Research Highlights
- GLP-1 receptor agonists are diabetes drugs (Ozempic, Trulicity, Byetta) being tested in Parkinson’s disease for possible disease-modifying effects. The hypothesis: shared metabolic-inflammatory pathways between type 2 diabetes and Parkinson’s mean drugs that work for one might help the other.
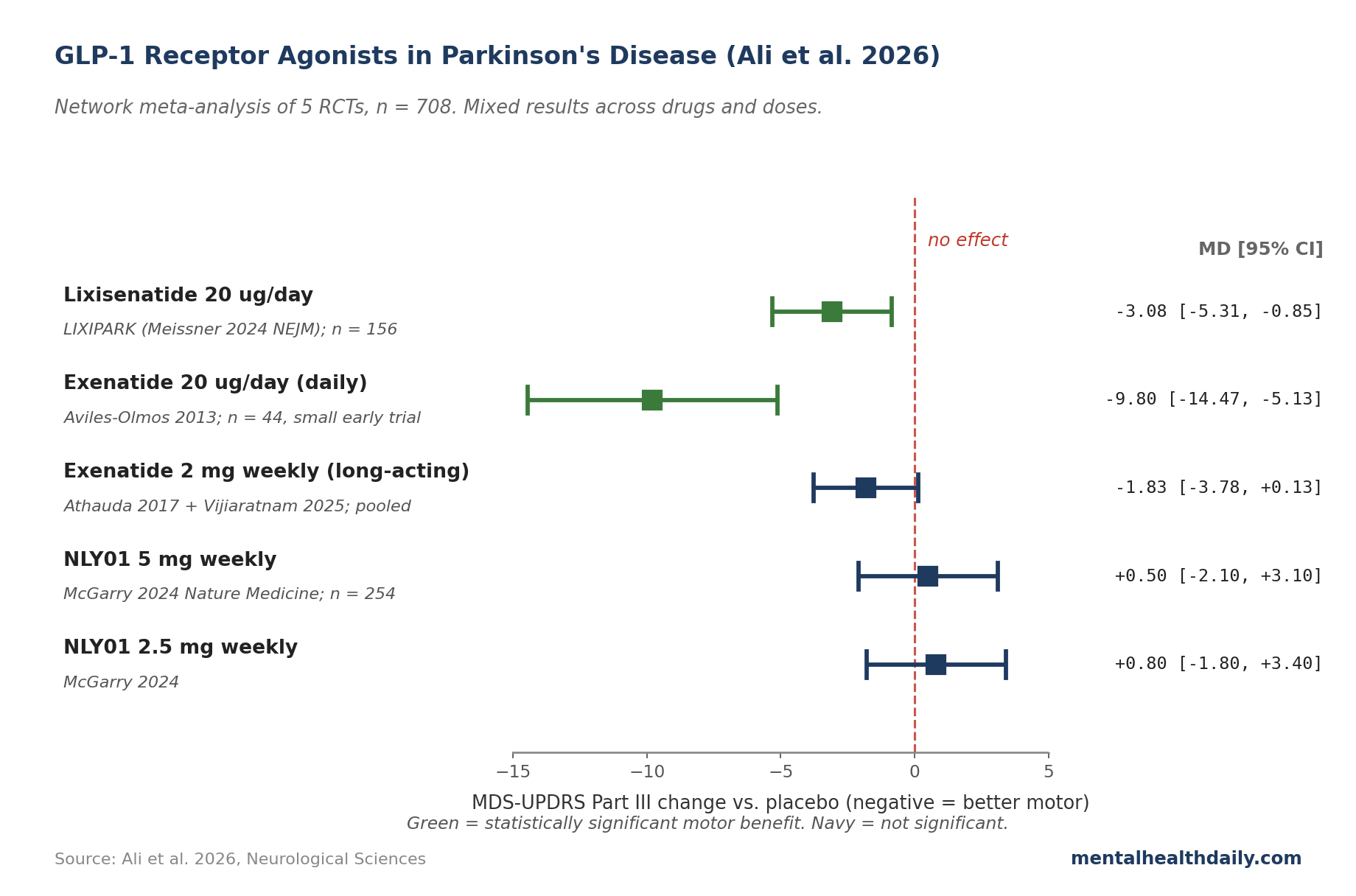
- A 2026 network meta-analysis pooled 5 trials and 708 patients. Pairwise analysis showed no overall motor benefit (MD −2.00 points on the standard movement scale, confidence interval crosses zero). Drilling down by specific drug and dose, exenatide 20 µg/day and lixisenatide 20 µg/day did show significant motor improvement.
