First-generation antipsychotic (FGA) shortages are not just pharmacy annoyances; they force medication changes in people who may have taken years to stabilize on a tolerable regimen.
Research Highlights
- 95,968 patients: Tabah 2026 used Komodo claims data to study people with psychotic-spectrum diagnoses taking shortage-affected first-generation antipsychotics.
- Switching was not evenly distributed: molindone reached 100% switching, trifluoperazine 55.2%, and thiothixene 32.8% after shortage or discontinuation events.
- Timing mattered: time to first switch ranged from 22 days to 103 days, suggesting some shortages translated into rapid clinical disruption while others unfolded more slowly.
- Clinical meaning: claims data show medication changes, not symptom relapse, but forced antipsychotic switching can be consequential because stable psychiatric maintenance regimens are hard-won.
- Policy read: older generic drugs with thin manufacturing redundancy can become weak links in serious-mental-illness care.
This study covered FDA-declared shortages and discontinuations between 2016 and 2023, so the 2026 claims-data estimate is a time-bounded snapshot.
Future shortages, manufacturers, and prescribing patterns could change the exact numbers, but the mechanism is durable: fragile generic supply chains can push psychiatric patients into medication switches they did not clinically choose.1
95,968 Claims Patients: What Tabah 2026 Measured
Tabah, Swamy, and English studied people with psychotic-spectrum diagnoses who were taking oral first-generation antipsychotics (FGAs; older dopamine-blocking antipsychotics such as haloperidol and perphenazine) affected by an FDA shortage or discontinuation. The source database was Komodo Healthcare Map, a large US claims dataset covering roughly 330 million lives.1
The design was pre-post and observational. It asked a concrete question: after a shortage or discontinuation event, how often did patients switch from the affected FGA to another antipsychotic?
- Population: 95,968 patients with psychotic-spectrum diagnoses.
- Drug class: oral first-generation antipsychotics, including haloperidol, perphenazine, thiothixene, trifluoperazine, and molindone.
- Exposure: FDA shortage or discontinuation events during 2016–2023.
- Outcome: antipsychotic switching during follow-up, measured through claims.
This is not the same as a relapse study. Claims can show that a medication changed; they cannot show whether the switch caused hallucinations, hospitalization, metabolic adverse effects, or loss of function.
Claims data are a notable limitation. They show prescription changes, not symptoms, adverse effects, hospitalization, or loss of function.
Still, in maintenance treatment for schizophrenia and chronic psychotic disorders, a forced switch is clinically meaningful because medication stability is often the product of multiple prior failures.
Molindone 100%, Trifluoperazine 55.2%, Thiothixene 32.8%
