SSRIs raise bleeding risk on their own. DOACs (direct oral anticoagulants — apixaban, rivaroxaban, dabigatran, edoxaban) raise it more. The intuitive worry is that combining them stacks the two effects. A new BJGP Open analysis from Chau and colleagues argues the stacking is smaller than most popular framings claim, and that the real safety question is narrower: what happens in the first weeks after an SSRI is added on top of an existing DOAC.1
Research Highlights
- The headline DOAC + SSRI bleeding risk shrinks when the comparison is fair. In Chau and colleagues’ 2026 UK cohort of 75,527 anticoagulated patients, DOAC + SSRI users had no excess intracranial bleeding (HR 1.16, 99% CI 0.62–2.20), gastrointestinal bleeding (HR 1.09, 99% CI 0.83–1.41), or other bleeding (HR 1.01, 99% CI 0.78–1.29) versus DOAC + non-SSRI antidepressant.
- Earlier alarming numbers used the wrong comparator. Cohorts pitting DOAC + SSRI against DOAC alone reported 25% to 92% increased major bleeding. That gap mostly reflects depression itself (and the way depressed patients differ from non-depressed ones), not the SSRI.
- The initiation window is the only place a real signal appears. Starting an SSRI while already on a DOAC was associated with higher odds of “other bleeding” (case-crossover OR 1.64, 99% CI 1.14–2.35; Wald p = 0.002). Non-SSRI antidepressants showed no equivalent spike.
- SSRIs deplete platelet serotonin. Platelets don’t synthesize serotonin; they take it up via the same SERT transporter SSRIs block. Lower platelet serotonin means weaker aggregation when a vessel breaks — the documented mechanism behind SSRI bleeding signals.
- Practical read: stay on the SSRI, watch the first 30 days, treat NSAIDs and antiplatelets as the bigger lever. Avoidable interactions live in NSAID + SSRI co-prescription (a stronger and more replicated risk) and in starting an SSRI cold on a DOAC patient with active bleeding history.
Chau 2026: 75,527 Patients, No Excess Bleeding vs. Active Comparator
Chau and colleagues used UK Clinical Practice Research Datalink (CPRD) Aurum primary care data linked to hospital admissions from 2011 to 2021. They identified 35,782 DOAC + SSRI users and 39,745 DOAC + non-SSRI antidepressant users (mirtazapine, tricyclics, etc.) and ran two complementary designs in parallel.1
The cohort design used inverse-probability-of-treatment weighting (a method that re-weights patients so the SSRI and non-SSRI groups look statistically similar at baseline). The case-crossover design compared each individual’s drug exposure in the week before a bleed to their exposure two months earlier — a within-person design that controls for any patient trait that doesn’t change over time.
The cohort numbers are the headline. After propensity score weighting, DOAC + SSRI users had:
- Intracranial bleeding: HR 1.16 (99% CI 0.62–2.20) — CI crosses 1.1
- Gastrointestinal bleeding: HR 1.09 (99% CI 0.83–1.41) — CI crosses 1.1
- Other bleeding: HR 1.01 (99% CI 0.78–1.29) — CI crosses 1.1
(HR, or hazard ratio, is roughly the ratio of bleeding rates between groups; 1.0 means equal, above 1.0 means the SSRI group bleeds more. The 99% CI — confidence interval — is the range the true ratio is likely to sit in. When the interval crosses 1.0, the data don’t rule out “no difference.”)
None of the three intervals excludes 1.0. The point estimates sit essentially on the null. Compared to a patient already on a DOAC who needs an antidepressant, switching from a non-SSRI to an SSRI did not measurably raise bleeding hazard.