- The clearest trial signal so far is from LIXIPARK (Meissner 2024). Lixisenatide reduced motor decline over 12 months compared to placebo in 156 early-stage PD patients. The effect was modest (about 3 points on the motor scale) but statistically significant, and it was published in the New England Journal of Medicine, which carries weight.
- Quality of life and non-motor symptoms generally didn’t improve. The Parkinson’s Disease Questionnaire-39 and Non-Motor Symptoms Scale showed no consistent benefit. Whether GLP-1 drugs help with depression, sleep, or cognition in PD — symptoms that drive much of the disease burden — isn’t yet shown.
- Preliminary signal, not established disease-modifying treatment. The most-discussed earlier exenatide trial (Vijiaratnam 2025) was negative on its primary outcome over 96 weeks. The picture is mixed enough that GLP-1 drugs aren’t ready for routine PD prescribing, but the lixisenatide signal holds up across analyses and is worth tracking.
Parkinson’s disease is the second most common neurodegenerative disorder, affecting around 10 million people worldwide. The mental-health burden is heavy: depression affects 40-50% of patients9, anxiety 30-40%, sleep disorders most patients eventually, and dementia about a third of long-duration cases.
Standard treatment with levodopa controls motor symptoms but doesn’t slow the underlying neurodegeneration. The clinical field has been searching for a disease-modifying drug for decades. The 2026 Ali network meta-analysis pulls together the current GLP-1 trial data — one of the most promising candidate paths.1
Why GLP-1 Drugs Might Help Parkinson’s Disease
GLP-1 (glucagon-like peptide-1) receptor agonists were developed for type 2 diabetes. They mimic an intestinal hormone that triggers insulin release after meals, which lowers blood sugar.
Common agents include exenatide (Byetta, Bydureon), liraglutide (Victoza), semaglutide (Ozempic, Wegovy), and lixisenatide (Adlyxin).
Type 2 diabetes and Parkinson’s share several biological features (insulin resistance in dopaminergic neurons being the most direct overlap, summarized in Athauda and Foltynie’s 2016 review)8 that suggested overlap might be exploitable:
- Insulin resistance in the brain. Both diseases show signs of disrupted insulin signaling in neurons, which interferes with energy production and protein clearance. GLP-1 receptors are expressed in many of the brain regions affected by Parkinson’s, including the substantia nigra (where dopamine neurons die in PD).
- Mitochondrial dysfunction. Both diseases show damaged mitochondria (the cell’s energy factories). GLP-1 drugs improve mitochondrial function in lab models, which raised hopes they might do the same in dying dopamine neurons.
- Neuroinflammation. Chronic low-grade inflammation in the brain is implicated in PD progression. GLP-1 receptor activation reduces inflammatory signaling in animal studies.
The biological rationale was strong enough that several research groups launched clinical trials starting around 2013. The hope was that GLP-1 drugs — already proven safe at scale in diabetes — might slow PD progression rather than just ease symptoms.
2026 Meta-Analysis: GLP-1 Receptor Agonists vs. Parkinson’s Disease
Ali and colleagues’ 2026 systematic review and network meta-analysis in Neurological Sciences identified 5 randomized controlled trials of GLP-1 receptor agonists in PD, totaling 708 patients across all arms.1 The five trials, in chronological order:
- Aviles-Olmos 2013 — first proof-of-concept trial of exenatide in PD (44 patients, 12 months).5
- Athauda 2017 — Lancet trial of exenatide once-weekly, 60 patients, 48 weeks.4
- McGarry 2024 — NLY01 trial (a longer-acting GLP-1 formulation), 254 patients, 36 weeks. Negative on primary outcome.6
- Meissner 2024 — LIXIPARK trial of lixisenatide, 156 patients, 12 months, NEJM. Positive on motor outcome.
