For two decades, headlines have linked autism to maternal obesity, depression, anxiety, gestational diabetes, infection, autoimmune disease, and antidepressant use. The studies behind those headlines are well-replicated registry associations. A new design from the Aarhus University iPSYCH group offers the most careful answer yet to what they mean for an individual pregnancy. Most “maternal X causes autism” framings don’t survive the analyses that separate inherited liability from in-utero causation.
Research Highlights
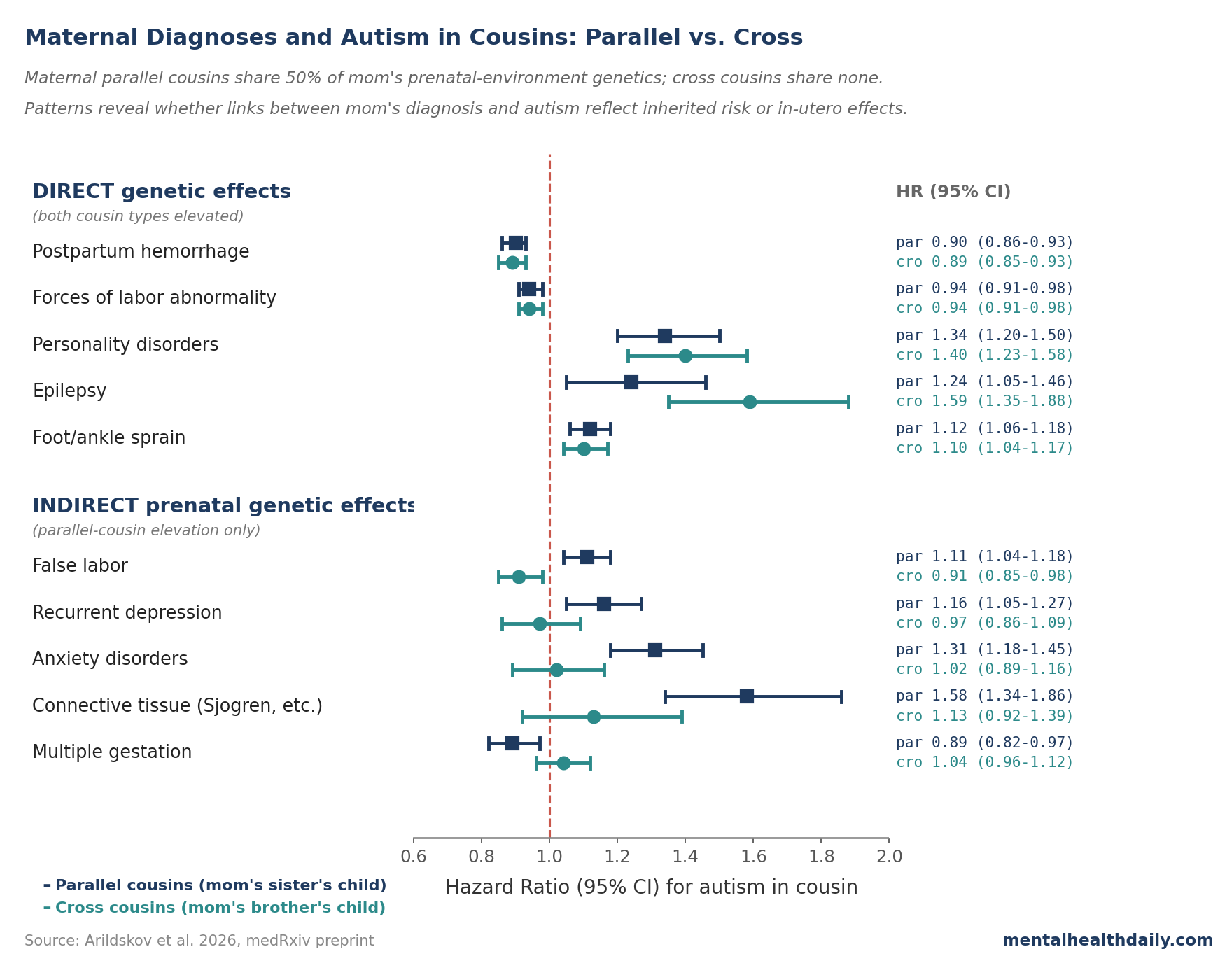
- Most observed links between maternal conditions in pregnancy and offspring autism are not direct in-utero causal effects. A new Danish 3-generation registry analysis of 1,131,899 children finds that for many maternal diagnoses previously associated with autism, the apparent link reflects shared family genetics rather than the diagnosis affecting the developing fetus.1
- Postpartum hemorrhage, personality disorders, and epilepsy in the mother track autism risk equally in cousins on her sister’s and brother’s side — the signature of inherited (direct genetic) liability passing through the family tree, not pregnancy itself.1
- Recurrent depression, anxiety disorders, false labor, and connective tissue conditions show stronger associations on the maternal-sister side only. That pattern is consistent with maternal-side genetic variants shaping the prenatal environment in ways that affect neurodevelopment — an indirect genetic effect, not an environmental one.1
- The same paper’s predecessor showed effects shrinking sharply in sibling comparisons. Khachadourian and colleagues’ 2025 Nature Medicine study on the same Danish cohort found that 30 maternal diagnoses initially associated with autism mostly shrank toward null after sibling comparison and paternal negative-control analyses.2
- This does not erase known causal exposures. Valproate during pregnancy roughly triples autism risk in a way that survives sibling comparison;3 rubella infection in early pregnancy is a long-established teratogen. The new findings narrow the list of plausibly causal maternal exposures, not eliminate it.
What the 3-Generation Cousin Design Tests
Arildskov and colleagues (2026, medRxiv preprint) used Denmark’s national registers to identify all 1,131,899 children born from 1998 to 2015, plus their parents and grandparents. From that source population they built two kinds of cousin pairs:1