The strongest signal was the drug-by-drug split. Switching rates differed sharply, which suggests that some shortage events were much harder for the system to absorb than others.
Molindone was the extreme case: 100% of patients switched after effective discontinuation. That number should not be read as an ordinary temporary shortage effect; it reflects a drug disappearing from practical availability.
Trifluoperazine and thiothixene showed large shortage-linked switching: 55.2% and 32.8%, respectively. Those rates imply that substantial fractions of stable patients could not simply wait out the supply problem.1
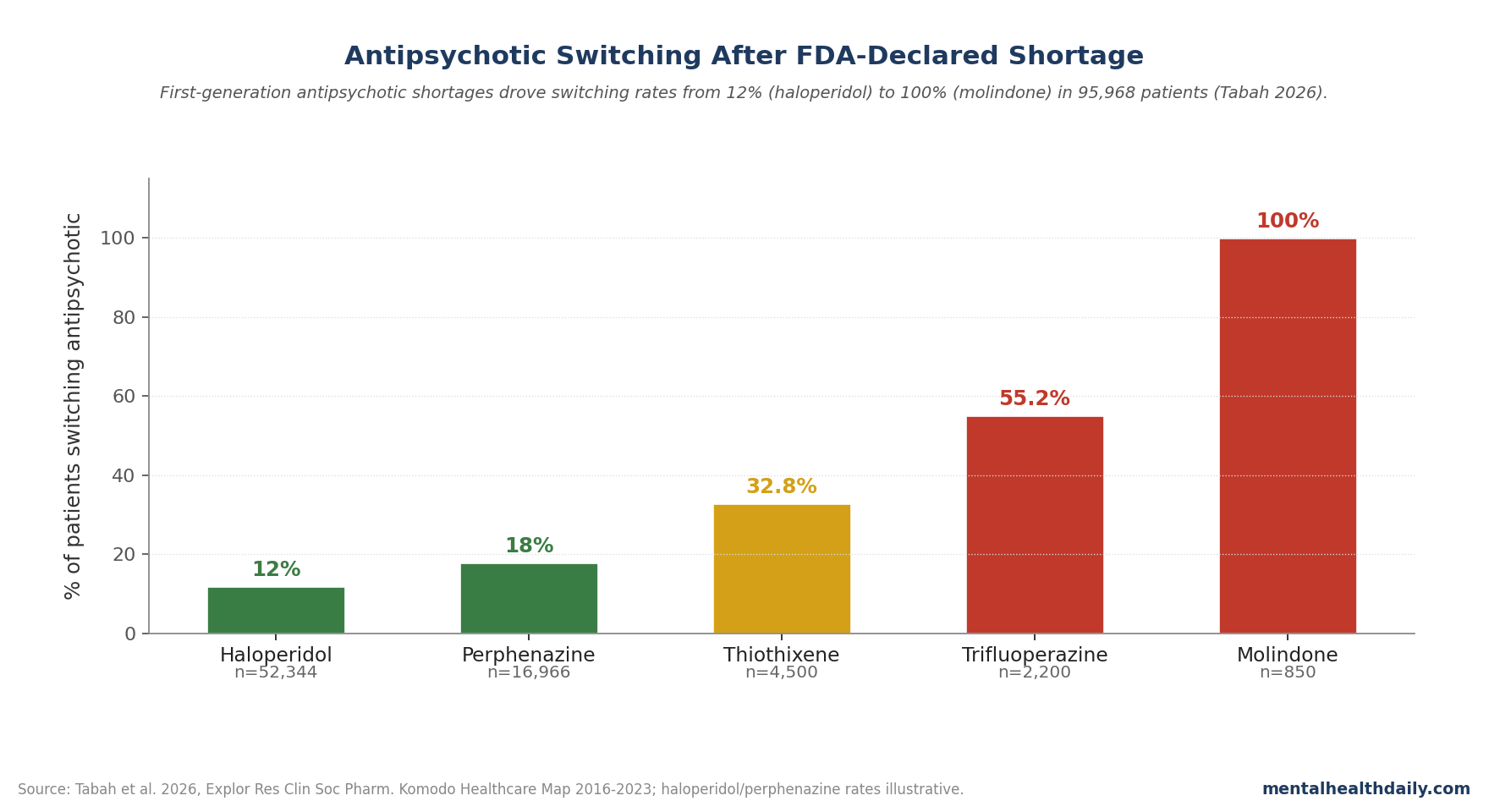
Haloperidol looked different. As the most common FGA in the cohort, it had more manufacturing and prescribing redundancy; a shortage from one manufacturer may be partly absorbed by other supply lines.
That difference is the policy lesson hiding inside the clinical data. A drug can be old, inexpensive, and medically useful while still being vulnerable if too few manufacturers keep producing it reliably.
Why Forced Antipsychotic Switching Is Clinically Different
In many medication classes, switching is annoying but usually straightforward. If one generic statin is temporarily unavailable, another statin can often cover the same clinical territory with modest adjustment.
Antipsychotics are not interchangeable in that way. Even drugs inside the same class differ in D2 receptor affinity, sedation, anticholinergic burden, metabolic risk, extrapyramidal symptoms, prolactin effects, and patient-specific tolerability.2
Three practical problems make forced switching especially risky:
- Prior failures matter. A patient stable on perphenazine or haloperidol may already have failed several alternatives.
- Cross-titration is not neutral. Dose overlap, taper speed, and receptor-profile mismatch can produce withdrawal symptoms, side effects, or symptom return.
- Stability has value. For chronic psychotic disorders, staying well can mean avoiding hospitalization, preserving housing, keeping work, and maintaining relationships.
First-generation and second-generation antipsychotics also carry different risk profiles. FGAs tend to carry higher extrapyramidal and tardive dyskinesia risk; second-generation agents often carry greater metabolic burden.3
So a forced switch from a tolerated FGA to a second-generation alternative is not simply a lateral move. It can trade one long-term harm profile for another.
Treatment-Resistant Schizophrenia Raises the Stakes
Treatment-resistant schizophrenia (TRS) is usually defined by persistent symptoms despite adequate trials of at least 2 antipsychotics. Roughly one-third of people with schizophrenia meet some version of this clinical problem over the illness course.4
For these patients, medication history is not background detail. It is the map of what already failed, what was intolerable, and what finally worked.
