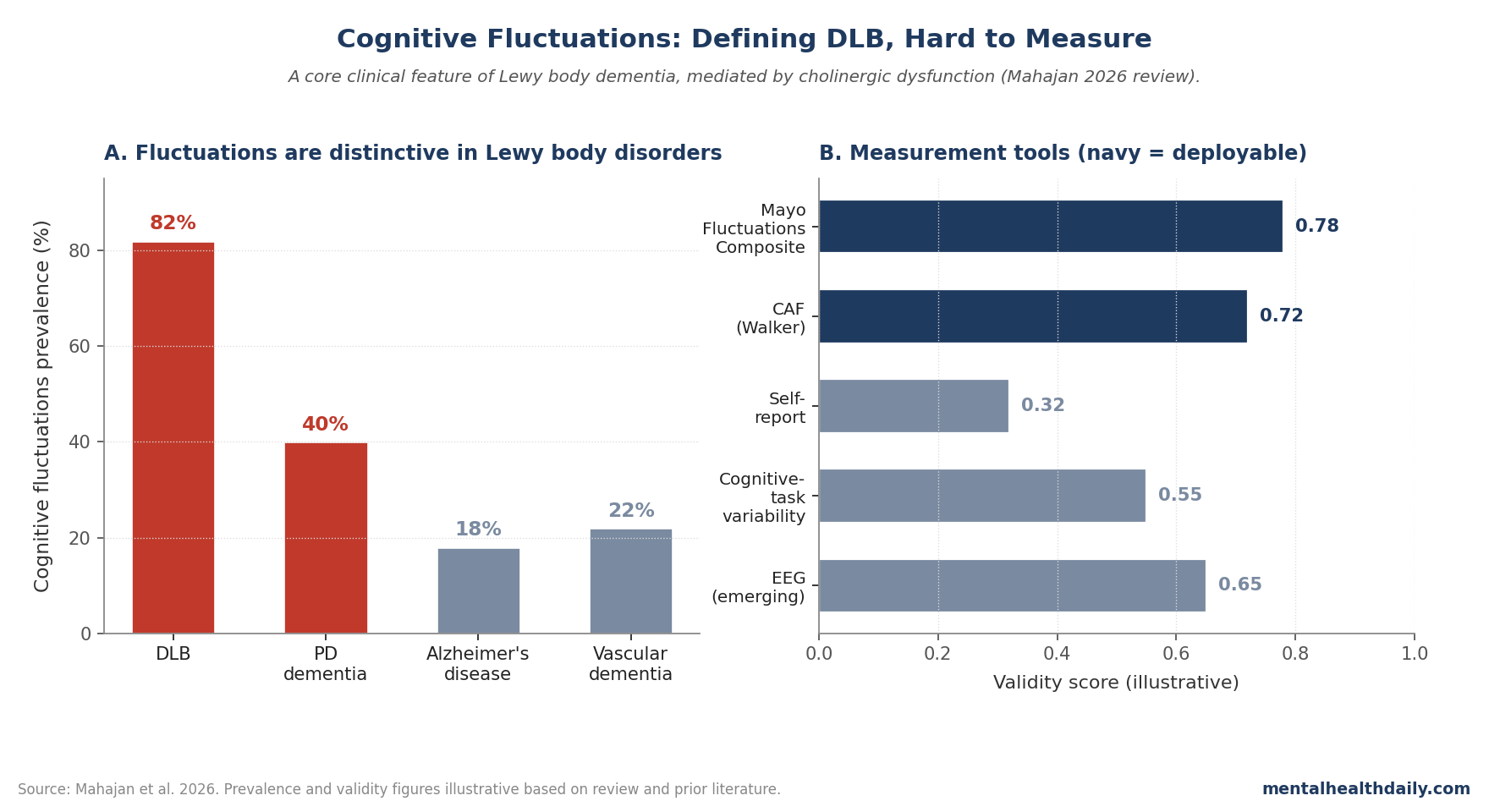
- Cognitive fluctuations are episodes of altered attention, alertness, and cognitive performance that can last seconds to days. They occur in ~75–90% of DLB patients and ~30–50% of Parkinson’s disease dementia patients, far less commonly in Alzheimer’s disease.2
- The Mahajan 2026 review integrates neuroimaging, electrophysiology, neuropsychiatric, and clinical-measurement evidence on fluctuations across the Lewy body disorder spectrum.1
- Underlying neurobiology implicates cholinergic dysfunction, thalamic circuit disruption, and altered cortical arousal — consistent with the cholinergic-deficit pattern in DLB and the partial response to cholinesterase inhibitors.3
- Measurement remains the hardest piece. Self-report is unreliable; clinician-rated scales (Mayo, Clinician Assessment of Fluctuation) have moderate validity; ambulatory tracking and EEG-based markers are emerging but not deployable.1,4
- Clinical implications: fluctuation severity tracks safety risk (falls, accidents), caregiver burden, and antipsychotic sensitivity. Better measurement could refine diagnosis and treatment matching.1,5
The 2017 DLB consensus criteria list cognitive fluctuations among the four core clinical features alongside REM sleep behavior disorder, parkinsonism, and visual hallucinations.2 Fluctuations are highly distinctive when present — AD patients show day-to-day cognitive variability but rarely the dramatic short-timescale episodes that DLB produces. The pattern of multiple core features co-occurring in DLB — fluctuations plus REM sleep behavior disorder plus visual hallucinations plus parkinsonism — helps distinguish the syndrome from AD even when individual features overlap with other dementias.