- Vijiaratnam 2025 — longer exenatide trial, 194 patients, 96 weeks. Negative on primary outcome.
The primary outcome across these trials is the MDS-UPDRS Part III — the Movement Disorder Society Unified Parkinson’s Disease Rating Scale, motor examination subsection. It’s a 0-132 point scale assessing tremor, rigidity, slowness, and gait. Higher scores mean worse motor symptoms; about a 3-point change is considered minimally clinically meaningful.
The pairwise meta-analysis (combining all 5 trials and all GLP-1 drugs into one pooled estimate) returned a non-significant overall effect: mean difference of −2.00 points (95% CI −5.46 to 1.46), with high heterogeneity (I² = 80.5%, meaning the trials disagree substantially about the magnitude of any benefit).
Pooling drugs that worked with drugs that didn’t washes out the signal.
The network meta-analysis (which estimates effects for individual drug-dose combinations using both direct and indirect comparisons) produced more informative results:
- Exenatide 20 µg/day: mean difference −9.80 (95% CI −14.47 to −5.13). A large motor improvement, but this dose comes from the 12-month Aviles-Olmos trial of daily-dose exenatide, which was a small early trial. Once-weekly long-acting exenatide formulations didn’t replicate the effect.
- Lixisenatide 20 µg/day: mean difference −3.08 (95% CI −5.31 to −0.85). A modest but statistically significant motor improvement — this is the LIXIPARK trial result and is the strongest current evidence for a GLP-1 drug in PD.
- Other agents (NLY01, weekly exenatide): no significant motor benefit.
The LIXIPARK Trial Findings
The LIXIPARK trial (Meissner 2024, New England Journal of Medicine) is the most rigorous current evidence for a GLP-1 drug in PD.2 156 patients with early-stage PD were randomized to lixisenatide (titrated up to 20 µg/day) or placebo for 12 months while continuing their standard PD medications. Headline findings:
Motor score change at 12 months (MDS-UPDRS Part III, ON-state, after morning levodopa): the placebo group worsened by 3.0 points (typical PD progression). The lixisenatide group worsened by 0.0 points — essentially no progression over the year. The between-group difference of approximately 3 points was statistically significant and clinically meaningful for early-stage PD.
The trial is the cleanest evidence to date that a GLP-1 drug might slow PD motor progression. Important caveats:
- Single 12-month timepoint. Whether the gap widens or narrows over years isn’t known. Disease-modification claims really need multi-year data, and LIXIPARK doesn’t have those yet.
- Modest absolute magnitude. Three points on a 132-point scale is detectable but not transformative for an individual patient. The clinical impact accumulates only if the effect persists.
- Gastrointestinal adverse events were common. Nausea, vomiting, and weight loss occurred more often on lixisenatide. Some patients couldn’t tolerate the maintenance dose. Real-world adherence may be worse than trial adherence.
How the Earlier Exenatide Got Complicated
The path to LIXIPARK ran through a high-profile trial that didn’t work.
Vijiaratnam and colleagues’ 2025 trial extended Athauda’s 2017 exenatide protocol to 96 weeks (almost 2 years) in 194 patients, and the primary motor endpoint did not reach significance.3
The Athauda 2017 result was widely covered in 2017 as a possible breakthrough — a meaningful motor effect that persisted 12 weeks after stopping the drug, raising the prospect of disease modification.
The longer Vijiaratnam follow-up was supposed to confirm and extend that finding.
When it didn’t, the field had to recalibrate.
Several explanations have been offered:
- The Athauda 2017 effect may have been noise from a small trial; subsequent better-powered work didn’t replicate.
- The longer-acting weekly exenatide formulation in Vijiaratnam may not deliver the same brain exposure as the daily-dose exenatide in the earlier proof-of-concept trial.
- The patient population in Vijiaratnam may have been further along in disease progression, where neuroprotection is harder to demonstrate.
- The treatment effect, if present, may be too small to detect even at 96 weeks with this sample size.
The truth is probably some combination.
