A 2026 Dutch stroke-center cohort found the opposite of the damaged draft’s claim: patients with a migration background were more likely to reach the hospital outside the therapeutic window for acute reperfusion therapy, and they had sharply lower odds of receiving endovascular thrombectomy.1
Research Highlights
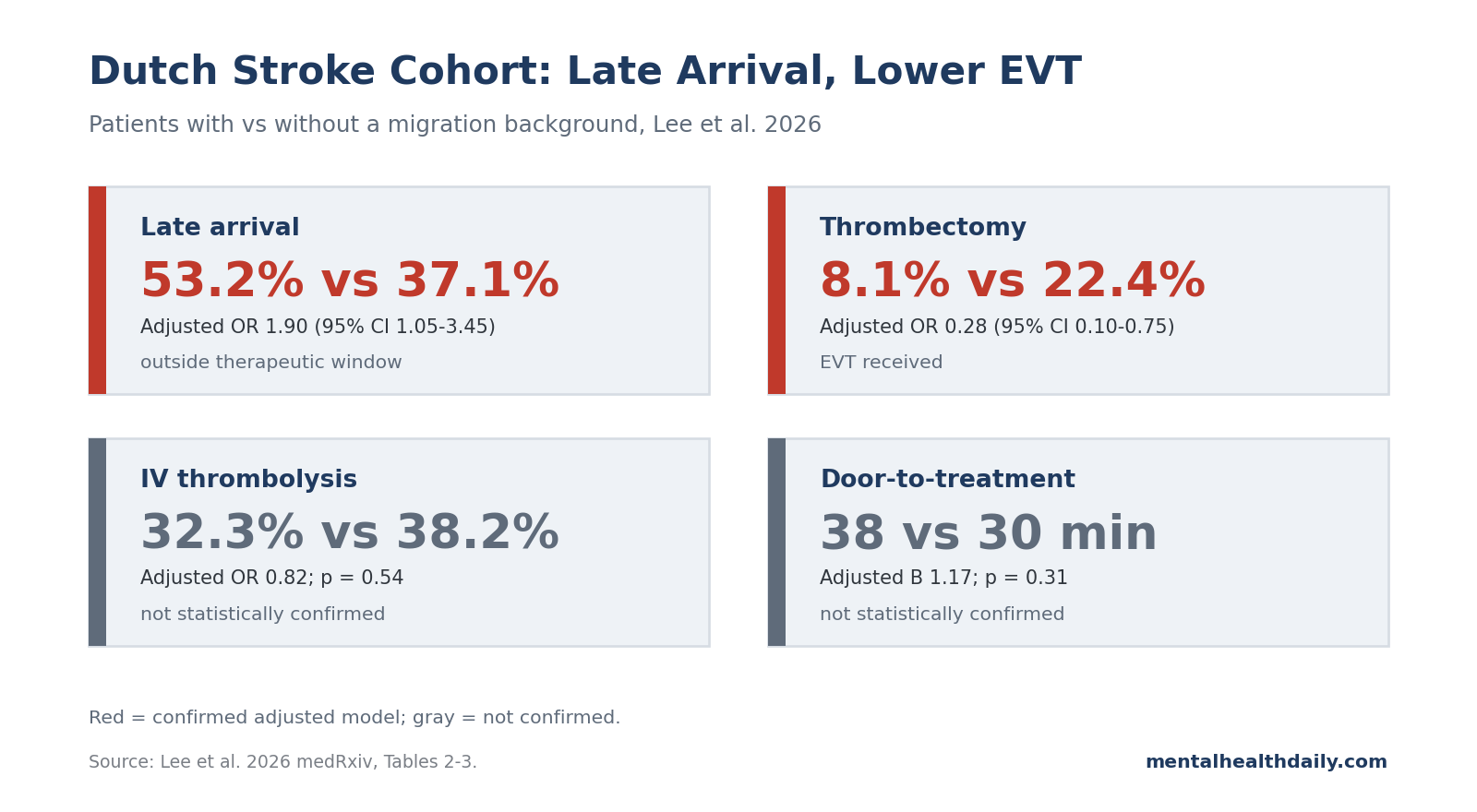
- Late arrival was the clearest disparity: 53.2% of migrant-background patients vs. 37.1% of non-migrant-background patients presented outside the therapeutic window; the adjusted odds ratio was 1.90 (95% CI 1.05–3.45; p = 0.03).1
- EVT separated more than IVT: endovascular thrombectomy occurred in 8.1% vs. 22.4% of patients (adjusted OR 0.28; 95% CI 0.10–0.75; p = 0.01), while IV thrombolysis was 32.3% vs. 38.2% (adjusted OR 0.82; p = 0.54).1
- Any-reperfusion treatment leaned lower but did not clear significance: 35.5% of migrant-background patients vs. 49.4% of non-migrant-background patients received IVT, EVT, or both (adjusted OR 0.62; 95% CI 0.32–1.20; p = 0.15).1
- In-hospital speed was not the proven bottleneck: median door-to-treatment time was 38 vs. 30 minutes, but the adjusted comparison was not statistically confirmed (B 1.17; 95% CI 0.86–1.61; p = 0.31).1
- The broader literature points the same way: a 2025 meta-analysis reported lower odds of IVT (OR 0.69; p = 0.001) and EVT (OR 0.87; p = 0.005) among Black vs. white patients, while a 2025 London cohort found lower thrombolysis odds in Black Caribbean patients (OR 0.56; 95% CI 0.40–0.80).2,4
Reperfusion therapy means treatment that tries to reopen blocked blood flow during ischemic stroke, usually by dissolving a clot with intravenous thrombolysis (IVT) or removing it with endovascular thrombectomy (EVT), a catheter procedure for large-vessel occlusion. Both are time-sensitive: delayed arrival can make a patient ineligible before the hospital team has a chance to treat.
The Lee et al. finding is not subtle once the tables are read directly. The cohort did not show faster migrant-background arrival with an in-hospital treatment gap.
