Research Highlights
- Media coverage of psychedelics is measurably more positive than coverage of antidepressants. A 2026 analysis of 6,805 newspaper articles from 2017-2024 found that psychedelic-related text was more strongly tied to positive emotion language (β = 0.21) and positive sentiment (β = 0.67), while antidepressant text was more strongly tied to risk and negative emotion (both β ≈ 0.24).1 The gap is consistent across three independent dictionaries.
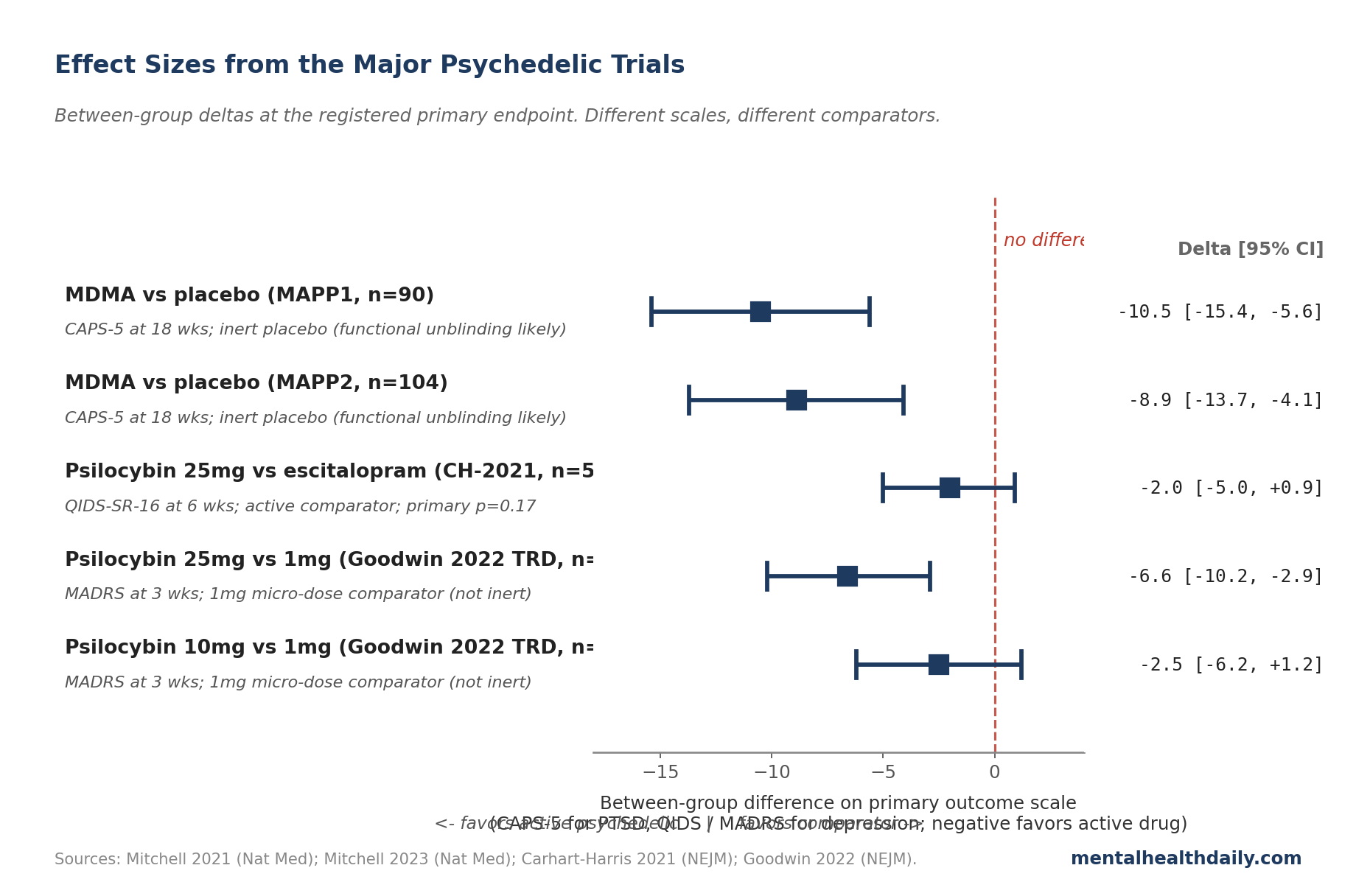
- The trial signal is meaningful but uneven. MDMA-assisted therapy for PTSD shows large between-group effects in two replicated Phase 3 trials (CAPS-5 differences of about 9-11 points vs. inert placebo).2,3 Psilocybin for depression shows smaller, less robust effects, especially against active comparators.4,5
- Functional unblinding pumps these effects up by an unknown amount. Psychedelics produce unmistakable subjective effects, so participants and raters can usually guess assignment within minutes. That breaks the trial’s blind and is a major reason the FDA rejected Lykos Therapeutics’ MDMA-PTSD application in August 2024.6
- Follow-up windows are short. The major trials’ registered primary endpoints range from 3 to 18 weeks. A separate review of 24 long-term studies found persistence at 6-12 months in some samples, but the durability evidence is observational and confounded by self-selection.7
- The honest read: signal is meaningful, evidence is immature. Calling psychedelics a cure for depression or PTSD overshoots the maturity of the data. Calling the trials fake undershoots it. The MDMA-PTSD effect is one of the larger drug-vs-placebo gaps in modern psychiatry; whether it survives a tighter blind, longer follow-up, and broader patient mix is the open question.
