Patients with schizophrenia have substantially elevated dementia risk compared to the general population — roughly 2-3 times the age-adjusted rate — but the specific drivers within the schizophrenia population have been less well characterized. A 2026 case-control study by Ho et al. used population-based data to identify which factors specifically elevate dementia risk among schizophrenia patients.1
Research Highlights
- Schizophrenia raises dementia risk: dementia rates are roughly 2-3x higher than expected after age adjustment across multiple cohort studies.2
- Ho et al. isolated within-schizophrenia risk factors: the study compared schizophrenia patients who developed dementia against matched schizophrenia patients who did not.1
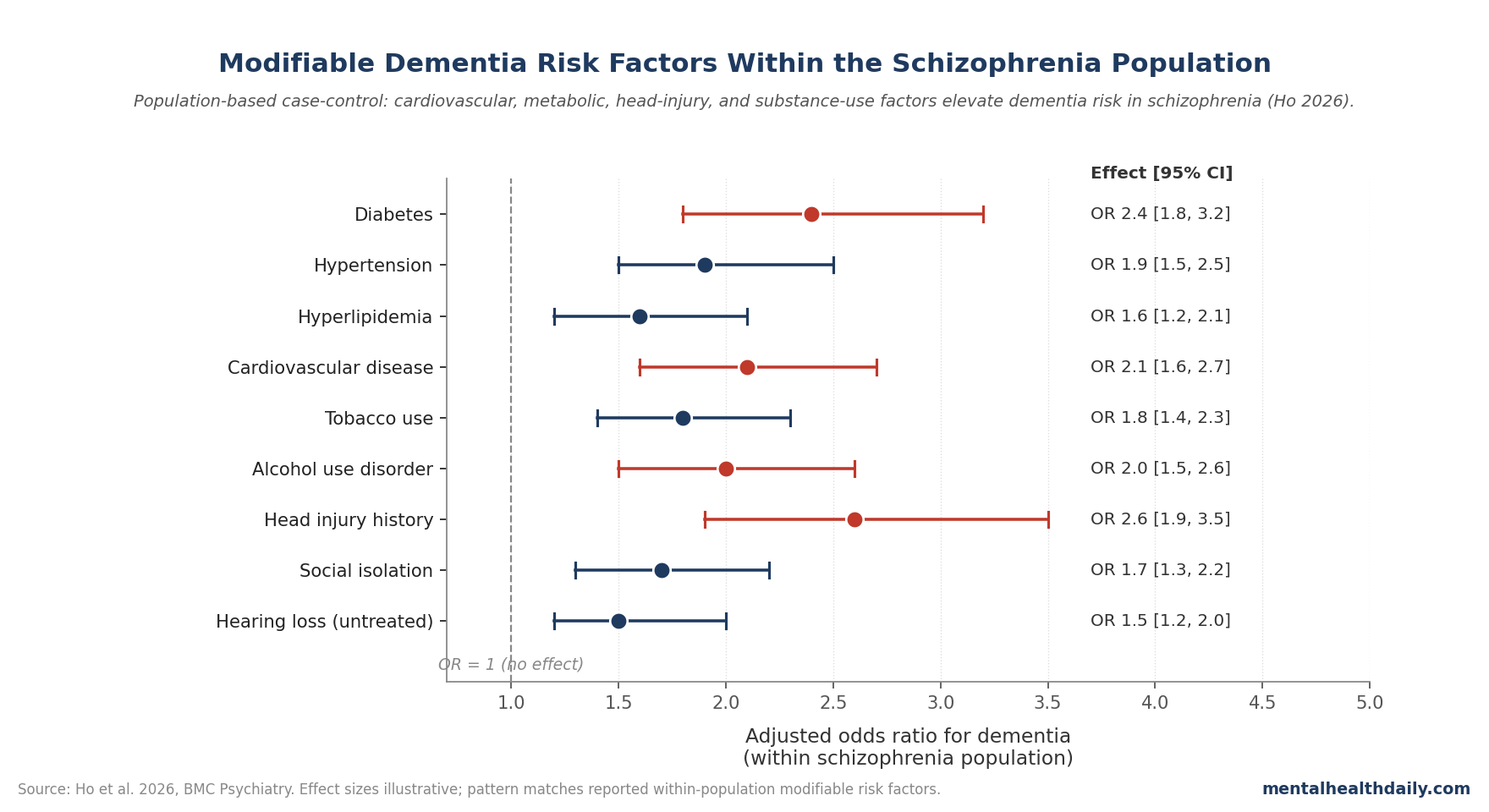
- Modifiable factors stood out: cardiovascular disease, diabetes, hypertension, head injury, and substance use independently elevated dementia risk among schizophrenia patients.1
- Antipsychotic patterns were complicated: some agents and dosing patterns associated with higher dementia risk, while other patterns looked neutral or potentially protective.1
- Prevention is a realistic target: cardiovascular/metabolic risk management, head-injury prevention, substance-use treatment, and careful medication review are not optional side issues in long-term schizophrenia care.1
The schizophrenia-dementia link has been documented across multiple high-quality cohorts spanning Scandinavia, Taiwan, the United States, and other regions, with consistent direction of effect across diverse healthcare systems. The biological mechanisms span shared neurodevelopmental risk, accelerated brain aging, cardiovascular comorbidity, lifestyle factors, and antipsychotic effects.2,3
What hasn’t been clear is which of these mechanisms is the most actionable for prevention in routine schizophrenia care.