- Maternal parallel cousins. The mothers are full sisters. Their children share 12.5% of inherited DNA AND share half of the maternal-side prenatal-environment genetics — the variants in the mother that shape how she carries a pregnancy.
- Maternal cross cousins. The index mother and the cousin’s father are full siblings. Their children share the same 12.5% of inherited DNA, but the cousin’s mother is genetically unrelated to the index mother — so they share none of the maternal-side prenatal-environment genetics.
The two cousin types are matched on transmitted (direct) genetic risk. They differ on whether they share maternal-side prenatal-environment genetics. That single contrast is the analytic engine.
If a maternal diagnosis is associated with autism in both cousin types, the most plausible reading is inherited risk — alleles that show up as the maternal diagnosis AND get transmitted to children, where they raise autism risk independently. The diagnosis itself isn’t doing anything to the fetus; it’s a flag for shared family genetics.
If a maternal diagnosis is associated with autism only on the maternal-sister side, maternal-side prenatal genetics are doing something the cross-cousin side is missing — how the mother metabolizes folate, how her immune system behaves in pregnancy, or another genetically-influenced feature of how she carries the fetus.
The design is elegant because it works without genotype data. It’s built entirely on Denmark’s register-based pedigree and ICD-10 diagnosis codes.
Where the Evidence Points to Inherited Risk, Not Pregnancy
Five maternal diagnoses produced the “both cousin types elevated” pattern consistent with direct genetic effects:1
- Postpartum hemorrhage — parallel HR 0.90, cross HR 0.89. Both cousin types had lower autism risk. The direction matches reduced fecundity in both maternal sisters and brothers of affected mothers, suggesting shared genetic factors driving both.
- Specific personality disorders — parallel HR 1.34, cross HR 1.40. Closely matched. Genetic correlations between psychiatric phenotypes and autism are well-documented in modern GWAS.10
- Epilepsy — parallel HR 1.24, cross HR 1.59. Both elevated, with the cross-cousin signal stronger — consistent with genetic liability passing through male relatives without going through the index pregnancy.