Popular coverage frames psychedelics as imminent breakthrough cures for depression and PTSD.
Evers and colleagues’ 2026 content analysis of nearly 7,000 newspaper articles confirms what clinicians have been saying anecdotally: media language around psychedelics tilts measurably more positive than language around FDA-approved antidepressants.1
That paper is a useful starting point, not the whole story. The deeper question is what the trial evidence supports — and the answer differs sharply between the MDMA-PTSD literature and the psilocybin-depression literature.
What Evers 2026 Measured (And What It Didn’t)
Evers and colleagues pulled 6,805 newspaper articles from LexisNexis covering psychedelics or FDA-approved antidepressants for major depressive disorder (MDD) and PTSD between 2017 and 2024. They analyzed language at the paragraph level using two computational dictionaries: LIWC (Linguistic Inquiry and Word Count, a standard psychology-research tool) and NRC (a word-emotion lexicon built by crowdsourcing).1
The pre-registered hypothesis was that psychedelic coverage would skew positive and antidepressant coverage would skew negative. The data confirmed it. Psychedelic-related paragraphs were more strongly associated with positive emotion words (β = 0.21, p < 0.001) and positive sentiment (β = 0.67, p < 0.001).
Antidepressant paragraphs were more strongly tied to risk language (β = 0.23) and negative emotion (β = 0.25), both at p < 0.001. The standardized betas (β here roughly equivalent to a correlation coefficient) are modest in absolute terms but consistent across three independent measures.
Two findings ran against predictions. Reward language — words like “benefits,” “access,” “taking” — was more associated with antidepressant paragraphs, probably because those articles discussed prescription practicalities.
The NRC negative-sentiment dictionary flagged psychedelic paragraphs as more negative than antidepressant ones, likely because words like “risk” and “dangerous” appear when journalists hedge novel-treatment stories.
What this paper doesn’t measure is whether the underlying trials justify the positive tone. That requires reading the trials.
MDMA-PTSD Phase 3: CAPS-5 Drops of -23.7 vs. Placebo’s -14.8
The MAPS-funded Phase 3 program is the strongest pillar of the modern psychedelic literature. Two trials — MAPP1 (Mitchell et al. 2021, Nature Medicine, n=90) and MAPP2 (Mitchell et al. 2023, Nature Medicine, n=104) — tested MDMA-assisted therapy against placebo-with-therapy in severe PTSD.2,3
MAPP1 reported a least-squares mean change on the CAPS-5 (the gold-standard clinician-administered PTSD severity scale) of −24.4 in the MDMA arm vs. −13.9 in the placebo arm at 18 weeks — a between-group difference of about 10.5 points (p < 0.001, Cohen’s d ≈ 0.91). MAPP2 replicated this in a moderate-to-severe sample with CAPS-5 changes of −23.7 vs. −14.8 (between-group difference 8.9 points, p < 0.001).
Those numbers are large by psychiatric-drug-trial standards. The typical SSRI-vs-placebo gap on the HAM-D (Hamilton Depression Rating Scale) is around 2-3 points; the MDMA-vs-placebo gap on CAPS-5 was roughly 9-11 points. In MAPP1, 67% of MDMA-treated participants no longer met DSM-5 PTSD criteria at study end, vs. 32% in placebo.