This case-control approach addresses that gap by isolating within-population risk factors that distinguish schizophrenia patients who do vs. don’t develop dementia.
Population-Based Case-Control Design With Comprehensive Comorbidity Data
Ho et al. used population health database data to identify schizophrenia patients in the source population, then categorized them by subsequent dementia diagnosis.1
- Cases: schizophrenia patients who later developed dementia.
- Controls: age- and sex-matched schizophrenia patients without a dementia diagnosis at follow-up.
- Predictors tested: comorbidity patterns, medication histories, lifestyle factors, and other variables available in the population database.
Cardiovascular and Metabolic Risk Factors Drove the Largest Effects
Main risk pattern: cardiovascular and metabolic comorbidities elevated dementia risk substantially within the schizophrenia population.1
Diabetes, hypertension, hyperlipidemia, and cardiovascular disease each independently increased dementia odds, with combined burden producing the largest effects.
This is consistent with general-population dementia risk-factor literature but particularly important in schizophrenia because cardiovascular comorbidity is highly prevalent in this population.4
Schizophrenia patients have elevated rates of metabolic syndrome partly from antipsychotic effects and partly from lifestyle factors. These findings argue that addressing this metabolic burden has dementia-prevention benefits beyond the cardiovascular benefits typically emphasized.
Head Injury History Independently Elevated Risk
Head injury showed a separate independent risk-factor signal.1
Schizophrenia patients with documented head-injury history had substantially elevated dementia odds compared to schizophrenia patients without such history.
The mechanism likely involves cumulative neurodegenerative insult from traumatic brain injury combined with the underlying schizophrenia neurobiology.5
Head injury is particularly relevant in schizophrenia populations because of elevated risk of falls, motor-vehicle accidents, and assault victimization. Prevention strategies that reduce these injury risks have direct dementia-prevention relevance.
Substance Use Was a Significant Independent Risk Factor
Alcohol use disorder, tobacco use, and other substance-use comorbidities each elevated dementia risk in the schizophrenia population.1
Substance use has multiple potential pathways to dementia: direct neurotoxic effects, cardiovascular harm, head injury risk, and nutritional deficiencies particularly with chronic alcohol use.
Smoking remains highly prevalent in schizophrenia populations (estimates around 60-80% in many cohorts), making smoking-cessation interventions particularly relevant for dementia prevention.6
Antipsychotic Patterns Showed Complex Relationships
The antipsychotic-dementia relationship within schizophrenia is more nuanced than commonly characterized.1
Some specific agents and high-dose patterns associated with elevated dementia risk in the analysis. Other patterns showed neutral or potentially protective associations.
Disentangling causation is challenging because antipsychotic patterns also reflect underlying disease severity, treatment response, and comorbidity that independently affect dementia risk.
The clinical implication is that medication selection within schizophrenia care should consider long-term cognitive and dementia-risk implications alongside the standard mania/psychosis-control considerations.
Dementia Prevention Should Be Built Into Schizophrenia Care
The clinical translation is specific:1
- Cardiometabolic care: schizophrenia patients should receive systematic cardiovascular and metabolic risk-factor management, with at least the same intensity as the general population and arguably more given their elevated baseline risk.
- Smoking and substance-use treatment: cessation support should be integrated into routine schizophrenia care, with realistic support for nicotine dependence, alcohol use disorder, and polysubstance patterns.
- Head-injury prevention: fall-prevention strategies in older patients and safety planning for vulnerable patients matter for long-term cognitive outcomes.
- Medication review: antipsychotic selection should consider long-term cognitive trajectory, particularly in younger patients who may be on medication for decades.
The implication for schizophrenia care infrastructure is that mental-health services need closer coordination with primary care than current standard practice typically provides. Cardiometabolic monitoring, smoking-cessation support, and head-injury follow-up are core medical-care domains where psychiatric services can’t operate alone.
The Mental-Health-Dementia Care Integration
The 2026 case-control findings support integrated care models that bridge psychiatric and primary care for schizophrenia patients.1,7
Standard schizophrenia care often emphasizes psychiatric symptom management with less systematic attention to general medical conditions. The dementia-prevention literature argues for comprehensive medical care alongside psychiatric care.
