Psychosis is one of 19 neuropsychiatric syndromes the American College of Rheumatology recognizes as attributable to systemic lupus erythematosus (SLE), an autoimmune disease that disproportionately affects young women.2 A 2026 systematic review and meta-analysis from Parperis and colleagues in Lupus pooled 65 studies and 31,495 patients.1
The synthesis matters less for the headline 4.5% prevalence than for what it implies about first-episode psychosis workups: when a young woman presents with new-onset hallucinations or delusions, her ANA, anti-dsDNA, anti-ribosomal P, and complement levels carry diagnostic weight that most psychiatric services don’t routinely check.
Research Highlights
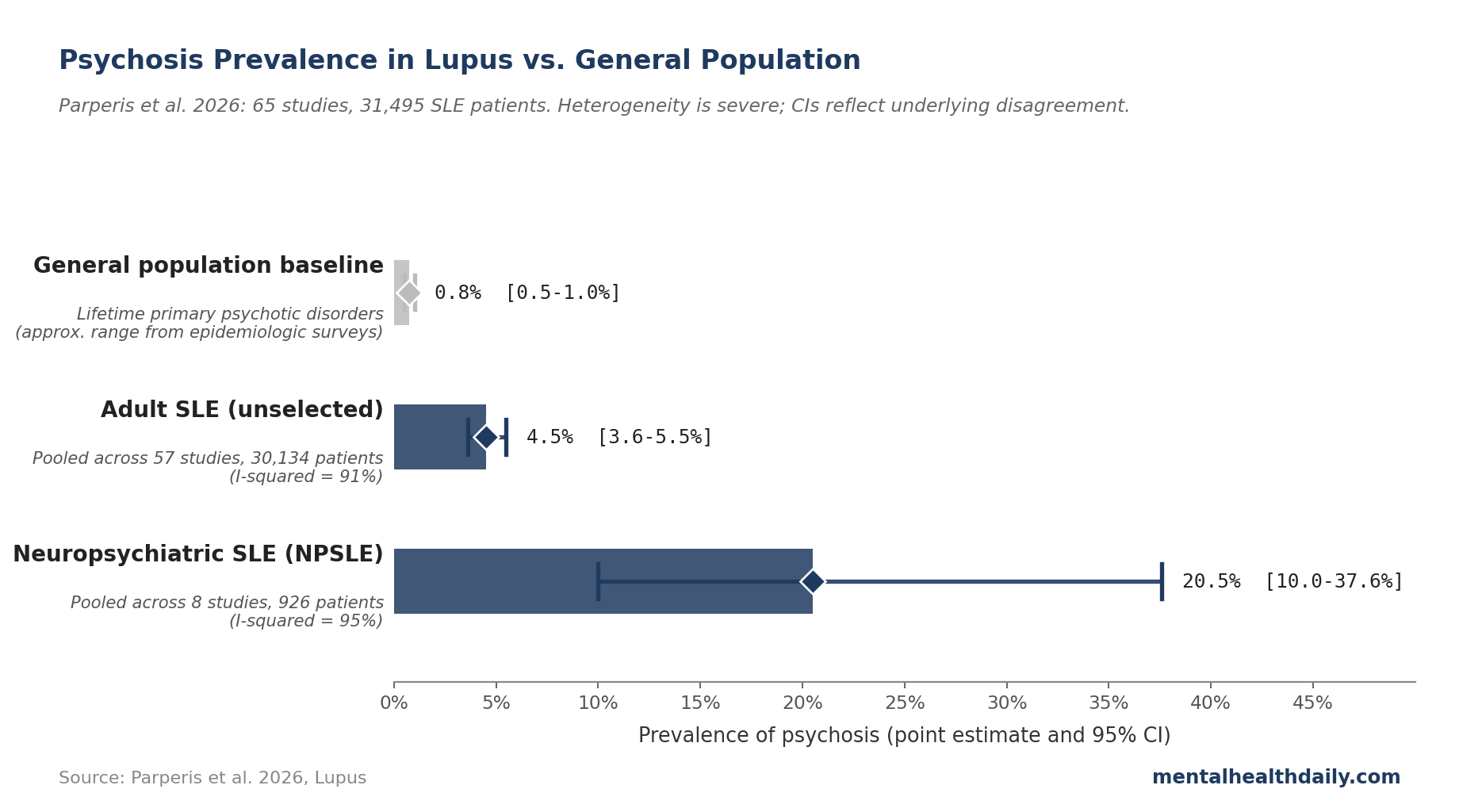
- Across 65 studies and 31,495 lupus patients, 4.5% develop psychosis (95% CI 3.6–5.5%). Among patients with neuropsychiatric lupus (NPSLE), the rate jumps to 20.5% (95% CI 10.0–37.6%).1 The 4.5% figure is roughly 50 to 100 times higher than the lifetime prevalence of primary psychotic disorders in age-matched general population samples.