Why Earlier Studies Reported 25%–92% Increased Bleeding
The Chau result lands inside a literature that mostly reads scarier. Several earlier studies reported substantial bleeding excess on DOAC + SSRI:
- A Belgian atrial fibrillation cohort by Grymonprez and colleagues reported HR 1.26 (95% CI 1.17–1.35) for major or clinically relevant non-major bleeding, HR 1.50 for intracranial, and HR 1.27 for gastrointestinal bleeding when DOAC patients used SSRIs/SNRIs.2
- A Taiwanese AF cohort reported a 38% increase in intracranial bleeding for DOAC + SSRI versus DOAC alone.3
- UK and South Korean nested case-control studies reported 68% and 92% increased odds of major bleeding for DOAC + SSRI versus DOAC alone.4,5
- A 2024 JAMA Network Open analysis by Rahman and colleagues, using the same CPRD data through 2021, reported a 25% higher major bleeding risk for DOAC + SSRI versus DOAC without SSRI.6
The shared design flaw across these studies: they compare DOAC + SSRI to DOAC alone. That comparison conflates the SSRI’s effect with depression itself, with whatever drove the SSRI prescription, and with how depressed patients differ from non-depressed ones (more falls, less follow-up, different comorbidity patterns).
Chau’s study used DOAC + non-SSRI antidepressant as the comparator. Both groups have a treated mood disorder, both are anticoagulated, both visited a prescriber for an antidepressant. The only systematic difference is which class of antidepressant was picked. That comparison strips out depression-the-confounder and isolates the question that matters at the bedside: does swapping an SSRI in for a non-SSRI antidepressant raise bleeding risk in someone already on a DOAC?
Two earlier studies that did use SSRI vs. non-SSRI antidepressant as the comparator agree with Chau. An Austrian nationwide cohort found no excess hospitalization for GI bleeding, cerebral hemorrhage, or anemia in oral-anticoagulant users on SSRIs versus non-SSRIs.7 An American AF cohort similarly found no major bleeding signal for DOAC + SSRI versus DOAC alone.8 The pattern is consistent: when the comparator controls for the indication, the SSRI signal mostly disappears.
Initiating an SSRI on a DOAC: A Transient 64% Bleeding Increase
The cohort design averages risk across whoever happens to be on both drugs at any given time. The case-crossover design asks something more specific: what happens in the days right after a new prescription gets added?