- Forces-of-labor abnormalities and foot/ankle sprain — both elevated similarly in both cousin types. Harder to interpret causally; likely reflect shared behavioral or personality factors rather than biological pregnancy effects.
When a maternal condition’s autism signal shows up just as strongly in children of the mother’s brothers as in children of her sisters, the pregnancy itself is not the mechanism. Inherited risk running through the family is.
Where Maternal-Side Prenatal Genetics Look Like They Matter
Five conditions produced the parallel-only pattern consistent with indirect prenatal genetic effects:1
- Recurrent major depressive disorder. Parallel HR 1.16 (1.05–1.27); cross HR 0.97 (0.86–1.09). Ratio 1.19 (p = 0.025).
- Other anxiety disorders. Parallel HR 1.31 (1.18–1.45); cross HR 1.02 (0.89–1.16); ratio 1.29 (p = 0.004). Among the strongest indirect-genetic signals in the data.
- False labor. Parallel HR 1.11; cross HR 0.91; ratio 1.22 (p = 4.3 × 10−5) — the only indirect-effects finding that survives FDR correction in the primary analysis.
- Connective tissue conditions (the M35 category covering Sjogren’s syndrome and joint hypermobility). Parallel HR 1.58 (1.34–1.86); cross HR 1.13 (0.92–1.39); ratio 1.39 (p = 0.012).
- Multiple gestation. Parallel HR 0.89 (lower risk on the maternal-sister side); cross HR 1.04. The two cousin types point opposite ways.
For these, the most plausible reading is that maternal-side genetics shape the prenatal environment — serotonin biology, immune-system behavior, labor character — in ways that affect neurodevelopment. That is still “genetics,” meaningfully, but acting through the womb rather than through transmitted alleles.
This is not the popular framing. “Maternal anxiety raises autism risk” suggests pregnancy stress hormones harming the fetus. The Arildskov result is more specific: maternal-side genetic variants linked to anxiety appear to influence the prenatal environment, but anxiety in the unrelated cross-cousin’s mother does not produce the same signal.
How the Cousin Design Improves on Sibling Comparisons
The Khachadourian 2025 Nature Medicine paper from this same Danish cohort is the immediate predecessor. It tested 236 maternal diagnoses, found 30 statistically associated with autism, and then re-ran the analyses using sibling comparisons and paternal negative controls.2 Sibling comparison contrasts full siblings of the same mother where one was exposed to her diagnosis and one wasn’t — differences within sibships can’t be due to factors siblings share, including 50% of mom’s genetics.4 The paternal-negative-control move tests whether the father’s version of the same diagnosis predicts offspring autism just as strongly; if it does, the maternal pregnancy isn’t the mechanism.
Most associations shrank sharply in those analyses, which is why the press releases read “no evidence maternal sickness during pregnancy causes autism.” That language oversells in the other direction. The right calibration is “most of the popular maternal-risk-factor associations look confounded, but a handful retain weight.”
Arildskov 2026 picks up where Khachadourian 2025 left off. Sibling comparisons can’t fully separate two kinds of family confounding: directly transmitted alleles versus indirect genetic effects through the prenatal environment, because siblings share both. The cousin design does separate them, by exploiting that maternal cross cousins share inherited risk but not maternal-side prenatal-environment genetics.
Limitations of the Arildskov Cousin Design
It is a preprint, not yet peer-reviewed. The methodology is novel and the assumptions are clearly laid out, but the analysis hasn’t passed external review. Read the inferences as provisional.
Sex-asymmetric fecundity could bias the comparison. Several maternal diagnoses (alcohol use disorder, schizophrenia, bipolar disorder, eating disorders, OCD) reduced fecundity more in maternal brothers than in maternal sisters. That asymmetry could selectively filter which children end up in the cross-cousin pool, dragging some HR estimates around. Arildskov flags this explicitly.