- Psychosis usually shows up early. Mean interval between SLE diagnosis and first psychotic episode is 19.2 months; in several cohorts, 30 to 60% of psychotic episodes occurred at SLE onset — psychosis can be the presenting complaint, not a late complication.1
- Three immune markers track risk. Anti-ribosomal P antibodies show high specificity (around 86%) and high negative predictive value (around 97%) — a negative test argues strongly against SLE-attributable psychosis. Antiphospholipid antibodies and complement consumption (low C3, low C4) also flag elevated risk.1
- Most first-episode psychosis workups skip the lupus serologies. Standard psychiatric workups rarely include ANA, anti-dsDNA, anti-ribosomal P, or complement. SLE-attributable psychosis often gets treated as primary psychosis until rashes, joint pain, or kidney findings catch up.
- The 4.5% figure has wide error bars. Heterogeneity is severe (I² = 91%; the studies disagree wildly on what the rate is), and the meta-analysis cannot cleanly separate primary SLE psychosis from steroid-induced psychosis. The directional point: lupus is an underdiagnosed organic cause of psychosis, though the precise prevalence remains fuzzy.
Pooled Prevalence: 4.5% in SLE, 20.5% in NPSLE
The Parperis meta-analysis pooled 65 studies across Asia, Europe, the USA, and Africa, with diagnoses based on the 1999 ACR nomenclature for NPSLE.1
In unselected SLE populations, pooled prevalence of psychosis was 4.5% (95% CI 3.6–5.5%), drawn from 57 studies and 30,134 patients.1 The confidence interval (CI) — the range the true rate is most likely to sit within — is reasonably tight, but heterogeneity is severe (I² = 91%; the studies disagree wildly on the actual rate, with individual cohorts ranging from under 1% to over 17%).
Among patients already classified as having neuropsychiatric SLE, the pooled prevalence climbs to 20.5% (95% CI 10.0–37.6%).1 NPSLE is itself a heterogeneous category covering 19 syndromes from headache to seizures to cognitive dysfunction; roughly one in five of those patients carries psychotic symptoms as part of the picture.
For context, lifetime prevalence of primary psychotic disorders in general population samples runs roughly 0.5 to 1%. The 4.5% SLE rate is a 5- to 10-fold elevation over baseline; the 20.5% NPSLE rate is dramatically higher.
Anti-Ribosomal P, Antiphospholipid, and Complement: Which Antibodies Predict Risk
The most clinically useful part of the Parperis synthesis is the autoantibody data. Three immune markers track psychosis risk in SLE.
- Anti-ribosomal P antibodies are the most specific marker. Sensitivity was low (around 11%; many SLE-psychosis cases test negative), but specificity was high (around 86.5%) and negative predictive value very high (around 96.9%).1 A negative anti-ribosomal P test argues strongly against lupus as the cause; a positive test in the right clinical context substantially raises suspicion. Akbarian and colleagues found psychosis in 13% of anti-P-positive SLE versus 3% of anti-P-negative (OR 4.43, p = 0.005). Watanabe and colleagues observed that IgA and IgM anti-P levels peaked at psychosis onset and fell with remission — consistent with antibody-mediated involvement.1
- Antiphospholipid antibodies (aPL) — the same antibodies that drive clotting and pregnancy loss in antiphospholipid syndrome — track elevated psychosis risk in several SLE cohorts. The proposed mechanism is microvascular: aPL disrupt endothelial function and small-vessel flow, potentially compromising the blood-brain barrier.1,3
- Complement consumption — low C3, low C4, and low C1q — flags active immune-complex disease and tracks NPSLE risk strongly. Aso and colleagues reported low C3 with an odds ratio of 28.6 (p < 0.001) for severe neuropsychiatric flare; Magro-Checa and colleagues found low C4 with an OR of 3.8.1,4 Complement levels are cheap and routine in rheumatology and almost never drawn in primary psychiatric workups.
