The risperidone-stroke link in dementia is one of the longest-running safety signals in geriatric psychiatry, anchoring the FDA’s 2005 black-box warning on atypical antipsychotic mortality. Popular framing reduces it to “antipsychotics cause stroke in dementia” — a true sentence that flattens a multi-axis decision. Choma’s 2025 analysis of UK primary care records is the largest published evaluation of how that risk distributes by cardiovascular history, and the answer rebalances the discussion: relative risk is similar across subgroups, but absolute risk depends almost entirely on baseline.1
Research Highlights
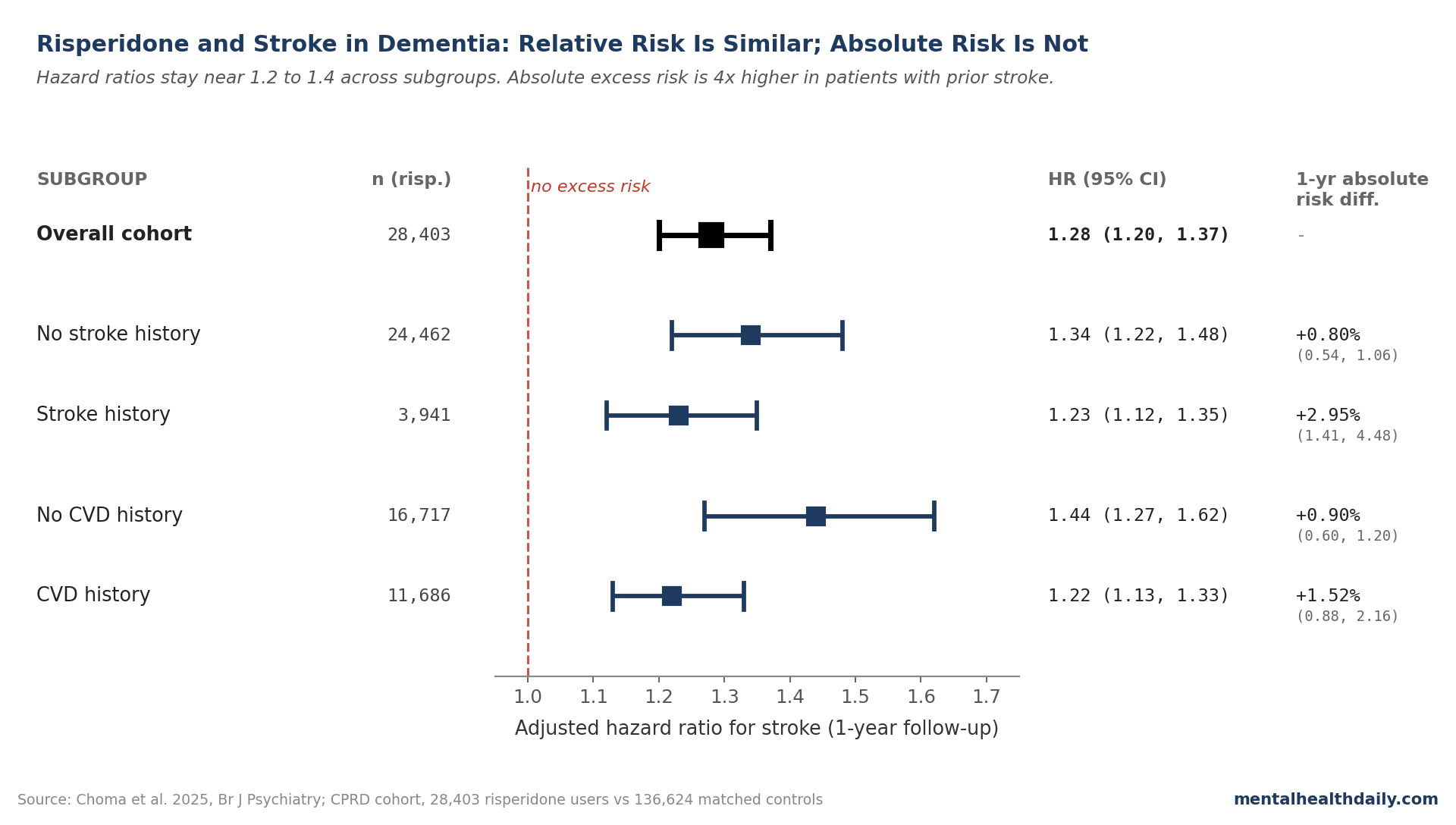
- Risperidone increases stroke risk ~28% overall in dementia. In a UK CPRD cohort of 28,403 risperidone users vs. 136,624 matched controls, the 1-year adjusted hazard ratio (HR) was 1.28 (95% CI 1.20–1.37).1
- Relative risk is similar across CVD and stroke history. HRs sit between 1.22 and 1.44 across all four subgroups. The hypothesis that prior CVD would amplify the relative effect was not supported.1
- Absolute risk differs by ~4x. 1-year absolute excess stroke risk is 0.80% in patients with no prior stroke vs. 2.95% in those with prior stroke; 0.90% no-CVD vs. 1.52% with CVD.1
- Risk concentrates in the first weeks. 12-week HRs (1.33 to 1.69) exceed 1-year HRs across every subgroup, consistent with prior literature on antipsychotic stroke timing.1,2
- “Antipsychotics cause stroke in dementia” flattens the decision. The comparator is rarely “no harm” — severe agitation and psychosis carry their own risks. Choma’s data turn the question into a quantifiable conversation about absolute risk in a specific patient.
Choma 2025: HR 1.28 Overall, 1.22 to 1.44 Across Subgroups
Choma and colleagues used the Clinical Practice Research Datalink (CPRD) Aurum, a large UK primary-care database linked to hospital and death records, to identify adults diagnosed with dementia after age 65 between 2004 and 2023. They compared 1-year stroke risk in 28,403 new risperidone users and 136,624 propensity-score-matched controls not on any antipsychotic at the index date.1