Chau’s case-crossover used a 6-parameter model that estimates separate odds ratios for six drug-initiation patterns — SSRI started while already on DOAC, DOAC started while already on SSRI, both started together, monotherapy initiation of either, and so on. The pattern that flagged a documented interaction signal was specifically initiating an SSRI while already taking a DOAC.
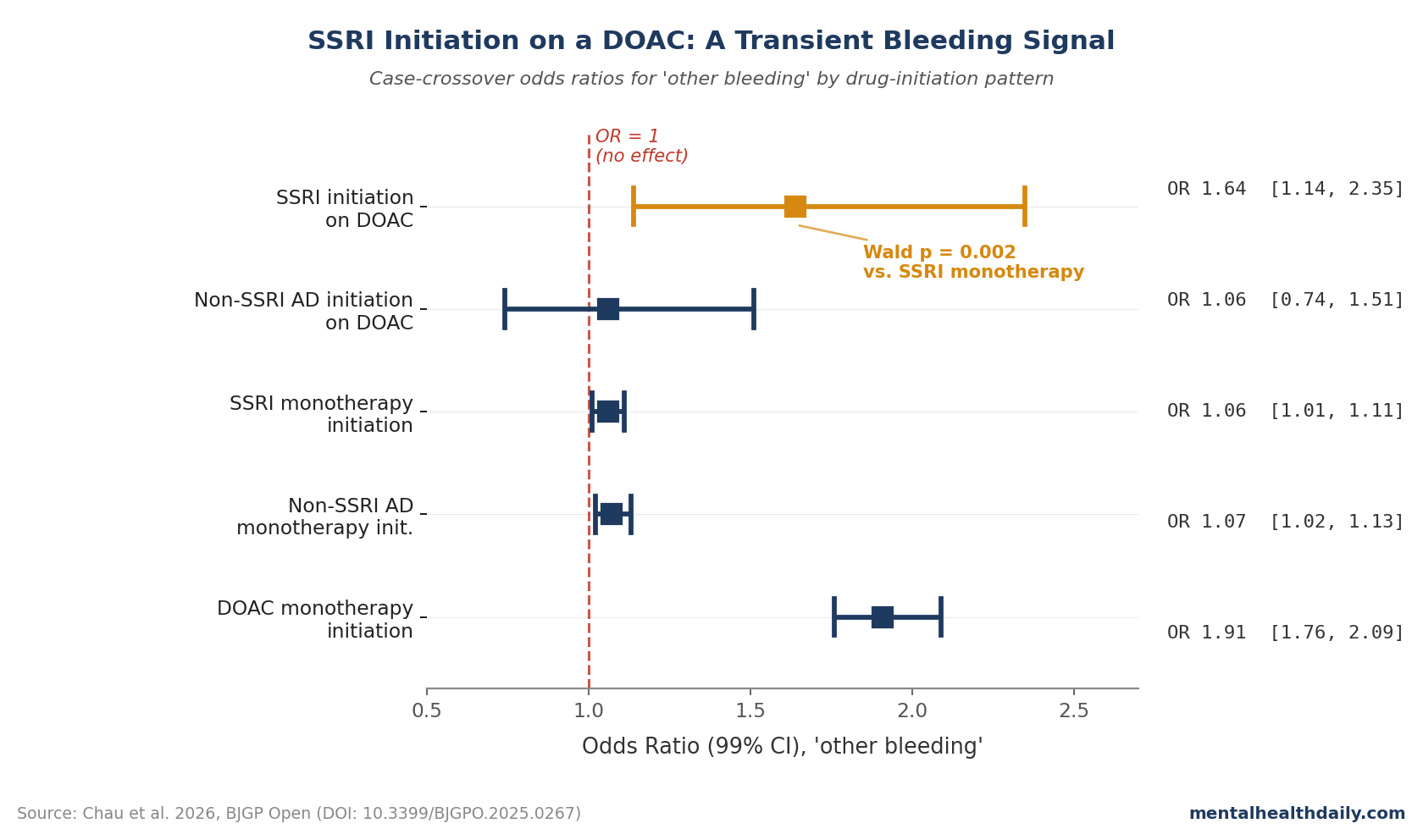
For “other bleeding” (a composite that excludes intracranial and GI — mostly skin, urinary, and respiratory tract bleeds), the odds ratio for SSRI initiation while on a DOAC was 1.64 (99% CI 1.14–2.35). Compared to SSRI monotherapy initiation (OR 1.06, 99% CI 1.01–1.11), the difference reached statistical significance (Wald p = 0.002).1
Three caveats deserve weight before reading this as a clinical alarm.
- The signal didn’t appear for intracranial or GI bleeding. Wald p-values were 0.42 and 0.18 respectively — consistent with chance. The interaction is restricted to a less-severe outcome category.
- The signal was design-specific. The cohort design, which weights average risk across all DOAC + SSRI patient-time, found no excess for any bleeding type. The case-crossover signal lives only in the narrow window right after a new SSRI prescription.
- Non-SSRI antidepressants showed no equivalent spike on initiation while taking DOAC (Wald p = 0.83 for other bleeding). That class-specificity argues for an SSRI-mediated mechanism rather than a general “starting any new antidepressant” effect.
The most defensible read: the average DOAC + SSRI patient is not at meaningfully elevated bleeding risk relative to a DOAC + non-SSRI antidepressant patient. The first few weeks after adding an SSRI to an existing DOAC may carry a modest, transient excess of minor and moderate bleeding events — worth a heads-up, not worth refusing the prescription.
Mechanism: Why SSRIs Affect Platelets at All
Platelets don’t make their own serotonin. They scoop it up from blood plasma via the serotonin transporter (SERT) — the same protein SSRIs block in neurons. Long-term SSRI use depletes intra-platelet serotonin stores, and serotonin is one of the cofactors platelets release to recruit other platelets when a vessel breaks. The net effect is weaker platelet aggregation, an antiplatelet effect comparable in size to low-dose aspirin in some assays.9
SSRIs alone modestly increase upper GI bleeding (Dalton 2003 reported an adjusted relative risk of 3.6 for current SSRI use, dropping to 1.3 once NSAIDs were excluded).10 SSRIs plus NSAIDs increase it more; SSRIs plus warfarin increase it more again. Chau’s data suggest DOACs fit lower on that gradient than warfarin did, with the residual signal concentrated in the initiation window rather than steady state.