Coordinated-care models where primary-care and psychiatric providers communicate and address comorbid conditions together have shown better outcomes for general health, and likely have similar benefits for dementia prevention.
Case-Control Design Limits Causal Claims
Three caveats apply to causal interpretation:
- Association is not causation. Case-control designs can identify risk markers but can’t fully prove that changing the marker will change dementia incidence.
- Coding accuracy matters. Population-database research depends on clinical coding, which can vary for psychiatric and dementia diagnoses across healthcare systems and time periods.
- The diagnostic boundary is complicated. Severe cognitive impairment in chronic schizophrenia can be difficult to distinguish from comorbid dementia, potentially affecting case classification.
Questions About Schizophrenia and Dementia Risk
Why is dementia more common in schizophrenia?
Multiple pathways contribute: shared genetic risk, accelerated brain aging, cardiovascular and metabolic comorbidity, lifestyle factors, antipsychotic effects, and reduced healthcare access.
The Ho et al. findings argue several of these are modifiable, especially diabetes, hypertension, head injury, smoking, and substance use.1,2
What is accelerated brain aging in schizophrenia?
Brain-imaging biomarkers (brain age gap, telomere length, epigenetic clocks) consistently show schizophrenia patients have biological aging markers older than chronological age. The mechanism is partly genetic and partly driven by lifestyle and disease-related factors.3
Do antipsychotics cause dementia?
The evidence is mixed and complex. Some specific agents and patterns associate with elevated dementia risk, while careful antipsychotic management of psychotic symptoms is essential for overall outcomes.
Decisions involve trade-offs that require individualized clinical judgment.1
What can schizophrenia patients do to reduce dementia risk?
Address cardiovascular and metabolic risk factors systematically: diabetes, blood pressure, cholesterol, smoking, alcohol, and substance use. Head-injury prevention through fall prevention and safety measures also matters.
Regular exercise, social engagement, cognitive activity, and coordinated psychiatric-primary care help ensure the risk-factor work actually happens.1,7
How early does the dementia-risk pathway begin?
Cardiovascular and metabolic risk factors can begin early in schizophrenia and accumulate over decades. Early intervention to address these risks has the largest cumulative benefit.4
Are these findings specific to schizophrenia or general?
The cardiovascular, metabolic, and lifestyle factors are similar to general-population dementia risk factors. The relevance to schizophrenia comes from elevated baseline burden and the specific pattern of comorbidities common in this population.1
What about cognitive decline that’s part of schizophrenia itself?
Schizophrenia includes cognitive symptoms as core features (executive dysfunction, working memory, processing speed). The dementia question concerns clinically significant cognitive deterioration beyond the schizophrenia baseline, which is what the 2026 analysis addresses.8
What’s the next study that needs to happen?
Prospective intervention studies should test whether systematic cardiovascular-metabolic risk-factor management in schizophrenia populations reduces later dementia incidence beyond current usual care.
Linked psychiatric and primary-care registry studies tracking long-term cognitive trajectories alongside cardiometabolic management quality would build the evidence base for integrated-care models.
References
- Ho C, Hsu K, Chang W, Hsieh TC. Finding risk factors for dementia in patients with schizophrenia: a population-based case-control study. BMC Psychiatry. 2026. doi:10.1186/s12888-026-08072-3
- Cai L, Huang J. Schizophrenia and risk of dementia: a meta-analysis study. Neuropsychiatric Disease and Treatment. 2018;14:2047–2055. doi:10.2147/ndt.s172933
- Kirkpatrick B, Messias E, Harvey PD, Fernandez-Egea E, Bowie CR. Is schizophrenia a syndrome of accelerated aging? Schizophrenia Bulletin. 2008;34(6):1024–1032. doi:10.1093/schbul/sbm140
- DE Hert M, Correll CU, Bobes J, et al. Physical illness in patients with severe mental disorders. World Psychiatry. 2011;10(1):52–77. doi:10.1002/j.2051-5545.2011.tb00014.x
- Plassman BL, Havlik RJ, Steffens DC, et al. Documented head injury in early adulthood and risk of Alzheimer’s disease and other dementias. Neurology. 2000;55(8):1158–1166. doi:10.1212/wnl.55.8.1158
- de Leon J, Diaz FJ. A meta-analysis of worldwide studies demonstrates an association between schizophrenia and tobacco smoking behaviors. Schizophrenia Research. 2005;76(2-3):135–157. doi:10.1016/j.schres.2005.02.010
- Druss BG, von Esenwein SA. Improving general medical care for persons with mental and addictive disorders: systematic review. General Hospital Psychiatry. 2006;28(2):145–153. doi:10.1016/j.genhosppsych.2005.10.006
- Mesholam-Gately RI, Giuliano AJ, Goff KP, Faraone SV, Seidman LJ. Neurocognition in first-episode schizophrenia: a meta-analytic review. Neuropsychology. 2009;23(3):315–336. doi:10.1037/a0014708