(A propensity score summarizes how likely a patient is to receive a treatment based on measured baseline traits. Matching on it mimics randomization on those measured variables — though it can’t account for unmeasured ones.)
The headline finding in the overall cohort:
- Adjusted HR for stroke at 1 year: 1.28 (95% CI 1.20–1.37).1
- Stroke incidence: 53.2 per 1,000 person-years on risperidone vs. 40.6 per 1,000 person-years in matched controls.1
The four prespecified subgroup HRs barely move:
- No stroke history: HR 1.34 (95% CI 1.22–1.48).1
- Prior stroke: HR 1.23 (95% CI 1.12–1.35).1
- No CVD history: HR 1.44 (95% CI 1.27–1.62).1
- CVD history: HR 1.22 (95% CI 1.13–1.33).1
The authors had hypothesized prior CVD would amplify the relative effect — consistent with preclinical work suggesting risperidone interacts directly with cardiovascular biology3 and an earlier secondary RCT analysis suggesting subgroup heterogeneity in cerebrovascular events.11 The data didn’t support that. Point estimates lean slightly the other way, with the largest relative risk in the no-CVD subgroup. CIs overlap; the formal between-subgroup interaction is not statistically distinguishable.1
Why Absolute Risk Tells a Different Story Than Relative Risk
The relative-risk uniformity across subgroups is what makes the absolute-risk variation matter. A 28% increase on a low baseline is small in absolute terms; a 23% increase on a high baseline can be a meaningful jump in stroke probability over a few months.
The 1-year absolute risk differences from Choma’s Kaplan-Meier curves:1
- No stroke history: +0.80% (95% CI 0.54%–1.06%) excess 1-year risk.
- Prior stroke: +2.95% (95% CI 1.41%–4.48%) — almost 4 times larger.
- No CVD history: +0.90% (95% CI 0.60%–1.20%).
- CVD history: +1.52% (95% CI 0.88%–2.16%).
Put differently: in dementia patients with prior stroke, baseline 1-year stroke incidence is 177 per 1,000 person-years; on risperidone it’s 222.1 Roughly 80% of the strokes seen in that subgroup would have happened anyway; the drug adds about 30 per 1,000 person-years on top.