The SSRIs also differ in SERT-binding affinity — paroxetine, sertraline, and fluoxetine bind tighter than citalopram and escitalopram — but bleeding-risk data aren’t consistent enough across agents to recommend switching on that basis alone.11
Limitations of the Chau Analysis
Power for intracranial bleeding is thin. Only 31 events occurred in the DOAC + non-SSRI group and 35 in the DOAC + SSRI group. The 99% CI (0.62–2.20) rules out a tripling of risk but not a doubling. A null result with this little power is reassuring, not definitive.
Adherence wasn’t captured. CPRD records prescriptions, not whether the patient took the pills. Nondifferential misclassification biases estimates toward the null, which can hide a small genuine interaction inside a non-significant point estimate.
The population was predominantly White and UK-based. The Taiwanese cohort that found a 38% intracranial bleeding excess raises a question about whether ethnicity-linked differences in CYP metabolism or co-prescription patterns matter here.1
Median follow-up was short — 27 days. Chronic-use bleeding risk over years isn’t tested in this analysis. It captures the early-window risk where the case-crossover signal lives, not steady-state risk over years.
Residual confounding can’t be ruled out. Propensity score weighting balances measured covariates only. The within-person case-crossover helps for time-varying confounders but not stable traits that affect both prescribing and bleeding.
Practical Co-Prescribing of SSRIs and DOACs
The co-prescription is common and the underlying need is well-documented. Atrial fibrillation, venous thromboembolism, and post-stroke status all elevate depression and anxiety risk; depressed patients with cardiovascular disease are routinely under-treated for the mood disorder, with measurable consequences for outcomes and adherence. Refusing an SSRI to a DOAC patient is rarely the right call.
- Don’t avoid the SSRI on bleeding risk alone. Active-comparator data (Chau 2026, Sheikh Rezaei 2019, Yao 2025) converge on no clinically meaningful excess at steady state.
- Watch the first 30 days. The case-crossover signal localizes to SSRI initiation on an existing DOAC, with a 64% relative increase in odds of minor and moderate bleeding. A check-in at 2 and 4 weeks for new bruising, gum bleeding, hematuria, or epistaxis is cheap insurance.
- Treat NSAIDs as the bigger lever. The Laporte meta-analysis pegged SSRI bleeding risk at OR 1.41 alone, jumping with NSAID co-prescription.11 Switching ibuprofen to acetaminophen does more for bleeding risk than switching the antidepressant class would.
- Add a PPI when the indication is GI-relevant. 54% of Chau’s cohort were already on PPIs. Peptic ulcer history, prior GI bleed, or NSAID use should trigger PPI co-prescription.
- Reconsider SSRI defaults if intracranial risk dominates. Renoux 2017 found higher intracranial hemorrhage signals for SSRIs than non-SSRI antidepressants — small absolute risk, documented relative signal.12 For patients with prior intracranial bleeding, mirtazapine is a reasonable first-line alternative.
Common Questions About SSRIs and Blood Thinners
Is it safe to take an SSRI like sertraline with apixaban or rivaroxaban?
For most patients, yes. The largest active-comparator cohort to date (Chau 2026, n = 75,527) found no excess intracranial, gastrointestinal, or other bleeding for DOAC + SSRI versus DOAC + non-SSRI antidepressant.1 The clinically meaningful signal, when present, lives in the first weeks after an SSRI is added on top of a DOAC — not at steady state.
Why do some studies say SSRIs increase bleeding by 25% or more on DOACs?
Most of those studies compared DOAC + SSRI to DOAC alone. That comparison conflates the SSRI’s effect with depression itself, with comorbidity patterns, and with whatever else differs between depressed and non-depressed anticoagulated patients. When studies use DOAC + non-SSRI antidepressant as the comparator (Chau 2026, Sheikh Rezaei 2019, Yao 2025), the excess largely disappears.1,7,8
Which SSRI is safest for bleeding risk?