What we can say cleanly: the strong “exenatide is disease-modifying” claims that appeared in lay coverage in 2017 didn’t survive longer trials. The lixisenatide signal in 2024 is modest enough that even a careful read can’t yet confirm it as disease modification.
What the Trial Doesn’t Measure
Most patients with PD — and most readers researching PD treatments — care about more than the motor exam score. PD’s burden is partly motor, partly cognitive, partly emotional, and partly autonomic.
Across the GLP-1 trial set, the non-motor outcomes have been mostly null:
- Quality of life (PDQ-39). The pooled mean difference across all 5 trials was −0.33 (95% CI −1.50 to 0.85), with no heterogeneity. Whatever motor improvement happens, patients aren’t reporting consistent quality-of-life improvements yet.
- Non-motor symptoms (NMSS). The Non-Motor Symptoms Scale captures cognition, mood, sleep, autonomic function, urinary, sexual, and gastrointestinal symptoms. NLY01 5 mg showed some signal at this measure; other GLP-1 agents did not.
- Activities of daily living (Part II) and motor complications (Part IV). No significant differences across the meta-analysis.
The mental-health-relevant point: GLP-1 drugs aren’t yet showing clear effects on the depression, anxiety, sleep, or cognitive symptoms that drive much of PD’s disability. If GLP-1 drugs eventually become standard PD treatment, the case will rest on motor benefit and possible long-term disease modification — not on improved daily-life experience or mental-health symptoms.
Gaps in the GLP-1-in-PD Literature
Several limitations cut across the trial set.
Sample sizes are small. The largest trial enrolled 254 patients; most enrolled fewer than 100 per arm. Pooled across the 5 trials, total n is 708 — modest by standards of established disease-modifying treatments in other neurological conditions.
Follow-up is short. Two trials ran 12 months; one ran 36 weeks; one ran 48 weeks; the longest ran 96 weeks. Disease modification is a multi-year question, and the trial set doesn’t yet have the duration to answer it.
Drug heterogeneity makes pooling problematic. Mulvaney and colleagues’ 2020 Cochrane review of GLP-1 drugs in PD reached similar mixed conclusions across the older trial set.7 Different GLP-1 agents have different brain penetration, different receptor subtype affinity, and different dosing. The pairwise pooled effect averages across drugs that may not have the same biological profile in the brain.
Patient selection skews early-stage. Most trials enrolled patients with mild to moderate PD (Hoehn-Yahr stage ≤2.5). Whether GLP-1 drugs help patients with more advanced disease is unknown.
The disease-modification claim isn’t formally tested. Showing slower motor decline over 12 months is consistent with disease modification but also consistent with symptomatic effects that mask underlying progression. The cleanest test would require a randomized withdrawal or delayed-start design over multiple years — which hasn’t been done.
Where GLP-1 Drugs Stand for PD Treatment
The honest read on the current evidence:
- GLP-1 drugs are not currently first-line treatment for PD. Standard PD pharmacology — levodopa, dopamine agonists, MAO-B inhibitors — remains the foundation. GLP-1 drugs aren’t FDA-approved for PD and aren’t recommended in current PD treatment guidelines.
- Lixisenatide has the strongest current evidence and is being investigated further. If you have early-stage PD and are interested in GLP-1 therapy, the appropriate path is enrollment in an ongoing clinical trial rather than off-label prescription. Several extension and replication trials are underway.
- Off-label use of GLP-1 drugs (Ozempic, etc.) for PD is not supported by current evidence. The trials that worked tested specific agents at specific doses. Newer GLP-1 drugs marketed for diabetes and weight loss have not been tested in PD trials. The gastrointestinal adverse-effect profile is consistent across the class; the brain effects may not be.
- For PD patients who also have type 2 diabetes, this calculus shifts. A GLP-1 drug prescribed for diabetes that may also have small motor-modifying effects in PD is a different cost-benefit conversation than off-label use without a primary indication. Discuss with your neurologist and endocrinologist.
- Watch for the next generation of trials. The field is moving fast. Replication of the LIXIPARK signal, longer-duration follow-ups, and tests of brain-penetrant GLP-1 agents specifically designed for PD are in the pipeline. The story is far from settled.