Anti-ribosomal P findings haven’t been fully consistent. Kampylafka and colleagues found only one anti-P-positive case among 16 psychotic SLE patients; Brey and colleagues found no significant association.1 The signal is well-replicated but not universal — consistent with anti-P being one of several converging mechanisms rather than the single causal pathway.
Why First-Episode Psychosis Workups Should Include ANA, Anti-dsDNA, and Complement
A young woman presenting with new-onset paranoid delusions or auditory hallucinations is far more likely to land in a psychiatric emergency room than a rheumatology clinic.
Standard first-episode psychosis workups typically include CBC, metabolic panel, urine toxicology, TSH, B12, RPR or HIV, and brain imaging if neurological signs are present. ANA, anti-dsDNA, anti-ribosomal P, and complement aren’t on most psychiatric checklists.
Demographics align. SLE disproportionately affects women (roughly 9:1 female:male in adult cohorts) of childbearing age, peak incidence in the 20s and 30s — the same window as first-episode psychosis. Black, Hispanic, and Asian populations have higher SLE prevalence and, in several cohorts, higher SLE-psychosis rates.1
Timing aligns. Pego-Reigosa and colleagues reported that 60% of SLE patients with psychosis had it as the initial SLE manifestation, and 80% developed it within the first year.1 Across the meta-analysis, mean interval between SLE diagnosis and psychosis onset was 19.2 months — psychosis often arrives before the rheumatologic diagnosis is made, not after.
Other lupus features can be subtle at psychosis onset. Malar rash, photosensitivity, joint pain, oral ulcers, alopecia, and Raynaud’s phenomenon often precede or accompany the psychiatric presentation but rarely make it into the psychiatric history. A targeted lupus review of systems plus a simple ANA + complement panel costs little and rules in or out a treatable organic cause.
The treatment implication is non-trivial. Primary psychotic disorders are treated with antipsychotics, often long-term. Active-disease SLE psychosis is treated with immunosuppression (high-dose corticosteroids, sometimes cyclophosphamide or rituximab) plus antipsychotics acutely, with the antipsychotic tapered as lupus comes under control.3,5 Missing the SLE diagnosis means missing the immunosuppression step.
Distinguishing Primary SLE Psychosis from Steroid-Induced Psychosis
Corticosteroids — particularly prednisone above 40 mg/day — can themselves cause psychosis, hallucinations, mania, and severe affective symptoms. Because SLE flares are commonly treated with high-dose steroids, and because psychosis often appears around flare time, separating primary SLE psychosis from steroid-induced psychosis is one of the harder bedside problems in NPSLE.
Appenzeller and colleagues, in one of the few studies that attempted formal attribution, classified 59 cases as primary CNS-SLE psychosis, 28 as corticosteroid-induced, and 2 as unrelated psychiatric disorders.6 Roughly 32% of acute psychotic episodes in their cohort were steroid-attributable rather than primary lupus — a non-trivial fraction.