For a patient with no prior cerebrovascular disease, baseline is 19 per 1,000 person-years and risperidone raises it to 27. Most strokes in that subgroup are caused by the drug rather than by underlying disease — but the underlying disease load is small enough that absolute excess is a third of the prior-stroke group.
Where this lands clinically: the same 1.28 hazard ratio means very different things for a patient with prior stroke vs. one without. Risk communication has to be in absolute terms — “about a 1-in-30 chance over the next year on top of an already 1-in-5 baseline” is a different conversation than a 1-in-110 attributable risk in a patient with cleaner cerebrovascular history.
Stroke Risk Concentrates in the First 12 Weeks
Antipsychotic-associated stroke risk peaks early. Choma’s restricted 12-week analysis showed higher relative risks across every subgroup than the 1-year estimates:1
- No stroke history: 12-week HR 1.61 (95% CI 1.37–1.90), vs. 1-year HR 1.34.
- Prior stroke: 12-week HR 1.33 (95% CI 1.16–1.52), vs. 1-year HR 1.23.
- No CVD history: 12-week HR 1.69 (95% CI 1.37–2.08), vs. 1-year HR 1.44.
- CVD history: 12-week HR 1.37 (95% CI 1.21–1.54), vs. 1-year HR 1.22.
The pattern matches earlier literature. Douglas and Smeeth’s BMJ self-controlled case series — a design using each patient as their own control — found a stroke rate ratio of 2.32 for atypicals (95% CI 1.73–3.10), falling sharply with longer exposure.2 Sacchetti’s systematic review converged on the same picture.4
UK licensing limits risperidone use in dementia to 6 weeks; EU specifies short-term; US prescribing is off-label entirely.1 Continued use beyond the acute window is when the risk-benefit math gets hardest to defend — symptom benefit plateaus while cumulative stroke exposure keeps adding up.
What “Antipsychotics Cause Stroke” Misses About the Counterfactual
Three things the popular framing underweights, each of which Choma’s data quantify.
- The “no antipsychotic” comparator is rarely “no harm.” Untreated severe agitation and psychosis in dementia carry their own mortality, fall, and hospitalization risks.5 The comparison isn’t antipsychotic vs. nothing; it’s antipsychotic vs. continued non-pharmacological management or alternative drugs (off-label quetiapine, olanzapine, the SSRI citalopram), each with its own risk profile.
- Not all antipsychotics carry identical risk. Maust 2015’s number-needed-to-harm analysis of 90,786 dementia patients found haloperidol most harmful (12.3% mortality excess, NNH 8) and quetiapine least (3.2%, NNH 31), with risperidone and olanzapine between.6 Wang 2005 found typical antipsychotics carried higher mortality than atypicals at every interval.7 Switching to typicals to avoid the atypical signal is a worse trade.
- Stroke is one outcome among several. Mok 2024’s BMJ analysis of 173,910 UK dementia patients found antipsychotic use associated with elevated risks of stroke, venous thromboembolism, myocardial infarction, heart failure, fracture, pneumonia, and acute kidney injury.8 The risk profile is broader than stroke alone.
The CATIE-AD trial, the largest randomized comparison of olanzapine, quetiapine, and risperidone in Alzheimer’s, found adverse effects offset efficacy advantages over placebo across all three drugs.9 The drugs work modestly for severe symptoms; harms are quantifiable; appropriate use is short, monitored, and reserved for cases where non-drug approaches have failed.
How Choma Fits With the Older Risperidone Literature
The 1.28 overall HR sits inside a stable historical band:
- Schneider 2005 meta-analysis (15 RCTs, 5,110 dementia patients): atypicals increased mortality (OR 1.54, 95% CI 1.06–2.23) and cerebrovascular events (OR 1.9, 1.08–3.35) vs. placebo — the dataset that drove the FDA’s 2005 class-wide warning.10
- Douglas & Smeeth 2008 (BMJ self-controlled case series): rate ratio 2.32 for atypicals overall; 3.50 in dementia specifically.2
- Sacchetti 2010 systematic review: 1.3- to 2-fold elevated stroke risk in users vs. non-users; no clear class difference between typicals and atypicals.4
- Mok 2024 (BMJ, 173,910 dementia patients): stroke HR ~2 in the first 90 days, attenuating thereafter; broader harm profile spanning VTE, MI, heart failure, fracture, pneumonia.8
RCT estimates run higher than cohort estimates — relative risks above 3 in pooled trial data vs. ~1.3 in real-world cohorts.1,10 Two plausible explanations: patients prescribed risperidone in routine care differ from trial enrollees in unmeasured ways, and RCTs are short (6 to 12 weeks), capturing the early high-risk window while cohort studies dilute over longer follow-up.
