Lisdexamfetamine remains the only FDA-approved medication for binge-eating disorder — a serious condition affecting roughly 1.2% of adults. A 2026 human laboratory study by McKee and colleagues tests whether naltrexone/bupropion, already approved for obesity, reduces the food-reward mechanisms that drive binge episodes.1
Research Highlights
- Binge-eating disorder (BED) is the most common eating disorder in the United States, affecting roughly 1.2% of adults and producing serious medical, psychiatric, and quality-of-life consequences. Treatment options remain limited.2
- Naltrexone/bupropion (NB) is FDA-approved for obesity. The combination targets the brain’s reward and hypothalamic appetite circuits: bupropion stimulates POMC neurons; naltrexone blocks opioid-receptor-mediated POMC autoinhibition.3
- The McKee 2026 study ran a human laboratory paradigm in 50 adults with BED (72% with obesity) embedded within a 12-week randomized clinical trial of NB.1
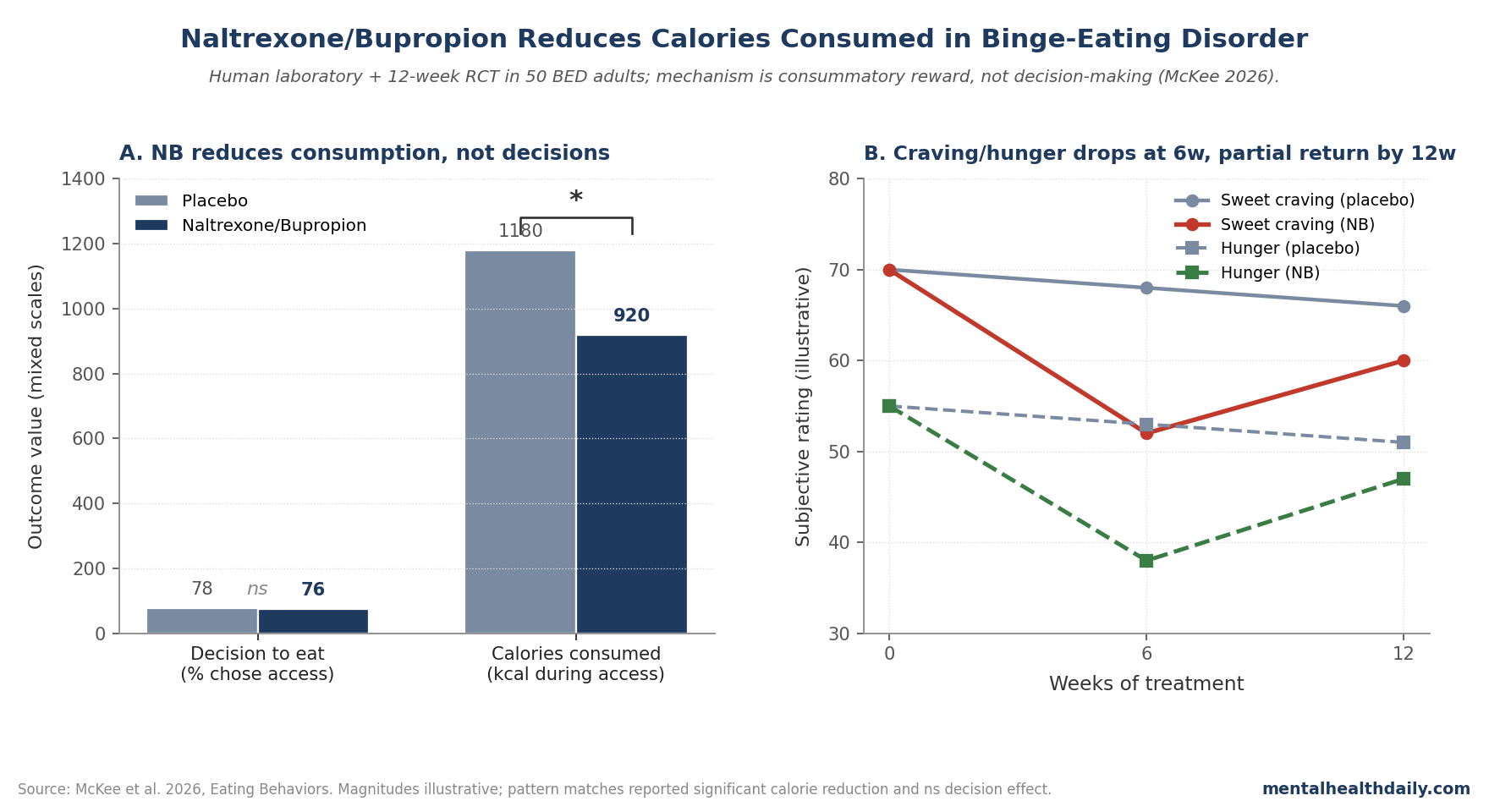
- NB significantly reduced calories consumed when participants were given access to preferred high-caloric snack food, with reductions correlating with plasma levels of 6β-naltrexol and bupropion. Craving for sweet snacks and hunger ratings were also reduced at 6 weeks.1
- NB did NOT change initial decisions to eat — it changed how much was consumed once eating started. The mechanistic implication: NB acts on consummatory food reward, not on preceding decision-making.1
BED was officially recognized in DSM-5 (2013), and lisdexamfetamine was approved for it in 2015 — the first and so far only medication. Lisdexamfetamine has important contraindications (history of substance misuse) and a labeling caveat that it isn’t recommended for weight reduction. Patients with BED and obesity often need a treatment that addresses both, with a more permissive risk profile.4
Naltrexone (an opioid antagonist) and bupropion (a norepinephrine-dopamine reuptake inhibitor) both target food-reward circuits. The combination was developed for obesity based on synergistic effects on hypothalamic POMC neurons and brain reward systems. Whether it would also reduce binge-eating-specific behaviors has been a logical next question.3,5
McKee 2026: Human Laboratory + Randomized Clinical Trial Design
The trigger paper enrolled 50 adults with DSM-5 BED, 72% with comorbid obesity. Participants completed a human laboratory paradigm at baseline, 6 weeks, and 12 weeks during a 12-week randomized trial of NB vs placebo.1
Three primary outcomes were measured at each lab visit:
- Decision to eat: ability to resist their preferred high-caloric snack food during a structured choice paradigm.
- Calories consumed: total energy intake during a free-access eating period.