The distinctiveness has significant diagnostic value precisely because DLB is otherwise often confused with AD or with Parkinson’s disease dementia. A patient who shows clear fluctuations — episodes of staring, periods of decreased alertness lasting minutes to hours, dramatic shifts between coherent conversation and confused presentation across the same day — is much more likely to have DLB than typical AD. This matters clinically because DLB has a different treatment profile than AD: cholinesterase inhibitors are more responsive in DLB, antipsychotics are more dangerous, and the prognosis includes additional features (visual hallucinations, REM sleep behavior disorder, parkinsonism) that AD doesn’t typically produce. Recognizing fluctuations early can change the entire trajectory of clinical care.2,5
Mahajan 2026: A Comprehensive Review of Fluctuations
The trigger paper is a comprehensive review covering definition, mechanism, measurement, and clinical implications of cognitive fluctuations in Lewy body disorders.1 Three core themes:
- Phenomenology: Fluctuations span attentional lapses, episodes of unresponsiveness or decreased alertness, transient confusional states, and pronounced day-to-day variability in cognitive performance.
- Mechanism: Cholinergic dysfunction is the most replicated mechanism, with thalamic involvement in the disrupted attentional network. EEG studies show altered alpha rhythm dominance, intermittent slowing, and reduced complexity.
- Measurement: Multiple scales exist, with the Mayo Fluctuations Composite and the Clinician Assessment of Fluctuation showing the best validity. Self-report is poor; informant report and clinician observation are more reliable.4
Why Cholinergic Dysfunction Matters
The cholinergic deficit in DLB is more severe than in Alzheimer’s disease at equivalent disease stages. Acetylcholine’s role in attention, arousal, and cortical activation is well-established — the link from cholinergic loss to cognitive fluctuations is mechanistically coherent.3
Clinical implications follow:
- Cholinesterase inhibitors (donepezil, rivastigmine) have shown benefit on cognitive fluctuations specifically, more than on global cognitive performance — consistent with the mechanism.6
- Anticholinergic medications can worsen fluctuations and should be minimized in Lewy body patients.