Steroid attribution leans on: psychosis onset within days to weeks of starting or escalating steroids, prominent affective features (mania, hypomania), rapid resolution after dose reduction, and absence of active-lupus markers (normal complement, low SLEDAI, no other NPSLE features).3,6 Primary SLE attribution leans on: positive anti-ribosomal P, low complement at onset, concurrent NPSLE features (seizures, encephalopathy), and persistence despite steroid taper. The Bortoluzzi attribution model formalizes this with weighted criteria.7
Plausible Mechanisms: Lupus and Psychosis
Anti-neuronal antibody binding. Anti-ribosomal P antibodies cross-react with a neuronal surface protein and, in animal models, alter behavior and impair memory after intracerebroventricular injection.1,8 Whether this happens at clinically relevant titers in humans, and whether anti-P crosses an intact blood-brain barrier or only a disrupted one, isn’t pinned down. The correlational data are strong; the causal pathway is plausible but not proven in humans.
Complement-mediated synaptic pruning. The complement system tags synapses for microglial elimination, and over-active complement signaling has been implicated in schizophrenia (via C4 variants) as well as in NPSLE.9 Low circulating C3 and C4 in active SLE may reflect consumption at the synaptic level. The mechanism is biologically coherent but largely inferential.
Blood-brain barrier disruption from antiphospholipid microangiopathy. aPL antibodies promote endothelial dysfunction and microvascular thrombosis, which in cerebral microvasculature can compromise the barrier separating circulating antibodies and cytokines from brain tissue.1,3 Imaging supports diffuse small-vessel changes in NPSLE; the direct link to psychosis is less specific.
Mendelian randomization work also suggests shared genetic susceptibility between SLE and schizophrenia — some patients may be predisposed to both, with lupus inflammation acting as the second hit.1
Limitations of This Research
Heterogeneity is severe. I² was 91% for unselected SLE prevalence and 95% for NPSLE — the studies disagree wildly on the rate. Individual cohorts range from under 1% (Kampylafka, Greece) to 17% (Mikdashi, USA; Sachse, Germany). The pooled 4.5% is the central estimate of a noisy distribution.
Attribution is messy. Most studies didn’t formally distinguish primary SLE psychosis from steroid-induced psychosis or comorbid primary psychotic disorders. Roughly a third of “SLE psychosis” cases in cohorts that did careful attribution work were steroid-attributable, suggesting the pooled prevalence overstates the rate of antibody-driven psychosis specifically.
Diagnostic methods varied. Some studies used the 1999 ACR NPSLE criteria, others DSM-IV or DSM-5; some applied structured psychiatric interviews, others relied on chart review. Risk of bias was moderate to high in many cohorts: selection bias (specialty referral cohorts oversample severe disease), incomplete control for confounders, and varying follow-up duration all push against precise estimation.
Childhood-onset SLE was excluded. The meta-analysis restricted to adult SLE (age 18+), but childhood-onset lupus has higher rates of NPSLE and psychosis. The adult numbers don’t generalize to pediatric or adolescent populations.
When to Order a Lupus Panel in First-Episode Psychosis
- For first-episode psychosis with any of the following, order ANA, anti-dsDNA, anti-ribosomal P, C3, C4, and antiphospholipid antibodies. Female under 45; personal or first-degree-relative autoimmune history; rash (malar or discoid); joint pain; unexplained fatigue, fever, or weight loss; oral ulcers; Raynaud’s phenomenon; alopecia; recurrent miscarriages or thrombotic events; abnormal CBC (cytopenias).
- For known SLE patients with new psychiatric symptoms, distinguishing primary SLE psychosis from steroid-induced psychosis is the key question. Timing relative to steroid dose changes is the strongest signal.
- A negative anti-ribosomal P test is informative. Given ~97% negative predictive value, a clean anti-P substantially reduces the probability that an active psychotic episode is lupus-attributable, even in known SLE.
- A positive ANA in isolation isn’t enough. Roughly 5 to 15% of healthy women test ANA-positive at low titers without active autoimmune disease.
- Treatment for confirmed primary SLE psychosis is a rheumatology-psychiatry collaboration. Acute management combines antipsychotics with immunosuppression, with antipsychotics tapered as lupus comes under control.
Common Questions: Lupus vs. Psychosis
Can lupus cause psychosis?
Yes. The 2026 Parperis meta-analysis pooled 65 studies and 31,495 SLE patients and found 4.5% pooled prevalence of psychosis (95% CI 3.6–5.5%), rising to 20.5% in neuropsychiatric lupus.1 Psychosis is one of 19 neuropsychiatric syndromes the American College of Rheumatology recognizes as attributable to SLE.2
What does lupus psychosis look like?