The most defensible read: a 12-week HR around 1.6 to 2.0 best estimates the acute effect; 1-year HR around 1.3 over longer treatment; and absolute risk depends overwhelmingly on baseline cerebrovascular history.
Limitations of the Choma Analysis
Confounding by indication is the central caveat. Risperidone in UK primary care is licensed only for severe, persistent aggression. Patients who get prescribed are systematically sicker and more agitated than matched controls in ways the propensity model can’t fully capture.1 Mueller 2021 found risperidone prescribed for psychosis carried higher stroke risk than risperidone prescribed for agitation — an indication signal CPRD couldn’t reliably distinguish.12
Stroke severity wasn’t captured. CPRD records the event but not the disabling outcome. The hospital-admission-only sensitivity analysis — likely capturing more severe events — produced consistent HR estimates.1
Dementia subtype was not reliably distinguishable. The cohort is “all-cause dementia.” Lewy body dementia carries a separate, well-documented hypersensitivity to antipsychotics, and risperidone is generally contraindicated there.5 Choma’s results don’t specifically apply to Alzheimer’s, vascular, or Lewy body subtypes.
Adherence and dose were inferred from prescription records. CPRD captures issued prescriptions, not collected pills. Misclassification typically attenuates effects toward the null, so true HRs may be slightly higher than reported.
The dataset is UK-specific. Risperidone is the only antipsychotic licensed for severe aggression in dementia in the UK and EU; in the US it’s off-label and quetiapine dominates prescribing. Generalizability to US practice is approximate.
Practical Use of Risperidone in Dementia
The data don’t support either extreme — “never prescribe” or “the warning was overblown.”
- Non-pharmacological approaches are first-line. Environmental modification, behavioral interventions, addressing unmet needs (pain, infection, dehydration, sleep disruption), and caregiver education all reduce agitation in trials and should be exhausted before any antipsychotic.5
- Treat short when pharmacotherapy is needed. 6 weeks is the licensed UK ceiling for risperidone in dementia for a reason. The 12-week HR of 1.6 in patients without prior CVD is the period the drug is doing the most measurable damage. Beyond the acute window, taper and re-evaluate.
- Quantify absolute risk for the specific patient, not just relative risk. A 1-year +0.80% excess for a low-baseline patient is a different conversation than a +2.95% excess for a patient with prior stroke.
- Prior stroke doesn’t change the relative risk much, but it changes the math. Patients with prior cerebrovascular disease aren’t disproportionately vulnerable to risperidone in relative terms — they’re just much more likely to have a stroke regardless. Risperidone in that group adds roughly 1 stroke per 33 patients treated for a year, on top of an already-high baseline.
- Consider alternatives where indication allows. Quetiapine carries the lowest mortality signal in Maust’s analysis, though efficacy for severe agitation is more modest.6 Citalopram reduced agitation in Alzheimer’s with a different but non-trivial side-effect profile. The choice is rarely between risperidone and nothing.
- Monitor frequently in the first weeks. Check-ins at 1, 2, and 6 weeks are reasonable given the front-loaded risk profile.1
Common Questions About Risperidone and Stroke Risk in Dementia
Does risperidone really cause stroke, or is it just associated?
Multiple study designs all point the same way — randomized trials, self-controlled case series, propensity-matched cohorts — with HRs from about 1.3 in long-window observational data to above 3 in trials.1,2,10 Convergence across designs with different confounding structures is what makes causation the most defensible reading. Preclinical work links risperidone to cardiovascular biology directly.3
How much extra stroke risk does risperidone add?