- Antipsychotic sensitivity in DLB — severe adverse reactions to typical and even atypical antipsychotics — is partly mediated by the cholinergic deficit and partly by D2 receptor sensitivity.5
The cholinergic-deficit framework also explains why DLB patients respond particularly well to cholinesterase inhibitors compared to AD patients receiving the same drugs. Donepezil and rivastigmine produce statistically significant improvements in cognition, fluctuations, and visual hallucinations in DLB trials, with effect sizes that exceed those typically seen in AD. The pattern reflects the more severe cholinergic deficit in DLB at equivalent disease stages: when more cholinergic signaling has been lost, restoring some of it via inhibitor mechanisms produces larger functional gains. Clinically, this argues for low-threshold cholinesterase inhibitor trials in suspected DLB, with careful monitoring for fluctuation reduction as a specific treatment-response indicator.6
Measurement: The Persistent Bottleneck
Fluctuations are difficult to measure because they happen by definition between assessments, and the most informative episodes often occur at home rather than in clinic. A patient who appears coherent during a 30-minute clinical encounter may have had a 2-hour episode of unresponsiveness the night before that the family witnessed but couldn’t document. Standard cognitive screens like MMSE or MoCA capture a single time-point and can miss fluctuating patients entirely. Three measurement approaches address this challenge:
Clinician-rated scales (Mayo, Clinician Assessment of Fluctuation, One-Day Fluctuation Assessment) capture caregiver- or clinician-observed variability. They have modest reliability and require training but are the current standard.4
Cognitive-task variability (within-session reaction-time variability, attentional lapse counts) provides objective metrics but captures only short-timescale fluctuations.
Ambulatory tracking via wearables, smartphone-based testing, and continuous EEG is emerging but not yet validated for clinical deployment. Promising signals from EEG slowing and reduced complexity correlate with clinical fluctuation severity.1
Why This Matters Clinically
Three practical implications:
First, recognizing fluctuations is critical for differential diagnosis. DLB is underdiagnosed; cognitive fluctuations are one of the most useful clinical features distinguishing DLB from AD when present.2
Second, fluctuation severity tracks safety risk. Patients with severe fluctuations are at elevated risk of falls, driving accidents, and acute decompensation requiring hospitalization. Care planning should account for this.5
Third, treatment response can be measured by fluctuation reduction even when global cognitive scores don’t improve. This may explain some heterogeneity in clinical-trial outcomes for DLB therapeutics where fluctuation-specific endpoints would have detected benefit that global scores missed.6
The caregiver burden associated with fluctuations is also important and underappreciated. Caregivers of DLB patients consistently report higher burden than caregivers of AD patients with comparable global severity, and fluctuation severity is one of the strongest specific predictors of caregiver distress. The unpredictability of episodes — not knowing whether the loved one will be coherent or confused at any given time, the social-isolation effect of canceled plans because of an unexpected episode, the safety concerns of an alert-then-suddenly-impaired loved one — produces chronic stress that exceeds what steady cognitive impairment alone tends to produce. Treatment that reduces fluctuations may improve caregiver well-being even when patient cognition itself is only modestly affected.5
Limitations of the Mahajan Review
Three caveats apply:
- Reviews summarize literature; they don’t generate new data. The mechanistic and measurement claims rest on the underlying primary literature, which has its own heterogeneity.
- Operationalization variability across studies makes meta-synthesis difficult. Different definitions of “fluctuation” can produce different prevalence estimates and treatment-effect sizes.
- Clinical trial endpoints have not standardized fluctuation measurement, limiting cross-trial comparison.
What Popular Coverage Often Misses
Two framings deserve calibration. First, “cognitive fluctuations” is sometimes confused with general dementia variability. The DLB phenomenon is more distinctive: episodes of pronounced decreased alertness, blank staring, transient confusion, distinct from gradual day-to-day variation.2
Second, fluctuations are not random. They tend to track sleep quality, illness, medication changes, and environmental factors — suggesting modifiable contributors that clinical care can target.1
Practical Implications
For clinicians, systematic fluctuation assessment using validated scales should be part of cognitive evaluation in older adults with possible Lewy body disorder.
For caregivers, recognizing fluctuations as a feature of the disorder — rather than as the patient being “difficult” or non-cooperative — can change the emotional weight of caregiving substantially.