It showed more late arrival, a lower EVT rate, a non-significant any-reperfusion gap, and no statistically confirmed difference in door-to-needle or door-to-groin time.1
53.2% of Migrant-Background Patients Arrived Outside the Treatment Window
Cohort setup: Lee et al. analyzed 232 adults with first-ever ischemic stroke who presented to Haaglanden Medical Center in The Hague between September 2020 and September 2021. Of those patients, 62 (26.7%) had a migration background under the Dutch Statistics Netherlands definition: the patient was born outside the Netherlands or had at least 1 parent born outside the Netherlands.1
Timing result: presentation outside the therapeutic time window happened in 33 of 62 patients with a migration background (53.2%) and 63 of 170 patients without a migration background (37.1%). After adjustment for age and sex, migration background remained associated with higher odds of late presentation (OR 1.90; 95% CI 1.05–3.45; p = 0.03).1
the time window decides eligibility. For IVT, the traditional window is measured in hours from symptom onset; for EVT, selected patients can be treated later, but eligibility still depends on imaging, occlusion type, tissue viability, and how fast the stroke pathway moves.
Patient-level pooled trial data have tied faster arrival-to-thrombectomy treatment to more healthy life-years, so a patient who arrives too late can look undertreated in the final data even if the hospital team follows protocol.6
The old draft’s “faster arrival, less treatment” framing was therefore not a calibration problem. It was the wrong direction.
The corrected read is less flashy and more useful: late presentation appears to be a major upstream contributor, but it does not fully explain every treatment pattern.
EVT Was 8.1% vs. 22.4%, While IVT Did Not Show the Same Gap
The treatment results split by therapy type. Any acute reperfusion therapy was lower in the migrant-background group: 22 of 62 patients (35.5%) vs. 84 of 170 (49.4%).
