Deep brain stimulation works for Parkinson’s, but it requires implanted electrodes. A 2026 randomized crossover trial by Stalter and colleagues tested whether transcranial temporal interference stimulation — a non-invasive technique designed to reach deep targets without exciting overlying cortex — can move the needle on motor symptoms when aimed at the putamen.1
Research Highlights
- Transcranial temporal interference stimulation (tTIS) uses two high-frequency currents (e.g. 2 kHz and 2.1 kHz) that beat against each other only at their target, producing a low-frequency envelope that excites deep brain regions while leaving overlying cortex unaffected.2
- The Stalter 2026 trial enrolled 19 PD patients (mean age 64.7, on dopaminergic medication) and 19 age- and sex-matched healthy controls in a double-blind crossover design, applying intermittent theta-burst (iTBS) tTIS to the right putamen versus sham across two sessions.1
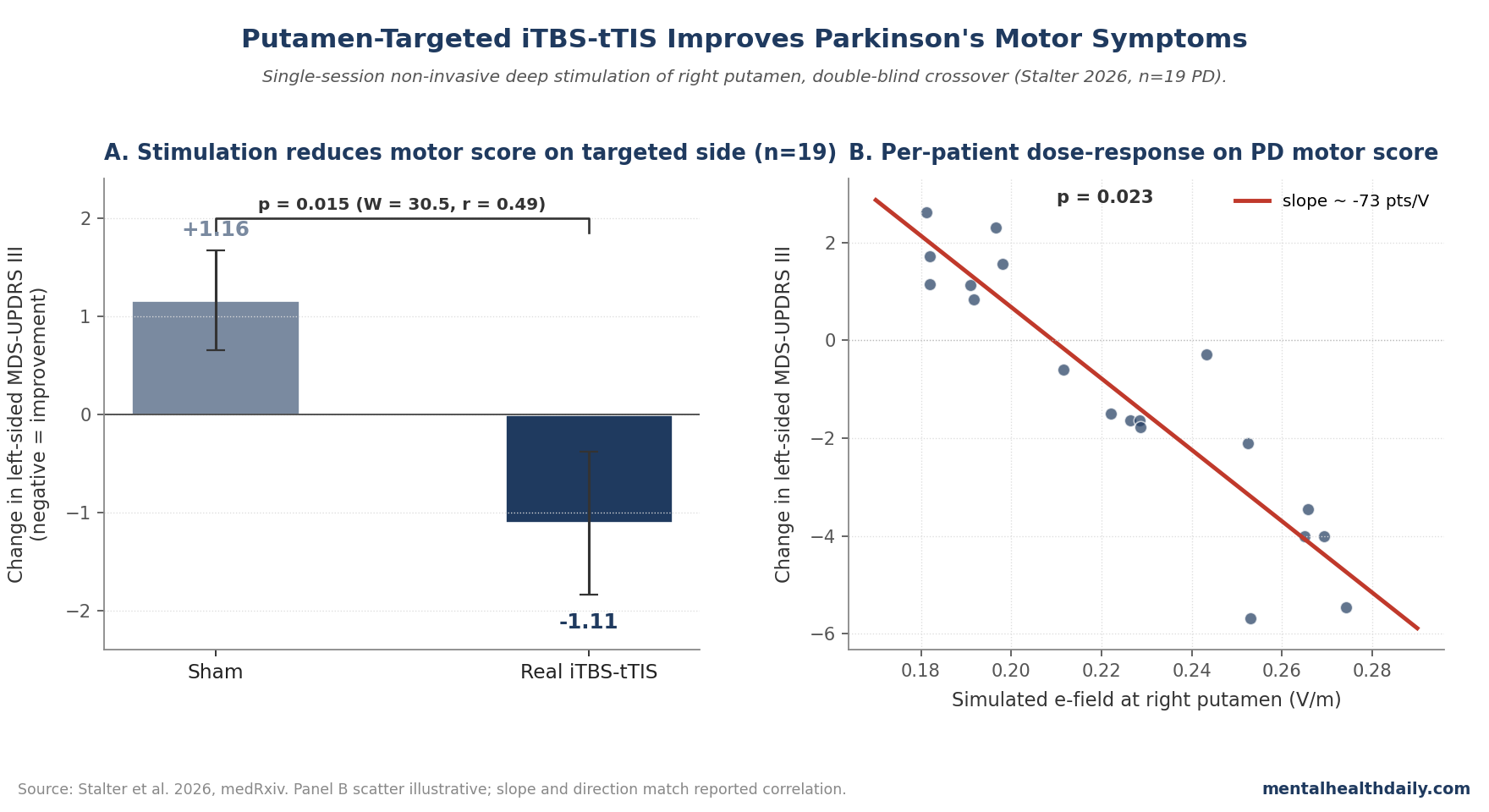
- Left-sided MDS-UPDRS III scores improved significantly with real stimulation versus sham (Wilcoxon W = 30.5, p = 0.015, r = 0.49). The effect was concentrated on the contralateral hand — consistent with stimulating the right basal ganglia.1
- Higher simulated electric field in the right putamen predicted greater motor improvement on a per-patient basis (Pearson p = 0.023), supporting a target-specific mechanism rather than a generic placebo or scalp effect.1
- No significant effects on motor learning or tapping speed. The clinical-rating signal didn’t translate to lab tasks, and no severe adverse events were reported across either group.1
Surgical deep brain stimulation of the subthalamic nucleus is highly effective in Parkinson’s disease but reserved for advanced patients who can tolerate intracranial surgery. A non-invasive method that reaches the same anatomy at a fraction of the risk profile would change the treatment landscape — not as a replacement for DBS, but as an option for earlier-stage patients and as a way to test deep targets before committing to surgery.3
Conventional non-invasive techniques like rTMS and tDCS face a depth-focality tradeoff: hitting deep targets requires intensities that hit cortex even harder. Temporal interference was proposed to break that tradeoff by exploiting the fact that neurons follow envelope frequencies, not the underlying high-frequency carriers.2,4