Direct comparisons are limited. SERT-binding affinity correlates roughly with platelet effect, which suggests citalopram, escitalopram, and fluvoxamine sit at the lower end and paroxetine, sertraline, and fluoxetine at the higher end. The clinical bleeding-risk gap between agents has not been consistent enough across studies to support agent-level switching for bleeding alone. Comorbidities, concomitant drugs, and prior response usually matter more.11
What about SNRIs (venlafaxine, duloxetine) with DOACs?
SNRIs share SSRIs’ SERT-blocking effect on platelets and likely carry a similar bleeding profile. The Belgian cohort that flagged a 26% major bleeding increase pooled SSRIs and SNRIs together.2 Chau 2026 used non-SSRI antidepressants excluding SNRIs as the active comparator, so it doesn’t speak directly to SNRIs.
What signs of bleeding should I watch for?
New or unusual bruising, prolonged nosebleeds, bleeding gums, blood in urine, black or tarry stools, coffee-ground vomit, severe headache, or sudden unexplained weakness. The first 30 days after adding an SSRI to an existing DOAC are the highest-risk window for minor and moderate bleeding; severe bleeds aren’t elevated above baseline DOAC risk.
References
- Potential interactions between SSRIs and DOACs: population-based cohort and case-crossover study. Chau M et al. BJGP Open. 2026. doi:10.3399/BJGPO.2025.0267
- Pharmacodynamic Drug-Drug Interactions and Bleeding Outcomes in Patients with Atrial Fibrillation Using Non-Vitamin K Antagonist Oral Anticoagulants: a Nationwide Cohort Study. Grymonprez M et al. Cardiovascular Drugs and Therapy. 2024;38(2):209-221. doi:10.1007/s10557-023-07535-z
- Major Bleeding Risk in Patients With Non-valvular Atrial Fibrillation Concurrently Taking Direct Oral Anticoagulants and Antidepressants. Chang KH et al. Frontiers in Aging Neuroscience. 2022;14:791285. doi:10.3389/fnagi.2022.791285
- Risk of major bleeding among users of direct oral anticoagulants combined with interacting drugs: A population-based nested case-control study. Zhang Y et al. British Journal of Clinical Pharmacology. 2020;86(6):1150-1164. doi:10.1111/bcp.14227
- Concomitant Use of NSAIDs or SSRIs with NOACs Requires Monitoring for Bleeding. Lee MT et al. Yonsei Medical Journal. 2020;61(9):741-749. doi:10.3349/ymj.2020.61.9.741
- Concomitant Use of Selective Serotonin Reuptake Inhibitors With Oral Anticoagulants and Risk of Major Bleeding. Rahman AA et al. JAMA Network Open. 2024;7(3):e243208. doi:10.1001/jamanetworkopen.2024.3208
- SSRI co-medication with NOAC or VKA does not increase hospitalisation for bleeding: A retrospective nationwide cohort study in Austria 2010-2015. Sheikh Rezaei S et al. International Journal of Geriatric Psychiatry. 2019;34(8):1194-1199. doi:10.1002/gps.5119
- Evaluating Major Bleeding Risks with Concomitant Use of Direct Oral Anticoagulants and Selective Serotonin Reuptake Inhibitors in Nonvalvular Atrial Fibrillation Patients. Yao SY et al. Pharmacoepidemiology. 2025;4(1):6. doi:10.3390/pharma4010006
- Serotonin Reuptake Inhibitors and Risk of Abnormal Bleeding. Andrade C & Sharma E. Psychiatric Clinics of North America. 2016;39(3):413-426. doi:10.1016/j.psc.2016.04.010
- Use of selective serotonin reuptake inhibitors and risk of upper gastrointestinal tract bleeding: a population-based cohort study. Dalton SO et al. Archives of Internal Medicine. 2003;163(1):59-64. doi:10.1001/archinte.163.1.59
- Bleeding risk under selective serotonin reuptake inhibitor (SSRI) antidepressants: A meta-analysis of observational studies. Laporte S et al. Pharmacological Research. 2017;118:19-32. doi:10.1016/j.phrs.2016.08.017
- Association of Selective Serotonin Reuptake Inhibitors With the Risk for Spontaneous Intracranial Hemorrhage. Renoux C et al. JAMA Neurology. 2017;74(2):173-180. doi:10.1001/jamaneurol.2016.4529