Assortative mating could erode the cross-cousin contrast. The design assumes the index mother shares no maternal-side prenatal genetics with the cross-cousin’s mother. If people mate non-randomly on traits associated with the diagnoses, the cross-cousin’s mother is partially “related” to the index family, which shrinks the apparent indirect-genetic signal.
Most indirect signals don’t survive FDR correction. Only false labor survives FDR adjustment in the primary analysis (Q = 0.0006); the others (recurrent depression Q = 0.07, anxiety Q = 0.07, connective tissue Q = 0.08, multiple gestation Q = 0.08) are nominally significant but fail multiple-testing correction. Treat them as plausible signals worth further investigation, not as confirmed pathways.
Some prior sibling findings don’t replicate here. Maternal obesity and OCD effects shrank by more than 80% in Khachadourian’s sibling analyses but didn’t produce a cross-cousin gap large enough to register here. The two designs answer subtly different questions, and the indirect-effects list shouldn’t be treated as exhaustive.
Valproate, Rubella, and the Short List of Causal Exposures
The careful read is not that nothing in pregnancy matters for autism risk. It’s that the list of maternal exposures with credible direct causal effects is much shorter than popular coverage implies. A few entries on it are very well-established:
Valproate. Christensen and colleagues’ 2013 JAMA study reported a 4.42% absolute risk of autism in valproate-exposed children versus 1.53% in unexposed, with adjusted hazard ratios near 3.0 that hold up after adjusting for parental psychiatric history and epilepsy.3 The biology is strong (valproate is a histone deacetylase inhibitor with documented teratogenic effects), and the effect size dwarfs anything in the registry-association literature for confounded exposures. Valproate is the clearest causal medication exposure for autism.
Rubella in early pregnancy. Long-established before modern epidemiology, with congenital rubella syndrome producing autism rates orders of magnitude above population baseline before universal vaccination.
Severe early-pregnancy infection. Atladottir and colleagues’ original Danish cohort showed elevated autism risk after viral infection requiring hospitalization in the first trimester.5 Brynge and colleagues’ 2022 Lancet Psychiatry sibling analysis pulled most of the maternal-infection-autism signal toward null (within-sibling HR 0.94, 95% CI 0.82–1.08).6 A small residual signal for severe early-pregnancy infection requiring hospitalization is biologically plausible via maternal immune activation; mild infections don’t survive sibling adjustment.
Antidepressants do not belong on this list. Hviid and colleagues’ 2013 NEJM Danish cohort of 626,875 births found a small unadjusted SSRI-autism association that became non-significant after adjustment.7 Sujan and colleagues’ 2017 JAMA Swedish sibling analysis found no within-sibship effect of first-trimester antidepressant exposure (sibling HR 0.83, 95% CI 0.62–1.13).8 Prenatal SSRI exposure is not a credible direct cause of autism; the crude associations reflected confounding by the underlying maternal depression.
The clinical implications run calmer than the average headline. Treating maternal depression and anxiety in pregnancy — with therapy, with SSRIs where indicated — is not credibly causing offspring autism.
Valproate remains the single most important medication to discuss with women of childbearing age who have epilepsy or bipolar disorder, with alternatives preferred in pregnancy where clinically possible. The long worry-list (gestational diabetes, mild SSRI use, maternal anxiety, mild infection) mostly isn’t actionable for autism risk specifically — standard pregnancy care addresses these for the mother’s sake.
Common Questions About Maternal Health and Autism
Does maternal depression during pregnancy cause autism?
The crude association is well-documented; the causal interpretation is not. Recurrent depression in mothers is associated with autism in their children (HR 1.16 in Arildskov 2026), but the signal concentrates on the maternal-sister side of the family tree — meaning maternal-side genetics are doing the work, not depression itself.1 Sibling-comparison and paternal-negative-control analyses point the same way.2
Are SSRIs in pregnancy linked to autism?