Stalter 2026: 19 PD Patients, Right Putamen Target, Crossover Design
The study recruited 19 patients with clinically diagnosed PD (Movement Disorder Society criteria), mean age 64.7 years, mean disease duration 6.3 years, mean Hoehn & Yahr stage 2.3, and mean levodopa equivalent daily dose ~495 mg.1 Nineteen age- and sex-matched healthy controls (mean age 68.6) were enrolled in parallel. All participants were right-handed.
The design was randomized, double-blind, crossover with three sessions per participant: clinical assessment plus structural MRI at session 1, and the two stimulation conditions (real iTBS-tTIS vs sham) at sessions 2 and 3, separated by at least 10 days of washout. PD patients were tested in the on-medication state.
The stimulation protocol used four scalp electrodes (placements F5/C5 and F6/C6) running carriers at 2 kHz and 2.1 kHz, peak-to-peak 2 mA per pair, producing an amplitude-modulated 100 Hz envelope at the right putamen. The iTBS pattern delivered 2-second trains followed by 8-second pauses for the duration of stimulation. Each participant’s scalp/skull/brain anatomy was used to simulate the actual electric field reaching the target on a per-patient basis.1
Significant Motor-Score Improvement on the Targeted Side
The pre-registered primary outcome was MDS-UPDRS Part III (the motor exam). The analysis focused on left-sided extremity items because the right putamen drives left-side motor function. The change-from-baseline difference between real and sham stimulation was significant: Wilcoxon W = 30.5, p = 0.015, effect size r = 0.49.1
Breakdown by sub-score showed the largest absolute changes in rigidity and total left-sided score, with smaller and non-significant trends for bradykinesia and tremor.1 The authors note that the rigidity and bradykinesia sub-scores have too few items to support sub-score-specific significance testing, so the meaningful claim is on the total.
The effect is small in absolute units (~1.1-point reduction on left-side total), but consistent with what a single short non-invasive session would be expected to produce.
DBS with implanted electrodes drives several-point reductions on the full bilateral UPDRS; a single 5-minute non-invasive session producing a measurable lateralized effect is the proof-of-concept goal here, not chronic symptomatic management.