Common Questions: GLP-1 Drugs in Parkinson’s
Should I ask my doctor about taking Ozempic for Parkinson’s?
Not yet. The trials that show benefit used specific agents (exenatide, lixisenatide) at specific doses. Semaglutide (Ozempic) has not been tested in PD trials. Off-label use without trial evidence is not currently supported, and GI side effects may not be worth tolerating without a primary indication.
Why did some trials work and others didn’t?
Probably some combination of drug differences (different agents have different brain penetration), dosing (daily vs. weekly formulations differ), trial duration, and statistical noise from small samples. The earlier exenatide signal (Athauda 2017) didn’t replicate in a longer trial (Vijiaratnam 2025); the lixisenatide signal (Meissner 2024) is newer and hasn’t been independently replicated yet.
Do GLP-1 drugs help with the depression or anxiety in PD?
Not on the available evidence. The non-motor symptoms scale and quality-of-life measures across the 5 trials were mostly null. Standard treatments for depression and anxiety in PD — SSRIs, CBT, and where appropriate, DBS — remain the recommended approaches.
Are GLP-1 drugs neuroprotective?
Animal and cell models show neuroprotective effects. Whether this translates to humans is the open question. The lixisenatide trial showed reduced motor decline over 12 months, which is consistent with neuroprotection but also consistent with symptomatic effects. Multi-year trials with delayed-start designs would settle this; they haven’t been done yet.
What are the side effects?
Nausea, vomiting, decreased appetite, and weight loss are the most common. These are usually worst during the dose-titration phase and improve over time. Pancreatitis is a rare but serious risk. The PD trials report side-effect profiles consistent with diabetes use.
If LIXIPARK was positive, why isn’t lixisenatide approved for PD?
A single positive 12-month trial isn’t enough for FDA approval. Regulators typically require replication, longer-duration data, and clear demonstration of disease modification rather than just symptomatic effects. The lixisenatide story is at “promising signal needing replication” rather than “approval-ready.”
References
- Efficacy of GLP-1 receptor agonists in Parkinson’s disease: a systematic review and exploratory network meta-analysis of randomized controlled trials. Ali M et al. Neurological Sciences. 2026;47:432. doi:10.1007/s10072-026-08929-1
- Trial of lixisenatide in early Parkinson’s disease. Meissner WG et al. New England Journal of Medicine. 2024;390(13):1176-1185. doi:10.1056/NEJMoa2312323
- Exenatide once weekly versus placebo as a potential disease-modifying treatment for Parkinson’s disease in the UK: a multicentre, randomised, double-blind, placebo-controlled phase 3 trial. Vijiaratnam N et al. The Lancet. 2025;405(10434):1330-1340. doi:10.1016/S0140-6736(25)00404-7
- Exenatide once weekly versus placebo in Parkinson’s disease: a randomised, double-blind, placebo-controlled trial. Athauda D et al. The Lancet. 2017;390(10103):1664-1675. doi:10.1016/S0140-6736(17)31585-4
- Exenatide and the treatment of patients with Parkinson’s disease. Aviles-Olmos I et al. Journal of Clinical Investigation. 2013;123(6):2730-2736. doi:10.1172/JCI68295
- A randomised trial of NLY01 in early Parkinson disease. McGarry A et al. Nature Medicine. 2024;30(12):3576-3583. doi:10.1038/s41591-024-03281-3
- Glucagon-like peptide-1 receptor agonists for the treatment of Parkinson’s disease: a meta-analysis of randomized controlled trials. Mulvaney CA et al. Cochrane Database of Systematic Reviews. 2020. doi:10.1002/14651858.CD012990.pub2
- Insulin resistance and Parkinson’s disease: a new target for disease modification? Athauda D, Foltynie T. Progress in Neurobiology. 2016;145-146:98-120. doi:10.1016/j.pneurobio.2016.10.001
- Depression and Parkinson disease: a systematic review and meta-analysis. Reijnders JS et al. Movement Disorders. 2008;23(2):183-189. doi:10.1002/mds.21803