It depends on baseline. With no prior stroke or CVD, about 0.80% to 0.90% extra 1-year risk — roughly 1 additional stroke per 110 to 125 patients treated for a year. With prior stroke, about 2.95% — roughly 1 per 33 patients. Relative risk is similar across groups; absolute risk is not.1
Is risperidone worse than other antipsychotics for stroke?
Within the atypical class, head-to-head data are limited. Sacchetti’s review found no clear class difference between typicals and atypicals for stroke specifically; Maust’s mortality analysis ranked haloperidol > risperidone or olanzapine > quetiapine.4,6 Risperidone is the most-studied atypical for dementia agitation because it’s the only one licensed for that indication in the UK and EU.
When does the stroke risk peak?
In the first 12 weeks. Choma’s 12-week HRs (1.33 to 1.69 across subgroups) are higher than the 1-year HRs (1.22 to 1.44).1 Treatment courses longer than the licensed 6 weeks accumulate more risk without proportionate symptom benefit.
Does the FDA black-box warning still apply?
Yes. The 2005 FDA boxed warning on increased mortality with atypicals in elderly dementia patients (extended to typicals in 2008) remains in effect. Choma’s data don’t contradict the warning; they refine it by quantifying absolute risk in clinically relevant subgroups.
What about Lewy body dementia specifically?
Patients with Lewy body dementia have a separate, well-documented neuroleptic hypersensitivity, and risperidone is generally contraindicated. Choma’s analysis didn’t reliably distinguish Lewy body cases from other dementias, so its results don’t apply there.5
References
- Risk of stroke associated with risperidone in dementia with and without comorbid cardiovascular disease: population-based matched cohort study. Choma J et al. British Journal of Psychiatry. 2025. doi:10.1192/bjp.2025.10419
- Exposure to antipsychotics and risk of stroke: self controlled case series study. Douglas IJ & Smeeth L. BMJ. 2008;337:a1227. doi:10.1136/bmj.a1227
- Whole transcriptome in-silico screening implicates cardiovascular and infectious disease in the mechanism of action underlying atypical antipsychotic side-effects. Malekizadeh Y et al. Alzheimer’s & Dementia: Translational Research & Clinical Interventions. 2020;6:e12078. doi:10.1002/trc2.12078
- Cerebrovascular accidents in elderly people treated with antipsychotic drugs: a systematic review. Sacchetti E et al. Drug Safety. 2010;33(4):273-288. doi:10.2165/11319120-000000000-00000
- Neuroleptic drugs in dementia: benefits and harm. Ballard C & Howard R. Nature Reviews Neuroscience. 2006;7(6):492-500. doi:10.1038/nrn1926
- Antipsychotics, Other Psychotropics, and the Risk of Death in Patients With Dementia: Number Needed to Harm. Maust DT et al. JAMA Psychiatry. 2015;72(5):438-445. doi:10.1001/jamapsychiatry.2014.3018
- Risk of Death in Elderly Users of Conventional vs. Atypical Antipsychotic Medications. Wang PS et al. New England Journal of Medicine. 2005;353(22):2335-2341. doi:10.1056/NEJMoa052827
- Multiple adverse outcomes associated with antipsychotic use in people with dementia: population based matched cohort study. Mok PLH et al. BMJ. 2024;385:e076268. doi:10.1136/bmj-2023-076268
- Effectiveness of Atypical Antipsychotic Drugs in Patients with Alzheimer’s Disease. Schneider LS et al. New England Journal of Medicine. 2006;355(15):1525-1538. doi:10.1056/NEJMoa061240
- Risk of death with atypical antipsychotic drug treatment for dementia: meta-analysis of randomized placebo-controlled trials. Schneider LS, Dagerman KS & Insel P. JAMA. 2005;294(15):1934-1943. doi:10.1001/jama.294.15.1934
- Baseline characteristics and treatment-emergent risk factors associated with cerebrovascular event and death with risperidone in dementia patients. Howard R et al. British Journal of Psychiatry. 2016;209(5):378-384. doi:10.1192/bjp.bp.115.177683
- Antipsychotic use in dementia: the relationship between neuropsychiatric symptom profiles and adverse outcomes. Mueller C et al. European Journal of Epidemiology. 2021;36(1):89-101. doi:10.1007/s10654-020-00643-2