Delusions and hallucinations dominate — paranoid delusions, grandiose delusions, auditory hallucinations, and visual hallucinations are the most reported features.1 Disorganized thinking and catatonia have also been documented in patients with high disease activity. The presentation overlaps substantially with primary psychotic disorders, which is part of why the diagnosis gets missed.
How early in lupus does psychosis appear?
Often very early. Mean interval between SLE diagnosis and first psychotic episode was 19.2 months. In several cohorts, 30 to 60% of psychotic episodes occurred at the time SLE was diagnosed — psychosis can be the presenting complaint that leads to the lupus workup, not a late complication.1
Should a first-episode psychosis workup include lupus testing?
For most cases, yes — particularly for women under 45 or patients with any autoimmune history, rash, joint pain, oral ulcers, alopecia, or unexplained cytopenias. ANA, anti-dsDNA, anti-ribosomal P, C3, C4, and antiphospholipid antibodies are inexpensive and informative. They aren’t on standard psychiatric checklists, which is part of why SLE-attributable psychosis is underdiagnosed.
How is lupus psychosis treated?
Acute management combines antipsychotics for symptom control with immunosuppression targeting the underlying lupus — typically high-dose corticosteroids, sometimes cyclophosphamide or rituximab for severe or refractory cases.3,5 Antipsychotics are usually tapered as the lupus comes under control. The treatment is genuinely different from primary psychosis, which is why the diagnosis matters.
Does lupus psychosis go away?
Often, yes — with appropriate immunosuppression, primary SLE psychosis frequently resolves during disease remission. Paholpak and colleagues reported a 2.9% relapse rate over 44 months.1 The trajectory is meaningfully different from primary psychotic disorders, where chronic management is the norm.
References
- Psychosis in patients with systemic lupus erythematosus: a systematic literature review and meta-analysis. Parperis K, Zis P, Evangelou M et al. Lupus. 2026;35(6):592-610. doi:10.1177/09612033261432704
- The American College of Rheumatology nomenclature and case definitions for neuropsychiatric lupus syndromes. ACR Ad Hoc Committee on Neuropsychiatric Lupus. Arthritis & Rheumatism. 1999;42(4):599-608. doi:10.1002/1529-0131(199904)42:4<599::AID-ANR2>3.0.CO;2-F
- The diagnosis and clinical management of the neuropsychiatric manifestations of lupus. Govoni M, Bortoluzzi A, Padovan M et al. Journal of Autoimmunity. 2016;74:41-72. doi:10.1016/j.jaut.2016.06.013
- Low C4 as a risk factor for severe neuropsychiatric flare in patients with systemic lupus erythematosus. Aso K, Kono M, Kono M et al. Lupus. 2020;29(10):1238-1247. doi:10.1177/0961203320938453
- Neuropsychiatric lupus: new mechanistic insights and future treatment directions. Schwartz N, Stock AD, Putterman C. Nature Reviews Rheumatology. 2019;15(3):137-152. doi:10.1038/s41584-018-0156-8
- Acute psychosis in systemic lupus erythematosus. Appenzeller S, Cendes F, Costallat LT. Rheumatology International. 2008;28(3):237-243. doi:10.1007/s00296-007-0410-x
- Development and validation of a new algorithm for attribution of neuropsychiatric events in systemic lupus erythematosus. Bortoluzzi A, Scirè CA, Bombardieri S et al. Rheumatology. 2015;54(5):891-898. doi:10.1093/rheumatology/keu384
- Prospective study of neuropsychiatric events in systemic lupus erythematosus. Hanly JG, Su L, Farewell V et al. Journal of Rheumatology. 2009;36(7):1449-1459. doi:10.3899/jrheum.081133
- Psychosis in systemic lupus erythematosus: results from an international inception cohort study. Hanly JG, Li Q, Su L et al. Arthritis & Rheumatology. 2019;71(2):281-289. doi:10.1002/art.40764