This is the signal that drove the FDA to grant MDMA breakthrough therapy designation in 2017 and that Lykos Therapeutics submitted for full approval in 2023. The effect is meaningful, not a hype artifact. The catch is what comes next.
Psilocybin Depression Trials: MADRS Effects Shrink Against Active Comparators
The psilocybin-depression literature has a similar shape but smaller effects, and the picture changes substantially based on what psilocybin is compared to.
The Carhart-Harris 2021 trial in The New England Journal of Medicine randomized 59 patients with moderate-to-severe depression to either two doses of psilocybin (25 mg) plus placebo escitalopram, or daily escitalopram (10-20 mg) plus two 1 mg psilocybin micro-doses.4 The primary outcome — change on the QIDS-SR-16 (a 16-item self-rated depression scale) at 6 weeks — favored psilocybin numerically (between-group difference −2.0, 95% CI −5.0 to 0.9) but didn’t reach statistical significance (p = 0.17). Several secondary outcomes favored psilocybin; the registered primary failed.
The COMPASS Pathways Phase 2b trial (Goodwin et al. 2022, NEJM, n=233) tested 25 mg, 10 mg, and 1 mg psilocybin in treatment-resistant depression (TRD).5 At 3 weeks, mean MADRS (Montgomery-Åsberg Depression Rating Scale, the most commonly used clinician-rated depression scale) reduction was −12.0 for 25 mg, −7.9 for 10 mg, and −5.4 for 1 mg. The 25 mg vs. 1 mg between-group difference was −6.6 points (95% CI −10.2 to −2.9, p < 0.001). The 10 mg dose did not separate cleanly from the 1 mg micro-dose.
By 12 weeks the 25 mg vs. 1 mg gap had shrunk and most patients had relapsed or partially relapsed. A single dose of psilocybin is not a durable monotherapy for TRD; the Phase 3 program is testing repeat dosing for that reason.
Two reads on the psilocybin literature stand together. The acute antidepressant effect is well-replicated and sometimes large — a single dose can produce a MADRS drop comparable to several weeks of SSRI treatment. The durability and the size advantage over an active comparator are weaker than coverage suggests.
Functional Unblinding: Why Psychedelic Trials Inflate Effect Sizes
Every drug trial relies on blinding: neither the patient nor the rater is supposed to know who got the active drug. That assumption fails badly with psychedelics. MDMA produces 4-6 hours of marked emotional and perceptual changes; psilocybin produces 4-6 hours of dose-dependent altered consciousness. Inert placebo doesn’t. Within the first dosing session, most participants and therapists can identify assignment with high accuracy.
This is called functional unblinding, and its effect on measured outcomes is hard to bound. The patient knows they got active drug, expects benefit (especially given the cultural enthusiasm Evers’ analysis quantifies), and reports lower symptoms; the rater knows the patient got drug, has the same expectation, and may rate more leniently. Both effects are unconscious and well-documented in trials of treatments with obvious side-effect signatures.8
Yaden and colleagues’ 2024 review of the placebo problem in psychedelic trials catalogs how badly standard inert-placebo design fails. In several psilocybin trials, more than 90% of participants correctly guessed their assignment after the first session. The implication is that headline effect sizes — the −10.5 CAPS-5 gap in MAPP1, the −6.6 MADRS gap in Goodwin — include some unknown amount of expectation effect on top of any pharmacological signal.8
Active-comparator designs reduce the unblinding problem but don’t eliminate it. The Carhart-Harris 25 mg vs. 1 mg setup still leaves the active group with strong subjective effects the comparison group lacks. The psilocybin-vs-escitalopram trial was the most defensible on blinding because both arms got something pharmacologically active — and that’s the trial whose primary outcome failed to separate.
Why Lykos’s 2024 FDA Rejection Was Predictable from the Trial Designs
In August 2024, the FDA rejected Lykos Therapeutics’ new drug application for MDMA-assisted therapy for PTSD, asking for an additional Phase 3 trial.6 The decision followed a 9-2 advisory-committee vote against approval and a sharply critical Institute for Clinical and Economic Review (ICER) report.9
The reasons tracked closely with pre-existing methodological concerns. Functional unblinding was a major theme — reviewers pointed out that the trials couldn’t separate pharmacological effect from expectation effect. The FDA also flagged irregularities in trial conduct, including allegations of therapist misconduct and protocol deviations. ICER concluded the evidence was insufficient to support approval given the design limitations.