The reframe from “he’s being stubborn” to “he’s in a low-alertness episode that will pass” reduces interpersonal conflict and reduces caregiver guilt and frustration in a chronic situation.
Education materials and DLB-specific support resources are available through the Lewy Body Dementia Association and similar organizations, and connecting families with peer support during the diagnostic period substantially reduces caregiver isolation.1,5
Questions About Cognitive Fluctuations in Lewy Body Disorders
What is dementia with Lewy bodies?
A neurodegenerative dementia caused by Lewy body pathology, characterized by cognitive impairment, fluctuations, parkinsonism, visual hallucinations, and REM sleep behavior disorder. It’s the second most common dementia after AD.2
How are fluctuations different from sundowning?
Sundowning is a circadian pattern of evening confusion seen in various dementias. DLB fluctuations are more variable in timing and more pronounced in alertness shifts, often including episodes of staring or unresponsiveness lasting seconds to minutes. Sundowning predictably coincides with evening; DLB fluctuations can occur at any time of day with relatively unpredictable timing, which makes them particularly difficult for caregivers to anticipate or manage.2
Do cholinesterase inhibitors help?
Yes. Donepezil and rivastigmine have shown benefit in DLB, particularly for fluctuations and visual hallucinations. They’re first-line cognitive treatment in DLB.6
Are antipsychotics safe in DLB?
Caution is required. DLB patients can have severe adverse reactions (neuroleptic sensitivity) including parkinsonism, autonomic dysfunction, and even neuroleptic malignant syndrome. Quetiapine and clozapine are preferred when antipsychotics are needed; haloperidol and risperidone should be avoided.5
How is DLB distinguished from Alzheimer’s?
By the presence of core clinical features (fluctuations, REM sleep behavior disorder, parkinsonism, visual hallucinations), supportive features, and biomarkers including DAT scan abnormalities and MIBG cardiac imaging.2
Can cognitive fluctuations be measured at home?
Smartphone-based attention testing and wearables show promise for fluctuation tracking but aren’t yet validated for clinical deployment. Caregiver observation and clinician-rated scales remain standard.4
Do fluctuations predict prognosis?
Severe fluctuations track caregiver burden, fall risk, and hospitalization risk. Whether they predict accelerated cognitive decline is less clear — the relationship is bidirectional with overall disease severity.1
What’s the next study that needs to happen?
Validated ambulatory fluctuation measurement combined with mechanism-targeted clinical trials testing cholinergic, glutamatergic, and other interventions specifically against fluctuation-severity endpoints.
References
- Mahajan A, Dwivedi AK, Galvin JE, et al. Cognitive fluctuations, neuropsychiatric burden and quality of life in moderate-advanced dementia with Lewy bodies: the role of dysautonomia. Neurology. 2026;106. doi:10.1212/WNL.0000000000214934
- McKeith IG, Boeve BF, Dickson DW, et al. Diagnosis and management of dementia with Lewy bodies: fourth consensus report of the DLB Consortium. Neurology. 2017;89(1):88–100. doi:10.1212/WNL.0000000000004058
- Tiraboschi P, Hansen LA, Alford M, et al. Cholinergic dysfunction in diseases with Lewy bodies. Neurology. 2000;54(2):407–411. doi:10.1212/wnl.54.2.407
- Walker MP, Ayre GA, Cummings JL, et al. The Clinician Assessment of Fluctuation and the One Day Fluctuation Assessment Scale. British Journal of Psychiatry. 2000;177:252–256. doi:10.1192/bjp.177.3.252
- McKeith I, Fairbairn A, Perry R, Thompson P, Perry E. Neuroleptic sensitivity in patients with senile dementia of Lewy body type. BMJ. 1992;305(6855):673–678. doi:10.1136/bmj.305.6855.673
- McKeith I, Del Ser T, Spano P, et al. Efficacy of rivastigmine in dementia with Lewy bodies: a randomised, double-blind, placebo-controlled international study. Lancet. 2000;356(9247):2031–2036. doi:10.1016/s0140-6736(00)03399-7