The adjusted estimate still leaned lower (OR 0.62), but its confidence interval crossed 1.00 (95% CI 0.32–1.20; p = 0.15), so the study did not statistically confirm an all-reperfusion gap.1
IVT also did not separate cleanly. It was used in 20 of 62 migrant-background patients (32.3%) and 65 of 170 non-migrant-background patients (38.2%), with an adjusted OR of 0.82 (95% CI 0.43–1.55; p = 0.54).
That point estimate leans lower, but the data are compatible with a small decrease, no difference, or even a modest increase.
EVT was different. Only 5 of 62 migrant-background patients (8.1%) underwent thrombectomy, compared with 38 of 170 patients without a migration background (22.4%).
The adjusted OR was 0.28 (95% CI 0.10–0.75; p = 0.01), meaning the strongest treatment disparity in this cohort was the procedure used for large-vessel occlusion stroke.1
That distinction prevents overcorrection. The Dutch data do not justify saying migrant-background patients were uniformly denied reperfusion treatment after reaching the hospital.
The precise finding is narrower: late presentation was more common, EVT use was substantially lower, and IVT did not show a confirmed independent gap.
Stroke Mechanism May Explain Part of the EVT Gap
EVT is not offered to every ischemic-stroke patient. It is mainly relevant when a large artery is blocked, especially when imaging shows a large-vessel occlusion that can be reached and reopened mechanically.
Lee et al. found that stroke mechanism differed by migration background, which complicates a simple access-only interpretation.1
Small-vessel disease was much more common among patients with a migration background: 43 of 62 (69.4%) vs. 81 of 170 (47.6%), with an adjusted standardized residual of 2.9. Cardioembolic stroke was less common: 3 of 62 (4.8%) vs. 26 of 170 (15.3%), with an adjusted standardized residual of −2.1.
Large-artery atherosclerosis was also numerically lower, 6.5% vs. 15.9%, though that post-hoc residual did not reach the same threshold.1
Small-vessel disease usually means a smaller penetrating artery is affected, often in the setting of chronic vascular risk such as hypertension or diabetes. Cardioembolism means a clot traveled from the heart, often from atrial fibrillation, and can lodge in a larger artery.
Because large-vessel occlusions are the usual EVT target, a cohort with more small-vessel disease and less cardioembolism can have less EVT eligibility even before any disparity in care delivery is considered.
Best read: the EVT odds ratio is strong, but the study was not large enough to fully untangle late arrival, occlusion type, stroke mechanism, imaging eligibility, language barriers, and clinician workflow.
The correct conclusion is not “bias caused the thrombectomy gap.” It is that the EVT gap remained a serious signal, with plausible contributions from both prehospital timing and stroke-subtype distribution.
Door-to-Treatment Times Were Directionally Longer but Not Confirmed
If the disparity were mainly inside the hospital, the timing table should show clearly slower treatment after arrival. It does not.
Median door-to-treatment time was 38 minutes in migrant-background patients vs. 30 minutes in non-migrant-background patients, but the adjusted log-transformed comparison was not statistically significant (B 1.17; 95% CI 0.86–1.61; p = 0.31).1
The same pattern held when the researchers separated the 2 treatment clocks. Door-to-needle time for IVT was 35.5 vs. 26 minutes (adjusted B 1.19; 95% CI 0.90–1.57; p = 0.22).
Door-to-groin time for EVT was 64 vs. 54 minutes (adjusted B 1.39; 95% CI 0.77–2.49; p = 0.26).1
These are not proof of equal hospital workflow. The point estimates all lean slower for migrant-background patients.
But a small cohort with only 5 EVT-treated migrant-background patients cannot cleanly rule in a within-hospital delay mechanism. The strongest supported hierarchy is: late arrival was statistically confirmed, EVT receipt was statistically confirmed, and in-hospital treatment-time differences were plausible but not confirmed.
Earlier Dutch and UK Data Put the Lee Result in Context
Amsterdam signal: the corrected Lee interpretation fits better with the surrounding literature than the damaged draft did. A 2011 Amsterdam hospital study by Coutinho et al. included 510 ischemic-stroke patients, 392 white and 118 non-white.