The E-Field Correlation: Strongest Evidence for Mechanism
The most mechanistically convincing result isn’t the group-mean improvement — it’s the per-patient correlation between simulated electric field strength at the right putamen and the clinical effect.1
Each patient’s structural MRI was used to compute the actual field strength reaching the putamen given individual anatomy and the standardized electrode montage.
Across the 19 PD participants, higher simulated field at the target predicted greater improvement on the left-sided MDS-UPDRS (Pearson, p = 0.023). Patients whose anatomy delivered weak fields to the target showed little improvement; patients whose anatomy delivered stronger fields showed more.
This is the kind of dose-response signature that would be hard to produce by placebo or by generic scalp/cortical effects. It suggests the iTBS-tTIS protocol is doing something specifically tied to engaging the targeted basal ganglia node.1
What Didn’t Work: Tapping Speed and Motor Learning
The secondary outcomes — an alternating tapping task and a sequential finger tapping motor-learning paradigm — did not show stimulation effects in PD patients.1
The healthy control group showed an isolated tapping improvement, but the authors flag that this was likely driven by an unbalanced baseline rather than a real stimulation effect.
This null is informative. The clinical rating scale captures rigidity, postural stability, and rest tremor — symptoms that depend on tonic basal ganglia output. Lab tapping tasks measure phasic motor speed and short-timescale learning, which depend more on cortico-cerebellar circuits.
iTBS-tTIS to the putamen may shift the basal ganglia state without measurably changing the cortico-cerebellar contribution to a 2-minute tapping task.1,5
How tTIS Actually Works at the Neuron Level
Temporal interference exploits a known property of neurons: they low-pass filter rapidly oscillating fields, responding to the envelope rather than the carrier.
Two scalp-applied currents at 2.0 and 2.1 kHz pass harmlessly through cortex because no neuron can follow a 2-kHz signal. Where the two beams cross, they sum to an amplitude-modulated waveform whose envelope oscillates at 100 Hz — a frequency neurons can follow.2
Pioneer rodent work showed this could selectively activate hippocampus without touching overlying cortex.2 Subsequent human studies extended the technique to motor cortex, striatum, and hippocampus, with controlled crossover designs showing target-specific behavioral signatures.4,6
The Stalter trial sits in this lineage and adds two design features that strengthen inference: a clinical disease population (not just healthy adults) and per-patient e-field modeling correlated with clinical response.
Limitations of the Stalter Trial
Four caveats deserve weight:
- Small sample (19 per group). The crossover design and within-subject comparison reduce the noise floor, but absolute effect sizes from single trials of this scale should be treated as preliminary.
- Single short session. The clinical question worth answering — whether repeated sessions could provide sustained symptomatic benefit — is unaddressed. iTBS protocols in TMS produce after-effects lasting 30–60 minutes from a single train; whether multi-session iTBS-tTIS produces compound or durable effects is the next study, not this one.
- On-medication only. PD patients were tested in the medON state. Whether the stimulation also benefits the medOFF state — arguably a more important clinical question for advanced patients — is unknown.
- Lateralized analysis. The right-putamen target was chosen partly because tTIS field optimization was easier on one side, and analysis focused on the contralateral hand. Real-world clinical use would need to address bilateral symptoms.
The motor-learning null, while interpretable, is also a limitation: any future use of iTBS-tTIS for rehabilitation would need a different design to demonstrate learning-relevant effects.1
What Popular Coverage Often Misses
“Non-invasive deep brain stimulation” is a useful shorthand but oversells the present capability.
tTIS does reach deep targets without intervening cortical activation, but the field strengths reaching the putamen are roughly 0.2–0.25 V/m — well below the suprathreshold intensities clinical DBS delivers via implanted electrodes.
The mechanism is more likely modulatory (shifting oscillatory state) than driver-level activation.1,4
The trial does not show that tTIS replaces DBS or changes the landscape for advanced PD.
It shows that a single session of carefully targeted putamen stimulation produces a small, lateralized, dose-responsive improvement in motor symptoms in mild-to-moderate PD — a meaningful proof of concept, not a treatment ready for clinic.