The shortage problem is therefore asymmetric: the patient taking a common drug with many acceptable substitutes has a different risk profile than the patient who stabilized only after years of trial and error. Claims data cannot identify every one of those histories, but the clinical logic is hard to ignore.
This is where supply-chain policy and psychiatric outcomes meet. A national shortage is experienced locally as a prescriber trying to decide whether to stretch a partial refill, locate another pharmacy, bridge with a different formulation, or initiate a switch that the patient never needed medically.
Generic Supply Chains Are a Psychiatric-Care Problem
FGA shortages fit a broader drug-shortage pattern: older generics often have thin profit margins, limited manufacturer redundancy, and fragile production capacity. A quality-control problem at one site can become a national treatment problem if few other suppliers can compensate.5
For psychiatric medications, that fragility is easy to underestimate because the drugs are old. Age does not mean dispensability. A 40-year-old antipsychotic can still be the specific medication keeping someone stable.
Useful prevention would be boring but meaningful:
- Redundant manufacturing: essential psychiatric generics should not depend on a tiny number of active producers.
- Earlier shortage alerts: prescribers and pharmacies need time to plan before forced switching becomes urgent.
- Psychiatric stockpiling rules: stable maintenance patients may need more refill flexibility than standard insurance rules allow.
- Drug-specific contingency plans: high-risk drugs such as less-common FGAs need pre-identified alternatives before shortage pressure hits.
None of this is glamorous. It is the infrastructure side of serious mental illness care, and the Tabah analysis shows why it belongs in the clinical conversation.
What the Claims Data Cannot Settle
Clinical outcomes are missing. The study does not show whether patients who switched relapsed, went to the emergency department, were hospitalized, or developed new adverse effects.
Switching reasons are inferred. A switch after a shortage event is plausibly shortage-related, but claims data cannot fully separate shortage pressure from ordinary clinical switching.
Adherence filtering shapes the cohort. The analysis focused on patients with at least 80% medication possession ratio, which captures a relatively adherent group. Patients with inconsistent access or lower baseline adherence may experience shortages differently.
Oral FGAs are only one part of the system. Long-acting injectables, second-generation antipsychotics, pharmacy-level substitution rules, and regional supply variation all need separate analysis.
The claims support a narrower conclusion: FDA shortage and discontinuation events were followed by substantial drug switching in a large psychosis-treated cohort. Relapse, hospitalization, and adverse-effect outcomes still need direct study.
Questions About Antipsychotic Shortages
How often do antipsychotic shortages occur?
In this analysis, every oral FGA had at least one manufacturer shortage during 2016–2023. That does not mean every patient lost access, but it shows that the class was repeatedly exposed to supply disruption.
What should patients do if their antipsychotic is in shortage?
Contact the prescriber quickly and do not stop abruptly. Antipsychotic discontinuation or rushed switching can be clinically risky, especially for patients with chronic psychotic disorders.
Are first-generation antipsychotics still important?
Yes. They remain useful for some patients because efficacy, side effects, cost, prior treatment failures, and individual tolerability do not line up the same way for everyone.
Does this prove shortages caused psychiatric relapse?
No. The study measured prescription switching in claims data, not symptoms. The clinical concern is that switching stable antipsychotic regimens can create relapse risk, not that this dataset directly measured relapse.
Why add 2026 to the slug?
Because this post is anchored to a 2026 claims-data paper using a defined 2016–2023 shortage window. Future claims analyses may produce different estimates, so the year has search and interpretation value here.
References
- Tabah A, Swamy N, English C. First-generation antipsychotic shortages in the United States: utilization and switching patterns. Explor Res Clin Soc Pharm. 2026;22:100725. doi:10.1016/j.rcsop.2026.100725
- Leucht S et al. Comparative efficacy and tolerability of 32 oral antipsychotics for the acute treatment of adults with multi-episode schizophrenia: a systematic review and network meta-analysis. Lancet. 2019;394(10202):939-951. doi:10.1016/s0140-6736(19)31135-3
- Huhn M et al. Comparative efficacy and tolerability of antipsychotics in schizophrenia. Lancet. 2019. doi:10.1016/s0140-6736(19)31135-3
- Howes OD et al. Treatment-resistant schizophrenia: TRRIP working group consensus guidelines. Am J Psychiatry. 2017;174:216-229. doi:10.1176/appi.ajp.2016.16050503
- Fox ER et al. Drug shortages: a complex health care crisis. Mayo Clin Proc. 2014;89(3):361-373. doi:10.1016/j.mayocp.2013.11.014