Non-white patients had lower odds of thrombolysis than white patients (OR 0.34; 95% CI 0.17–0.71).5
That older Dutch study predated the modern EVT era, so it cannot answer the thrombectomy question directly. It still established the same broad mechanism: lower acute treatment may partly start before arrival.
Symptom recognition, emergency activation, language access, and trust can all affect how quickly patients reach the emergency department.
London signal: a 2025 South London cohort by Emmett et al. adds a universal-health-system comparison. Among 7,280 participants with stroke, Black Caribbean patients had lower thrombolysis rates than white patients (adjusted OR 0.56; 95% CI 0.40–0.80) and more delayed arrival (arrival >4 hours: 60.0% vs. 51.2%).4
Black African and Black Caribbean patients also had poorer functional outcomes despite better survival than white patients.4
That UK finding is useful because it weakens a simplistic insurance-access explanation. Universal coverage can remove one barrier while leaving other barriers intact: symptom recognition, emergency communication, health-system trust, care navigation, vascular-risk burden, and neighborhood-level access patterns.
Meta-analytic signal: Biswas et al.’s 2025 DARTS meta-analysis covered 38 studies of access to reperfusion therapy and found lower odds of IVT and EVT among Black patients compared with white patients.2
Ikeme et al.’s 2022 systematic review of 30 US studies similarly found that white patients were more likely to arrive within 3 hours and more likely to receive acute reperfusion than Black, Hispanic, or Asian patients.3
The Mental-Health Link Runs Through Disability, Not Stroke Etiology
MHD should cover this topic because stroke is a vascular event with psychiatric and cognitive aftermath. Post-stroke disability changes depression risk, cognition, social independence, family burden, sleep, employment, and rehabilitation engagement.
An acute treatment disparity can therefore become a mental-health disparity months later.
Hackett and Pickles’ updated systematic review estimated that depression after stroke is common across observational studies, and post-stroke depression is not a cosmetic outcome. It is tied to quality of life, rehabilitation participation, cognitive burden, and long-term functioning.7
Reperfusion treatment is not a depression treatment, and Lee et al. did not measure post-stroke depression as the primary outcome. The link is indirect but clinically important: delayed arrival and lower EVT eligibility can worsen neurological disability, and worse disability increases the load on mental health after stroke.
For migrant-background patients, that load may be compounded by language barriers, care-navigation difficulty, and weaker access to culturally competent rehabilitation or mental-health follow-up.
What the Lee Study Can and Cannot Support
The study’s strongest contribution is its separation of 4 pieces that are often blended together: arrival timing, eligibility, treatment receipt, and in-hospital treatment speed. Once those pieces are separated, the repair target becomes clearer.
- Prehospital delay needs direct intervention. A 53.2% late-arrival rate cannot be solved only by improving door-to-needle time after the patient arrives.
- EVT eligibility needs better explanation. The 8.1% vs. 22.4% EVT gap may reflect late arrival, fewer large-vessel occlusions, referral workflow, imaging selection, language barriers, or a mix of those mechanisms.
- Hospital workflow still deserves audit. Door-to-treatment estimates leaned slower in migrant-background patients even though they were not statistically confirmed, so process mapping should not be dismissed.
- Language-related selection bias may understate the problem. The cohort excluded patients unable to complete Dutch questionnaires because of insufficient Dutch, severe cognitive problems, or severe aphasia; that exclusion can remove exactly the patients most likely to face communication barriers.
The practical takeaway is not a slogan about patient responsibility or hospital bias. It is a sequence: improve symptom recognition and emergency activation in migrant communities, measure large-vessel occlusion and EVT eligibility transparently, audit treatment clocks after arrival, and track 3-month disability and mental-health outcomes by migration background.
Reader Questions About Stroke Disparities
Did Lee 2026 show the disparity happened before or inside the hospital?