Practical Implications
The Stalter results matter most as a signal that putamen-targeted iTBS-tTIS is worth scaling to a multi-session, multi-day, larger-N trial — the format that would test whether the effect can be made durable enough for clinical relevance.
A useful next study would compare iTBS-tTIS to active sham (e.g. cortical-target tTIS) rather than just zero stimulation, and would include medOFF assessments and quality-of-life endpoints.
For now, this is a preprint result in a small cohort, not a deployable therapy. But the e-field correlation, the design rigor, and the safety profile make it one of the more credible non-invasive deep-stimulation results published to date.1
More About Temporal Interference Stimulation in Parkinson’s
How is tTIS different from regular tDCS or TMS?
tDCS and TMS reach deep targets only by also stimulating overlying cortex at high intensity, which limits their utility for basal ganglia. tTIS uses two scalp-applied high-frequency currents that interfere only at the target, producing a low-frequency envelope deep in the brain while leaving cortex unaffected.2
Is this the same as deep brain stimulation (DBS)?
No. DBS uses implanted electrodes inserted surgically into the basal ganglia, with continuous chronic stimulation. tTIS is fully non-invasive, applied by scalp electrodes in single sessions of minutes–to–hours, with much lower field strengths reaching the target.1,3
How big was the symptom improvement?
About a 1.1-point reduction on left-sided MDS-UPDRS Part III (motor score) versus sham. Small in absolute units, but statistically significant and dose-responsive on a per-patient basis. Single-session non-invasive stimulation isn’t expected to produce DBS-magnitude effects.1
Why target the right putamen specifically?
The putamen is a major basal ganglia input from the substantia nigra dopaminergic neurons that degenerate in PD. Right-sided targeting drives left-sided motor function; the trial used left-side MDS-UPDRS items as the contralateral readout.1
Were there side effects?
No severe adverse events. Mild and brief sensations were reported (redness, transient fatigue, mild dysesthesia), distributed roughly evenly between real and sham sessions.1
Could this replace DBS for advanced PD?
Not on the strength of this trial. Field strengths and effect sizes are modulatory, not driver-level. The plausible clinical niche is earlier-stage symptom modulation, presurgical target testing, and rehabilitation adjuncts — not advanced motor fluctuations.1,3
Why didn’t tapping speed improve?
Tapping tasks depend heavily on cortico-cerebellar timing networks, which weren’t the stimulation target. Clinical rating scales capture rigidity and postural-stability features more dependent on basal ganglia state.1,5
What’s the next study that needs to happen?
Multi-session iTBS-tTIS over days to weeks, larger sample, medOFF assessment, and quality-of-life endpoints — to test whether the single-session signal scales into clinically durable benefit.
References
- Stalter J, Stecher H, Bergmann L, et al. Intermittent theta-burst transcranial temporal interference stimulation focusing on the putamen improves motor functions in Parkinson’s disease — a randomized, controlled trial. medRxiv. 2026. doi:10.64898/2026.02.05.26345627.
- Grossman N, Bono D, Dedic N, et al. Noninvasive deep brain stimulation via temporally interfering electric fields. Cell. 2017;169(6):1029–1041.e16. doi:10.1016/j.cell.2017.05.024
- Deuschl G, Schade-Brittinger C, Krack P, et al. A randomized trial of deep-brain stimulation for Parkinson’s disease. New England Journal of Medicine. 2006;355:896–908. doi:10.1056/NEJMoa060281
- Violante IR, Alania K, Cassarà AM, et al. Non-invasive temporal interference electrical stimulation of the human hippocampus. Nature Neuroscience. 2023;26:1994–2004. doi:10.1038/s41593-023-01456-8
- Ni Z, Bahl N, Gunraj CA, et al. Increased motor cortical facilitation and decreased inhibition in Parkinson’s disease. Neurology. 2013;80(19):1746–1753. doi:10.1212/wnl.0b013e3182919029
- Wessel MJ, Beanato E, Popa T, et al. Non-invasive theta-burst stimulation of the human striatum enhances striatal activity and motor skill learning. Nature Neuroscience. 2023;26:2005–2016. doi:10.1038/s41593-023-01457-7