In population-level data, mild associations exist; in sibling comparisons that strip out shared family confounding, the associations vanish. Hviid 2013 (Denmark, 626,875 births)7 and Sujan 2017 (Sweden, sibling design)8 are the strongest tests: prenatal SSRI exposure does not credibly cause autism. The earlier case-control signal in Croen 20119 reflected confounding by the underlying maternal depression rather than the medication.
Does maternal infection during pregnancy cause autism?
Most of the original associations shrink sharply in sibling comparisons. Brynge 2022 in Lancet Psychiatry reported a within-sibling HR of 0.94 (0.82–1.08) for autism after maternal infection — effectively null.6 A small residual signal for severe early-pregnancy infection requiring hospitalization is biologically plausible (maternal immune activation has substantial preclinical support) but is much smaller than headlines have suggested.
What about valproate?
Valproate is in a different category. The Christensen 2013 JAMA data showed a roughly 3-fold elevation in autism risk that holds up after adjustment for parental epilepsy and psychiatric history,3 the biology (HDAC inhibition, documented teratogenicity for other neurodevelopmental outcomes) is well-supported, and the effect size dwarfs anything seen for confounded exposures. Women of childbearing age with epilepsy or bipolar disorder should discuss alternatives with prescribers where clinically feasible.
If most maternal-condition links to autism are genetic, what does “genetic” mean here?
It splits into two kinds. Direct genetic effects are the conventional version: alleles transmitted from parent to child that affect the child’s development. Indirect genetic effects refer to a parent’s genotype shaping the child’s environment in outcome-relevant ways — for autism, maternal-side genetics shaping the prenatal environment. Both are family-level factors, but only direct effects involve the child inheriting the relevant variant.
References
- Maternal health and autism risk: parsing direct and indirect genetic effects using 3-generation family linkage. Arildskov ES et al. medRxiv (preprint). 2026. doi:10.64898/2026.04.15.26350976
- Familial confounding in the associations between maternal health and autism. Khachadourian V et al. Nature Medicine. 2025;31:996-1007. doi:10.1038/s41591-024-03479-5
- Prenatal valproate exposure and risk of autism spectrum disorders and childhood autism. Christensen J et al. JAMA. 2013;309(16):1696-1703. doi:10.1001/jama.2013.2270
- Within-sibship genome-wide association analyses decrease bias in estimates of direct genetic effects. Howe LJ et al. Nature Genetics. 2022;54:581-592. doi:10.1038/s41588-022-01062-7
- Maternal infection requiring hospitalization during pregnancy and autism spectrum disorders. Atladottir HO et al. Journal of Autism and Developmental Disorders. 2010;40:1423-1430. doi:10.1007/s10803-010-1006-y
- Maternal infection during pregnancy and likelihood of autism and intellectual disability in children in Sweden: a negative control and sibling comparison cohort study. Brynge M et al. The Lancet Psychiatry. 2022;9(10):782-791. doi:10.1016/S2215-0366(22)00264-4
- Use of selective serotonin reuptake inhibitors during pregnancy and risk of autism. Hviid A, Melbye M, Pasternak B. New England Journal of Medicine. 2013;369:2406-2415. doi:10.1056/NEJMoa1301449
- Associations of maternal antidepressant use during the first trimester of pregnancy with preterm birth, small for gestational age, autism spectrum disorder, and attention-deficit/hyperactivity disorder in offspring. Sujan AC et al. JAMA. 2017;317(15):1553-1562. doi:10.1001/jama.2017.3413
- Antidepressant use during pregnancy and childhood autism spectrum disorders. Croen LA et al. Archives of General Psychiatry. 2011;68(11):1104-1112. doi:10.1001/archgenpsychiatry.2011.73
- Identification of common genetic risk variants for autism spectrum disorder. Grove J et al. Nature Genetics. 2019;51:431-444. doi:10.1038/s41588-019-0344-8