- Food craving: self-reported craving for preferred snacks before and after the access period.
Secondary outcomes: hunger ratings, mood, and trough plasma levels of NB metabolites (6β-naltrexol and bupropion).
The Headline: Calories Down, Decisions Unchanged
NB significantly reduced calories consumed compared to placebo. The reduction was dose-correlated — participants with higher trough levels of 6β-naltrexol and bupropion showed larger reductions in calories consumed, supporting a pharmacologically driven mechanism rather than a generic placebo effect.1
Critically, NB did not change participants’ initial decisions to eat. When given the choice to access high-caloric snack food, NB and placebo participants made similar decisions. Once they started eating, however, NB participants ate substantially less.
This dissociation is mechanistically informative. The decision-to-eat phase reflects appetitive (anticipatory) reward processing — whether the food is judged worth pursuing. The amount-consumed phase reflects consummatory reward processing — how reinforcing the food is once eating has begun. NB selectively shifted the consummatory phase.1,6
Craving and Hunger: Sweet-Specific Effects at 6 Weeks
NB reduced craving for preferred high-caloric sweet snacks but not for other food types. The effect was significant at 6 weeks but did not persist to 12 weeks — a partial-efficacy time course consistent with adaptation in the underlying mechanism.1
Hunger ratings showed the same pattern: reduced at 6 weeks, returning toward baseline by 12 weeks. The selective effect on sweet-food craving fits the pharmacology — mu-opioid receptors in the nucleus accumbens are particularly involved in reinforcement of palatable sweet foods, and naltrexone blocks this reinforcement.7
Mechanism: Reward Pathway, Not Decision Pathway
The McKee dissociation between “decisions to eat” and “calories consumed” maps onto distinct neural pathways:
Decision-making (intact under NB) recruits dorsolateral prefrontal cortex, anterior cingulate, and value-computation regions like ventromedial prefrontal cortex. These weigh costs, benefits, dietary intentions, and future-self concerns. NB doesn’t change this calculus.8
Consumption (reduced under NB) recruits striatal reward circuitry — nucleus accumbens, ventral pallidum, and the mu-opioid-receptor-rich hedonic hotspots that drive “liking” once palatable food is in the mouth. Naltrexone’s opioid blockade reduces this hedonic reinforcement; bupropion’s catecholamine effects further dampen the dopaminergic “wanting” signal.7,9
The clinical translation: NB doesn’t prevent patients from initiating an eating episode, but it makes the episode less reinforcing once underway, reducing total caloric intake and breaking the binge cycle.