Best read: the strongest confirmed disparity was before effective treatment could start: 53.2% of migrant-background patients vs. 37.1% of non-migrant-background patients arrived outside the therapeutic window. EVT receipt was also lower, but the study could not fully separate late arrival from stroke mechanism, imaging eligibility, and hospital workflow.1
Why was thrombectomy lower if IV thrombolysis was not clearly different?
Eligibility matters: EVT depends heavily on having a treatable large-vessel occlusion. Lee et al. found more small-vessel disease (69.4% vs. 47.6%) and less cardioembolism (4.8% vs. 15.3%) among migrant-background patients, which can reduce EVT eligibility.
That does not erase the disparity, but it makes the mechanism more complicated than “same strokes, different treatment.”1
Does migration background mean race, ethnicity, or country of birth?
In this Dutch study, migration background followed the Statistics Netherlands definition: born outside the Netherlands or having at least 1 parent born outside the Netherlands. It is not a clean race variable, and it combines different communities, languages, migration histories, and risk profiles into 1 analytic group.1
Could language barriers still matter if patients with insufficient Dutch were excluded?
Yes. The exclusion makes language barriers harder to measure, not less important.
If patients with insufficient Dutch were less likely to enter the analytic cohort, the study may undercount the subgroup most likely to experience communication delays during symptom recognition, ambulance activation, history-taking, and consent.
How should families use this finding during a possible stroke?
Use emergency services immediately. Do not wait for symptoms to resolve, do not drive around looking for a familiar clinic, and do not let uncertainty about language or documentation delay activation.
The Dutch data do not prove every late arrival was preventable, but the 53.2% late-window rate makes rapid emergency activation the most concrete patient-side lever.1
Why does this belong on a mental-health site?
Disability is the bridge: post-stroke depression, cognitive burden, and loss of independence are downstream mental-health outcomes. Acute stroke treatment affects disability; disability affects depression risk and rehabilitation engagement.
The mental-health relevance is strongest when the article tracks the pathway from emergency treatment access to later functioning, not when it treats stroke as a purely vascular topic.7
References
- Lee YX, Hurkmans PV, Arwert HJ, et al. Ethnic disparities in acute stroke presentation and reperfusion therapy in a Dutch comprehensive stroke center. medRxiv. 2026. doi:10.64898/2026.04.23.26351631
- Biswas R, Wijeratne T, Zelenak K, et al. Disparities in access to reperfusion therapy for acute ischemic stroke (DARTS): a comprehensive meta-analysis of ethnicity, socioeconomic status, and geographical factors. CNS Drugs. 2025;39(4):417–442. doi:10.1007/s40263-025-01161-z
- Ikeme S, Kottenmeier E, Uzochukwu G, Brinjikji W. Evidence-based disparities in stroke care metrics and outcomes in the United States: a systematic review. Stroke. 2022;53(3):670–679. doi:10.1161/STROKEAHA.121.036263
- Emmett ES, O’Connell MDL, Pei R, et al. Trends in ethnic disparities in stroke care and long-term outcomes. JAMA Network Open. 2025;8(1):e2453252. doi:10.1001/jamanetworkopen.2024.53252
- Coutinho JM, Klaver EC, Roos YB, Stam J, Nederkoorn PJ. Ethnicity and thrombolysis in ischemic stroke: a hospital based study in Amsterdam. BMC Neurology. 2011;11:81. doi:10.1186/1471-2377-11-81
- Almekhlafi MA, Goyal M, Dippel DWJ, et al. Healthy life-year costs of treatment speed from arrival to endovascular thrombectomy in patients with ischemic stroke: a meta-analysis of individual patient data from 7 randomized clinical trials. JAMA Neurology. 2021;78(6):709–717. doi:10.1001/jamaneurol.2021.1055
- Hackett ML, Pickles K. Part I: frequency of depression after stroke: an updated systematic review and meta-analysis of observational studies. International Journal of Stroke. 2014;9(8):1017–1025. doi:10.1111/ijs.12357