None of this means MDMA doesn’t work for PTSD. The signal is meaningful and is one of the larger drug-vs-placebo gaps in modern psychiatry. The rejection reflected the gap between “this looks like it works” and “this has been demonstrated to a regulatory standard.” The trials weren’t designed to confidently rule out expectation effects, and the FDA wasn’t willing to make an exception.
The rejection caught psychedelic-advocacy media off guard, while methodologists and trial statisticians had been flagging the design problems for years. That gap between specialist concern and popular framing is what Evers’ 2026 paper quantifies.
Critical Thinking about Psychedelic Research for Depression and PTSD
Even taking the trial results at face value, the evidence has substantial gaps.
Follow-up windows are short. MAPP1 and MAPP2 used 18-week primary endpoints. Goodwin 2022 ran to 12 weeks. Carhart-Harris 2021 ran to 6 weeks. Aday 2020’s review of long-term psychedelic effects pulled together 24 studies suggesting persistence at 6-12 months, but most of that evidence is observational or retrospective self-report — not the same standard as the registered trial endpoints.7
Patient samples are narrow. Trials excluded active substance use disorders, recent suicidality in many cases, psychotic-spectrum diagnoses, and significant cardiovascular conditions (MDMA’s blood-pressure effects make that a real safety constraint). How psychedelics work in the broader population walking into a community clinic is largely unstudied.
Adverse-event reporting has been challenged. Re-analyses of the MDMA trials by independent groups suggested higher rates of serious adverse events than initial reports captured, including suicidality and cardiovascular events. ICER and FDA review both flagged this; some of the discrepancy is definitional, but some appears to reflect under-capture.9
The therapy component is unstandardized. “MDMA-assisted therapy” is MDMA plus 8-10 hours of preparatory and integrative psychotherapy delivered by trained dyads. The protocol varies across sites, and the active-ingredient question — how much is the drug, how much is the therapy — can’t be pinned down from existing data.
Independent replication is limited. MDMA-PTSD evidence is dominated by MAPS/Lykos trials. Psilocybin-depression evidence is more diversified across sponsors (Compass Pathways, Usona, academic groups), but the largest trials still come from a small number of labs.
Psychedelic Therapy in 2026: Practical Takeaways
For someone Googling “do psychedelics cure depression” or “is MDMA-assisted therapy approved,” the calibrated answer has three parts.
- The formal answer. As of late 2026, no classical psychedelic is FDA-approved for any psychiatric indication. MDMA-assisted therapy was rejected in 2024 and would require a new Phase 3 trial to come back. Psilocybin for TRD is in Phase 3 (Compass Pathways). Esketamine, a ketamine derivative, is approved for TRD, but the classical psychedelics aren’t.
- The trial-evidence answer. The strongest signal is for MDMA-assisted therapy in severe PTSD, with two replicated Phase 3 trials showing large between-group effects against inert placebo. Psilocybin-for-depression evidence is meaningful but smaller and more design-dependent — effects shrink against active comparators and largely wear off by the 12-week follow-up in TRD trials.
- The practical answer. The trials don’t yet support self-treatment outside research settings. Psychedelics remain Schedule I, and the patient screening, dose preparation, therapeutic environment, and integrative therapy in the trials are substantively different from recreational use. The intervention that produced the trial results isn’t the same as buying mushrooms or MDMA on the street.
Enthusiasm in popular coverage runs ahead of the maturity of the evidence, but the evidence itself contains a meaningful signal that justifies continued rigorous research. Both halves of that sentence have to land for the calibration to be accurate.
Common Questions About Psychedelic Therapy
Is psilocybin therapy FDA-approved for depression?
No. Psilocybin has FDA breakthrough therapy designation for treatment-resistant depression, granted in 2018, but designation is not approval — it’s a status that streamlines the development pathway. Phase 3 trials are ongoing as of 2026. Approval, if it comes, is likely several years out.
Is MDMA-assisted therapy approved for PTSD?