Why This Matters for BED Treatment
Several practical implications:
First, the mechanism dissociation suggests NB pairs well with cognitive-behavioral therapy. CBT for BED targets decision-making, urge regulation, and triggering contexts — the front-end of the binge cycle. NB acts on the consummatory back-end. Combining them addresses two stages of the binge sequence rather than redundantly targeting one.10
Second, the dose-response correlation between drug levels and calorie reduction supports tolerable-dose maintenance and patient-level dose individualization. Patients with sub-therapeutic levels may benefit from dose increases.1
Third, the time-course nuance — sweet-craving and hunger reductions at 6 weeks but not 12 weeks — suggests that treatment monitoring should track whether early gains are maintained, with consideration of dose adjustment, behavioral augmentation, or alternative therapy if effects attenuate.
Limitations of the McKee Analysis
Three caveats deserve weight:
- Modest sample (n = 50). The human laboratory paradigm with 50 BED participants is statistically capable of detecting medium-to-large effects but underpowered for subgroup or moderator analyses. Replication and scaling are warranted.
- 12-week duration. Long-term effects on binge frequency, weight, and remission rates require extended follow-up. The accompanying RCT addresses this in part; the human laboratory component captures only the within-trial mechanism signal.
- 72% with comorbid obesity. Whether the mechanism findings extend to BED patients without obesity (the remaining 28% of BED population) needs separate testing — effects on the reward/consumption circuit may differ when baseline weight regulation is intact.
The attenuation of craving and hunger effects from 6 to 12 weeks is also interpretively complex — it could reflect tolerance, regression to the mean, or differential dropout patterns.1
How This Fits the Broader BED Pharmacotherapy Picture
BED pharmacotherapy has lagged behind eating-disorder psychotherapy in development. Lisdexamfetamine (the only FDA-approved BED medication) targets dopaminergic reward but with stimulant-class side effects and substance-misuse contraindications. SSRIs have shown modest benefit. Topiramate has been used off-label.11
The McKee mechanism work adds NB as a candidate option, particularly for BED with comorbid obesity where weight reduction is also a treatment goal. Direct head-to-head comparisons against lisdexamfetamine haven’t been done. The complementary mechanism — consummatory reward modulation rather than appetitive activation — suggests NB may pair differently with concurrent CBT than stimulant-class options.1,10
What Popular Coverage Often Misses
Two framings deserve calibration. First, NB isn’t FDA-approved for BED — only for obesity. The McKee work supports off-label consideration in BED+obesity patients, but doesn’t change regulatory status or guarantee insurance coverage for the indication.1
Second, “medication for binge eating” framings often imply elimination of binge episodes. NB’s mechanism — reducing calories consumed once eating starts, not preventing eating — means treatment success looks more like “smaller, less destructive episodes” than “binge-free.” Setting realistic expectations matters for treatment adherence.
Practical Implications
For patients with BED and comorbid obesity considering pharmacological options, NB joins a small list of mechanism-supported candidates. Decisions should be made with a clinician familiar with both the obesity and eating-disorder literatures, with attention to cardiac risk profile (bupropion seizure threshold considerations), substance-use history (bupropion is also bupropion), and concurrent psychiatric conditions.3,4
For research, the human-laboratory-plus-RCT design template that McKee used is worth highlighting. Embedding mechanism-targeted laboratory paradigms within efficacy trials produces evidence that’s simultaneously clinically grounded and mechanistically informative — rare in eating-disorder pharmacotherapy and worth scaling.1
Common Questions About Naltrexone/Bupropion for Binge Eating
What is binge-eating disorder?
A DSM-5 eating disorder defined by recurrent episodes of consuming unusually large amounts of food with a sense of loss of control, occurring at least once per week for three months, accompanied by marked distress — without the compensatory behaviors (purging, restriction) that define bulimia nervosa.2
What is naltrexone/bupropion?