No. The FDA rejected Lykos Therapeutics’ application in August 2024, citing concerns about functional unblinding, adverse-event reporting, and protocol conduct. A new Phase 3 trial would be needed for resubmission.6
How big are the effect sizes in the major psychedelic trials?
For MDMA in PTSD, the between-group CAPS-5 difference vs. inert placebo was about 9-11 points across MAPP1 and MAPP2 (Cohen’s d roughly 0.7-0.9) — large by psychiatric-drug-trial standards. For psilocybin in depression, the effect against active comparators was smaller and not always statistically significant (Carhart-Harris 2021 primary outcome p = 0.17). The headline numbers depend heavily on what the comparator is.2,3,4,5
What is functional unblinding and why does it matter?
In a blinded trial, neither the patient nor the rater is supposed to know who got active drug. Psychedelics produce obvious subjective effects, so most participants and therapists can correctly guess assignment after the first session. That broken blind lets expectation effects pump up reported benefit, and there’s no clean way to bound how much.8
Does psychedelic-assisted therapy work better than SSRIs?
The one head-to-head trial of psilocybin vs. escitalopram (Carhart-Harris 2021) didn’t show a statistically significant advantage on the primary outcome, though several secondary outcomes favored psilocybin numerically. There is no comparable head-to-head data for MDMA vs. SSRIs in PTSD. The strong popular framing of psychedelics as superior to SSRIs is not yet supported by direct comparison data.4
How long do the benefits last?
Registered primary endpoints run 3 to 18 weeks. Aday 2020’s review found persistence at 6-12 months in some samples, but most long-term evidence is observational and confounded by self-selection.7 Single-dose psilocybin in TRD wears off for most patients by 12 weeks.5
Is taking psychedelics on my own a substitute for the trial protocols?
No. Trial interventions involve patient screening, controlled dosing, prepared therapeutic environments, and 8-10 hours of structured psychotherapy around each dose. Self-administered use lacks all of that. Cardiovascular, perceptual, and psychiatric destabilization risks are also higher without screening and monitoring.
Why is media coverage so positive if the evidence has these gaps?
Evers 2026 confirmed that psychedelic-related media language tilts measurably more positive than antidepressant-related language, partly because the cultural moment favors novel-treatment narratives and partly because antidepressant coverage has shifted toward critical reporting on withdrawal and overprescribing.1 Positive coverage doesn’t mean the trials are fake; it does mean readers should weight popular framing carefully.
References
- Media hype regarding psychedelic treatments for depression and PTSD from 2017 to 2024. Evers A et al. Scientific Reports. 2026. doi:10.1038/s41598-026-50186-x
- MDMA-assisted therapy for severe PTSD: a randomized, double-blind, placebo-controlled phase 3 study. Mitchell JM et al. Nature Medicine. 2021;27(6):1025-1033. doi:10.1038/s41591-021-01336-3
- MDMA-assisted therapy for moderate to severe PTSD: a randomized, placebo-controlled phase 3 trial. Mitchell JM et al. Nature Medicine. 2023;29(10):2473-2480. doi:10.1038/s41591-023-02565-4
- Trial of psilocybin versus escitalopram for depression. Carhart-Harris RL et al. New England Journal of Medicine. 2021;384(15):1402-1411. doi:10.1056/NEJMoa2032994
- Single-dose psilocybin for a treatment-resistant episode of major depression. Goodwin GM et al. New England Journal of Medicine. 2022;387(18):1637-1648. doi:10.1056/NEJMoa2206443
- FDA’s rejection of MDMA-assisted therapy for PTSD — reasons and implications. McKenzie K. New England Journal of Medicine. 2024;391(20):1869-1871. doi:10.1056/NEJMp2410755
- Long-term effects of psychedelic drugs: a systematic review. Aday JS, Mitzkovitz CM, Bloesch EK, Davoli CC, Davis AK. Neuroscience & Biobehavioral Reviews. 2020;113:179-189. doi:10.1016/j.neubiorev.2020.03.017
- The challenge of the placebo control in psychedelic-assisted therapy trials. Yaden DB et al. JAMA Psychiatry. 2024;81(2):117-118. doi:10.1001/jamapsychiatry.2023.4458
- The effectiveness and value of MDMA-assisted psychotherapy for post-traumatic stress disorder. Institute for Clinical and Economic Review (ICER). 2024. icer.org/assessment/ptsd-2024