An FDA-approved combination medication for chronic weight management (brand name Contrave). Naltrexone is an opioid receptor antagonist; bupropion is a norepinephrine-dopamine reuptake inhibitor also used for depression and smoking cessation.3
Is NB approved for BED?
No. It’s FDA-approved for chronic weight management in adults with obesity. The McKee study supports off-label consideration in BED with comorbid obesity but doesn’t establish a new approved indication.1
How does NB reduce binge eating?
By dampening the consummatory food-reward signal once eating has begun — participants ate less of preferred snack foods despite making the same decision to access them. Naltrexone blocks opioid-receptor reinforcement of palatable foods; bupropion dampens dopaminergic “wanting.”1,7
What about lisdexamfetamine?
The only FDA-approved BED medication. It has substance-misuse contraindications and isn’t recommended for weight reduction. NB and lisdexamfetamine target different parts of the reward system and may have different patient-fit profiles.4
Does NB work without therapy?
The McKee data show pharmacological mechanism effects independent of psychotherapy, but combination treatment (NB plus CBT) is generally expected to outperform either alone. CBT targets the decision-making side, NB the consumption side — a complementary pairing.10
What are the side effects?
Common: nausea, headache, constipation, dizziness, insomnia. Bupropion lowers seizure threshold (contraindicated in patients with seizure disorders or active eating disorders with significant restriction/purging). Naltrexone is contraindicated with opioid use.3
What’s the next study that needs to happen?
Direct head-to-head trials of NB vs lisdexamfetamine in BED, longer-duration follow-up to test maintenance of effects, and combination protocols pairing NB with CBT to test whether the mechanism-complementary hypothesis pays off in clinical outcomes.
References
- McKee SA, Lydecker JA, Pittman B, et al. Naltrexone/bupropion for binge-eating disorder: a human laboratory investigation of mechanisms. Eating Behaviors. 2026. doi:10.1016/j.dadr.2026.100428
- Hudson JI, Hiripi E, Pope HG, Kessler RC. The prevalence and correlates of eating disorders in the National Comorbidity Survey Replication. Biological Psychiatry. 2007;61(3):348–358. doi:10.1016/j.biopsych.2006.03.040
- Yanovski SZ, Yanovski JA. Naltrexone extended-release plus bupropion extended-release for treatment of obesity. JAMA. 2015;313(12):1213–1214. doi:10.1001/jama.2015.1617
- McElroy SL. Pharmacologic treatments for binge-eating disorder. Journal of Clinical Psychiatry. 2017;78 Suppl 1:14–19. doi:10.4088/jcp.sh16003su1c.03
- Greenway FL, Fujioka K, Plodkowski RA, et al. Effect of naltrexone plus bupropion on weight loss in overweight and obese adults (COR-I): a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2010;376(9741):595–605. doi:10.1016/s0140-6736(10)60888-4
- Berridge KC. Food reward: brain substrates of wanting and liking. Neuroscience & Biobehavioral Reviews. 1996;20(1):1–25. doi:10.1016/0149-7634(95)00033-b
- Pecina S, Berridge KC. Hedonic hot spot in nucleus accumbens shell: where do mu-opioids cause increased hedonic impact of sweetness? Journal of Neuroscience. 2005;25(50):11777–11786. doi:10.1523/jneurosci.2329-05.2005
- Volkow ND, Wang GJ, Tomasi D, Baler RD. The addictive dimensionality of obesity. Biological Psychiatry. 2013;73(9):811–818. doi:10.1016/j.biopsych.2012.12.020
- Billes SK, Sinnayah P, Cowley MA. Naltrexone/bupropion for obesity: an investigational combination pharmacotherapy for weight loss. Pharmacological Research. 2014;84:1–11. doi:10.1016/j.phrs.2014.04.004
- Hilbert A, Petroff D, Herpertz S, et al. Meta-analysis of the efficacy of psychological and medical treatments for binge-eating disorder. Journal of Consulting and Clinical Psychology. 2019;87(1):91–105. doi:10.1037/ccp0000358
- Reas DL, Grilo CM. Pharmacological treatment of binge eating disorder: update review and synthesis. Expert Opinion on Pharmacotherapy. 2015;16(10):1463–1478. doi:10.1517/14656566.2015.1053465